Abstract
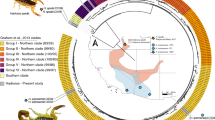
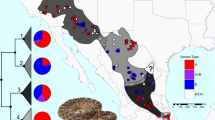
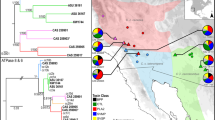
Snake venom can vary both among and within species. While some groups of New World pitvipers—such as rattlesnakes—have been well studied, very little is known about the venom of montane pitvipers (Cerrophidion) found across the Mesoamerican highlands. Compared to most well-studied rattlesnakes, which are widely distributed, the isolated montane populations of Cerrophidion may facilitate unique evolutionary trajectories and venom differentiation. Here, we describe the venom gland transcriptomes for populations of C. petlalcalensis, C. tzotzilorum, and C. godmani from Mexico, and a single individual of C. sasai from Costa Rica. We explore gene expression variation in Cerrophidion and sequence evolution of toxins within C. godmani specifically. Cerrophidion venom gland transcriptomes are composed primarily of snake venom metalloproteinases, phospholipase A\(_2\)s (PLA\(_2\)s), and snake venom serine proteases. Cerrophidion petlalcalensis shows little intraspecific variation; however, C. godmani and C. tzotzilorum differ significantly between geographically isolated populations. Interestingly, intraspecific variation was mostly attributed to expression variation as we did not detect signals of selection within C. godmani toxins. Additionally, we found PLA\(_2\)-like myotoxins in all species except C. petlalcalensis, and crotoxin-like PLA\(_2\)s in the southern population of C. godmani. Our results demonstrate significant intraspecific venom variation within C. godmani and C. tzotzilorum. The toxins of C. godmani show little evidence of directional selection where variation in toxin sequence is consistent with evolution under a model of mutation–drift equilibrium. Cerrophidion godmani individuals from the southern population may exhibit neurotoxic venom activity given the presence of crotoxin-like PLA\(_2\)s; however, further research is required to confirm this hypothesis.
Resumen
El veneno de las serpientes puede variar entre y dentro de las especies. Mientras algunos grupos de viperidos del Nuevo Mundo—como las cascabeles—han sido bien estudiadas, muy poco se sabe acerca del veneno de las nauyacas de frío (Cerrophidion) que se encuentran en las zonas altas de Mesoamérica. Comparadas con las extensamente estudiadas cascabeles, que estan ampliamente distribuidas, las poblaciones de Cerrophidion, aisladas en montañas, pueden poseer trayectorias evolutivas y diferenciación en su veneno unicos. En el presente trabajo, describimos el transcriptoma de las glándulas de veneno de poblaciones de C. petlalcalensis, C. tzotzilorum, y C. godmani de México, y un individuo de C. sasai de Costa Rica. Exploramos la variación en la expresión de toxinas en Cerrophidion y la evolución en las secuencias geneticas en C. godmani específicamente. El transcriptoma de la glándula de veneno de Cerrophidion esta compuesto principalmente de Metaloproteinasas de Veneno de Serpiente, Fosfolipasas A\(_2\) (PLA\(_2\)s), y Serin Proteasas de Veneno de Serpiente. Cerrophidion petlalcalensis presenta poca variación intraespecífica; sin embargo, los transcriptomas de la glandula de veneno de C. godmani y C. tzotzilorum difieren significativamente entre poblaciones geográficamente aisladas. Curiosamente, la variación intraespecífica estuvo atribuida principalmente a la expresión de las toxinas ya que no encontramos señales de selección en las toxinas de C. godmani. Adicionalmente, encontramos miotoxinas similares a PLA\(_2\) en todas las especies excepto C. petlalcalensis, y PLA\(_2\)s similares a crotoxina en la población sureña de C. godmani. Nuestros resultados demuestran la presencia de variacion intraespecífica presente en el veneno de C. godmani y C. tzotzilorum. Las toxinas de Cerrophidion godmani muestran poca evidencia de selección direccional, y la variación en la secuencias de las toxinas es consistente con evolucion bajo un modelo de equilibrio de mutación-deriva. Algunos individuos de C. godmani de la población del sur potencialmente tienen un veneno neurotóxico dada la presencia de PLA\(_2\)s similares a la crotoxina, sin embargo, se necesita más evidencia para corroborar esta hipótesis.






Similar content being viewed by others
Data Availability
The raw data-sets generated and analyzed during the current study are available in the National Center for Biotechnology Information (NCBI) repository under the accession numbers SRR19227107, SRR19227106, SRR19227101, SRR19227100, SRR19227099, SRR19227098, SRR19227097, SRR19227096, SRR19227095, SRR19227094, SRR19227105, SRR19227104, SRR19227103, and SRR19227102. The consensus transcriptomes generated and analyzed during the current study are available at the github repository https://github.com/RamsesRosales/Cerrophidion_Selection, a similar version is available at the DDBJ/EMBL/GenBank under the accession numbers: C. sasai GJZX00000000, the version described in this paper is the first version, GJZX01000000; C. petlalcalensis GKAV00000000, the version described in this paper is the first version, GKAV01000000; C. godmani GKCA00000000, the version described in this paper is the first version, GKCA01000000. C. tzotzilorum GKIB00000000, the version described in this paper is the first version, GKIB01000000. Scripts utilized in this work can be found in the online resources 3 and 4, and in the github repositories: https://github.com/RamsesRosales/Cerrophidion_Selection, https://github.com/RamsesRosales/ModelCrotA.
References
Aird SD, Aggarwal S, Villar-Briones A, Tin MMY, Terada K, Mikheyev AS (2015) Snake venoms are integrated systems, but abundant venom proteins evolve more rapidly. BMC Genom 16:1–20. https://doi.org/10.1186/s12864-015-1832-6
Aird SD, Arora J, Barua A, Qiu L, Terada K, Mikheyev AS (2017) Population genomic analysis of a pitviper reveals microevolutionary forces underlying venom chemistry. Genome Biol Evol 9:2640–2649
Aird SD, Watanabe Y, Villar-briones A, Roy MC, Terada K, Mikheyev AS (2013) Quantitative high-throughput profiling of snake venom gland transcriptomes and proteomes (Ovophis okinavensis and Protobothrops flavoviridis). BMC Genom 14:1–26
Almeida DD, Viala VL, Nachtigall PG, Broe M, Gibbs HL, Serrano SMdT, Moura-da Silva AM, Ho PL, Nishiyama-Jr MY, Junqueira-de Azevedo ILM (2021) Tracking the recruitment and evolution of snake toxins using the evolutionary context provided by the Bothrops jararaca genome. Proc Natl Acad Sci 118:e2015159118. https://doi.org/10.1073/pnas.2015159118
Amazonas DR, Portes-Junior JA, Nishiyama-Jr MY, Nicolau CA, Chalkidis HM, Mourão RH, Grazziotin FG, Rokyta DR, Gibbs HL, Valente RH, Junqueira-de Azevedo IL, Moura-da Silva AM (2018) Molecular mechanisms underlying intraspecific variation in snake venom. J Proteom 181:60–72. https://doi.org/10.1016/j.jprot.2018.03.032
Arni RK, Fontes MR, Barberato C, Gutiérrez JM, Díaz C, Ward RJ (1999) Crystal structure of myotoxin II, a monomeric Lys49-phospholipase A\(_2\) homologue isolated from the venom of Cerrophidion (Bothrops) godmani. Arch Biochem Biophys 366:177–182
Auwera GA, Carneiro MO, Hartl C, Poplin R, del Angel G, Levy-Moonshine A, Jordan T, Shakir K, Roazen D, Thibault J, Banks E, Garimella KV, Altshuler D, Gabriel S, DePristo MA (2013) From fastQ data to high-confidence variant calls: the genome analysis toolkit best practices pipeline. Curr Protocols Bioinfo 43:1–33. https://doi.org/10.1002/0471250953.bi1110s43
Junqueira-de Azevedo IDL, Ho PL (2002) A survey of gene expression and diversity in the venom glands of the pitviper snake Bothrops insularis through the generation of expressed sequence tags (ESTs). Gene 299:279–291
Borja M, Neri-Castro E, Castañeda-Gaytán G, Strickland JL, Parkinson CL, Castañeda-Gaytán J, Ponce-López R, Lomonte B, Olvera-Rodríguez A, Alagón A, Pérez-Morales R (2018) Biological and proteolytic variation in the venom of Crotalus scutulatus scutulatus from Mexico. Toxins 10:1–19
Brahma RK, McCleary RJ, Kini RM, Doley R (2015) Venom gland transcriptomics for identifying, cataloging, and characterizing venom proteins in snakes. Toxicon 93:1–10. https://doi.org/10.1016/j.toxicon.2014.10.022
Calvete JJ (2017) Venomics: integrative venom proteomics and beyond. Biochem J 474:611–634
Campbell JA, Lamar WW (2004) Venomous reptiles of the western hemisphere, vol 1. Cornell University Press, Ithaca
Campbell JA, Solórzano A (1992) The distribution, variation, and natural history of the middle American montane pitviper Porthidium godmani. In: Campbell JA, Brodie ED (eds) Biology of the pitvipers. Selva, Tyler, pp 223–250
Capella-Gutiérrez S, Silla-Martínez JM, Gabaldón T (2009) Trimal: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics. https://doi.org/10.1093/bioinformatics/btp348
Casewell NR, Huttley GA, Wüster W (2012) Dynamic evolution of venom proteins in squamate reptiles. Nat Commun 3:1066
Casewell NR, Wagstaff SC, Harrison RA, Renjifo C, Wuster W (2011) Domain loss facilitates accelerated evolution and neofunctionalization of duplicate snake venom metalloproteinase toxin genes. Mol Biol Evol 28:2637–2649. https://doi.org/10.1093/molbev/msr091
Casewell NR, Wagstaff SC, Wüster W, Cook DA, Bolton FM, King SI, Pla D, Sanz L, Calvete JJ, Harrison RA (2014) Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. Proc Natl Acad Sci USA 111:9205–9210. https://doi.org/10.1073/pnas.1405484111
Casewell NR, Wüster W, Vonk FJ, Harrison RA, Fry BG (2013) Complex cocktails: the evolutionary novelty of venoms. Trends Ecol Evol 28:219–229
Castoe TA, Daza JM, Smith EN, Sasa MM, Kuch U, Campbell JA, Chippindale PT, Parkinson CL (2009) Comparative phylogeography of pitvipers suggests a consensus of ancient Middle American highland biogeography. J Biogeogr 36:88–103. https://doi.org/10.1111/j.1365-2699.2008.01991.x
Chen YH, Wang YM, Hseu MJ, Tsai IH (2004) Molecular evolution and structure-function relationships of crotoxin-like and asparagine-6-containing phospholipases A\(_2\) in pit viper venoms. Biochem J 381:25–34
Chernomor O, von Haeseler A, Minh BQ (2016) Terrace aware data structure for phylogenomic inference from supermatrices. System Biol 65:997–1008. https://doi.org/10.1093/sysbio/syw037
Cingolani P, Platts A, Coon M, Nguyen T, Wang L, Land S, Lu X, Ruden D (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain \(W^{118}\); iso-2; iso-3. Fly 6:80–92.
Cock PJ, Antao T, Chang JT, Chapman BA, Cox CJ, Dalke A, Friedberg I, Hamelryck T, Kauff F, Wilczynski B et al (2009) Biopython: freely available python tools for computational molecular biology and bioinformatics. Bioinformatics 25:1422–1423. https://doi.org/10.1093/bioinformatics/btp163
Danecek P, Auton A, Abecasis G, Albers CA, Banks E, DePristo MA, Handsaker RE, Lunter G, Marth GT, Sherry ST, McVean G, Durbin R (2011) The variant call format and VCF tools. Bioinformatics. https://doi.org/10.1093/bioinformatics/btr330
Daza JM, Castoe TA, Parkinson CL (2010) Using regional comparative phylogeographic data from snake lineages to infer historical processes in Middle America. Ecography 33:343–354. https://doi.org/10.1111/j.1600-0587.2010.06281.x
Dowell NL, Giorgianni MW, Griffin S, Kassner VA, Selegue JE, Sanchez EE, Carroll SB (2018) Extremely divergent haplotypes in two toxin gene complexes encode alternative venom types within rattlesnake species. Curr Biol 28:1016–1026
Dowell NL, Giorgianni MW, Kassner VA, Selegue JE, Sanchez EE, Carroll SB (2016) The deep origin and recent loss of venom toxin genes in rattlesnakes. Curr Biol 26:2434–2445
Durban J, Juárez P, Angulo Y, Lomonte B, Flores-Diaz M, Alape-Girón A, Sasa M, Sanz L, Gutiérrez JM, Dopazo J, Conesa A, Calvete JJ (2011) Profiling the venom gland transcriptomes of Costa Rican snakes by 454 pyrosequencing. BMC Genom. https://doi.org/10.1186/1471-2164-12-259
Eswar N, Webb B, Marti-Renom MA, Madhusudhan M, Eramian D, My Shen, Pieper U, Sali A (2006) Comparative protein structure modeling using modeller. Curr Protocols Bioinfo 15:1–30. https://doi.org/10.1002/0471250953.bi0506s15
Faure G, Guillaume JL, Camoin L, Saliou B, Bon C (1991) Multiplicity of acidic subunit isoforms of crotoxin, the phospholipase A\(_2\) neurotoxin from Crotalus durissus terrificus venom, results from posttranslational modifications. Biochemistry 30:8074–8083
Fernández M, Martínez-Fonseca J, Salazar-Saavedra M, Gutiérrez L, Loza J, Sunyer J (2017) First verified record of Cerrophidion wilsoni (Reptilia: Squamata: Viperidae) from Nicaragua. Mesoamerican Herpetol 4:481–484
Fox JW, Serrano SM (2005) Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon 45:969–985
Fraczkiewicz R, Braun W (1998) Exact and efficient analytical calculation of the accessible surface areas and their gradients for macromolecules. J Comput Chem 19:319–333
Franco-Servín C, Neri-Castro E, Bénard-Valle M, Alagón A, Rosales-García RA, Guerrero-Alba R, Poblano-Sánchez JE, Silva-Briano M, Guerrero-Barrera AL, Sigala-Rodríguez JJ (2021) Biological and biochemical characterization of Coronado Island Rattlesnake (Crotalus helleri caliginis) venom and antivenom neutralization. Toxins 13:582
Fraser DJ, Weir LK, Bernatchez L, Hansen MM, Taylor EB (2011) Extent and scale of local adaptation in salmonid fishes: review and meta-analysis. Heredity 106:404–420
Fu L, Niu B, Zhu Z, Wu S, Li W (2012) CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics 28:3150–3152. https://doi.org/10.1093/bioinformatics/bts565
Gasteiger E, Hoogland C, Gattiker A, Duvaud S, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server. In: Walker JM (ed) The proteomics protocols handbook. Humana Press, Totowa, pp 571–607. https://doi.org/10.1385/1-59259-890-0:571
Gené J, Roy A, Rojas G, Gutiérrez J, Cerdas L (1989) Comparative study on coagulant, defibrinating, fibrinolytic and fibrinogenolytic activities of Costa Rican crotaline snake venoms and their neutralization by a polyvalent antivenom. Toxicon 27:841–848
Gibbs HL, Sanz L, Pérez A, Ochoa A, Hassinger AT, Holding ML, Calvete JJ (2020) The molecular basis of venom resistance in a rattlesnake-squirrel predator-prey system. Mol Ecol 29:2871–2888. https://doi.org/10.1111/mec.15529
Glenn J, Straight R, Wolt T (1994) Regional variation in the presence of canebrake toxin in Crotalus horridus venom. Compar Biochem Physiol C 107:337–346
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652
Gutiérrez J (2014) Current challenges for confronting the public health problem of snakebite envenoming in Central America. J Venom Anim Toxins Including Trop Dis 20:7. https://doi.org/10.1186/1678-9199-20-7
Gutiérrez JM, Gené JA, Rojas G, Cerdas L (1985) Neutralization of proteolytic and hemorrhagic activities of Costa Rican snake venoms by a polyvalent antivenom. Toxicon 23(6):887–893
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS (2018) UFBoot2: improving the ultrafast bootstrap approximation. Mol Biol Evol 35:518–522
Hofmann EP, Rautsaw RM, Strickland JL, Holding ML, Hogan MP, Mason AJ, Rokyta DR, Parkinson CL (2018) Comparative venom-gland transcriptomics and venom proteomics of four Sidewinder Rattlesnake (Crotalus cerastes) lineages reveal little differential expression despite individual variation. Sci Rep 8:15534. https://doi.org/10.1038/s41598-018-33943-5
Holding ML, Margres MJ, Mason AJ, Parkinson CL, Rokyta DR (2018) Evaluating the performance of de novo assembly methods for venom-gland transcriptomics. Toxins 10:1–21
Holding ML, Sovic MG, Colston TJ, Gibbs HL (2021) The scales of coevolution: comparative phylogeography and genetic demography of a locally adapted venomous predator and its prey. Biol J Linnean Soc 132:297–317
Holding ML, Strickland JL, Rautsaw RM, Hofmann EP, Mason AJ, Hogan MP, Nystrom GS, Ellsworth SA, Colston TJ, Borja M, Castañeda-Gaytán G, Grünwald CI, Jones JM, Freitas-de Sousa LA, Viala VL, Margres MJ, Hingst-Zaher E, Junqueira-de Azevedo ILM, Moura-da Silva AM, Grazziotin FG, Gibbs HL, Rokyta DR, Parkinson CL (2021) Phylogenetically diverse diets favor more complex venoms in North American pitvipers. Proc Natl Acad Sci 118:e2015579118. https://doi.org/10.1073/pnas.2015579118
Jadin RC, Townsend JH, Castoe TA, Campbell JA (2012) Cryptic diversity in disjunct populations of Middle American montane pitvipers: a systematic reassessment of Cerrophidion godmani. Zoologica Scripta 41:455–470
Jones L, Youngman NJ, Neri-Castro E, Guadarrama-Martínez A, Lewin MR, Carter R, Frank N, Fry BG (2022) Differential antivenom and small-molecule inhibition of novel Coagulotoxic variations in atropoides, Cerrophidion, Metlapilcoatlus, and Porthidium American viperid snake venoms. Toxins 14. https://www.mdpi.com/2072-6651/14/8/511
Kalyaanamoorthy S, Minh BQ, Wong TKF, von Haeseler A, Jermiin LS (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nat Methods 14:587–589
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol. https://doi.org/10.1093/molbev/mst010
Keil B (1992) Specificity of proteolysis. Springer, Berlin, Heidelberg
Kolde R (2019) pheatmap: pretty heatmaps. https://CRAN.R-project.org/package=pheatmap. R package version 1.0.12
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinfo 12:323
Li H (2013) Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. 00:1–3. arXiv:1303.3997
Lizano S, Angulo Y, Lomonte B, Fox JW, Lambeau G, Lazdunski M, Gutiérrez JM (2000) Two phospholipase A\(_2\) inhibitors from the plasma of Cerrophidion (Bothrops) godmani which selectively inhibit two different group-II phospholipase A\(_2\) myotoxins from its own venom: isolation, molecular cloning and biological properties. Biochem J 346:631
Lomonte B, Fernández J, Sanz L, Angulo Y, Sasa M, Gutiérrez JM, Calvete JJ (2014) Venomous snakes of Costa Rica: biological and medical implications of their venom proteomic profiles analyzed through the strategy of snake venomics. J Proteom 105:323–339. https://doi.org/10.1016/j.jprot.2014.02.020
Lomonte B, Rey-Suárez P, Tsai WC, Angulo Y, Sasa M, Gutiérrez JM, Calvete JJ (2012) Snake venomics of the pit vipers Porthidium nasutum, Porthidium ophryomegas, and Cerrophidion godmani from Costa Rica: Toxicological and taxonomical insights. J Proteom 75:1675–1689
López-Luna MA, Vogt RC, De la Torre-Loranca MA (1999) A new species of montane pitviper from Veracruz, México. Herpetologica 55:382–389
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with deseq2. Genome Biol 15:550
Mackessy SP (2008) Venom composition in rattlesnakes: trends and biological significance. In: Hayes WK, Beaman KR, Cardwell MD, Bush SP (eds) The biology of rattlesnakes. Loma Linda University Press, Loma Linda, pp 495–510
Mai U, Mirarab S (2018) Treeshrink: fast and accurate detection of outlier long branches in collections of phylogenetic trees. BMC Genom 19. https://bmcgenomics.biomedcentral.com/articles/10.1186/s12864-018-4620-2
Margres MJ, McGivern JJ, Seavy M, Wray KP, Facente J, Rokyta DR (2014) Contrasting modes and tempos of venom expression evolution in two snake species. Genetics 199:165–176. https://doi.org/10.1534/genetics.114.172437
Margres MJ, McGivern JJ, Wray KP, Seavy M, Calvin K, Rokyta DR (2014) Linking the transcriptome and proteome to characterize the venom of the eastern diamondback rattlesnake (Crotalus adamanteus). J Proteom 96:145–158. https://doi.org/10.1016/j.jprot.2013.11.001
Margres MJ, Patton A, Wray KP, Hassinger ATB, Ward MJ, Lemmon EM, Lemmon AR, Rokyta DR (2019) Tipping the scales: the migration-selection balance leans toward selection in snake venoms. Mol Biol Evol 36:271–282
Margres MJ, Wray KP, Hassinger ATB, Ward MJ, McGivern JJ, Moriarty Lemmon E, Lemmon AR, Rokyta DR (2017) Quantity, not quality: rapid adaptation in a polygenic trait proceeded exclusively through expression differentiation. Mol Biol Evol 34:3099–3110
Margres MJ, Wray KP, Sanader D, McDonald PJ, Trumbull LM, Patton AH, Rokyta DR (2021) Varying intensities of introgression obscure incipient venom-associated speciation in the timber rattlesnake (Crotalus horridus). Toxins 13:782
Margres MJ, Wray KP, Seavy M, McGivern JJ, Herrera ND, Rokyta DR (2016) Expression differentiation is constrained to low-expression proteins over ecological timescales. Genetics 202:273–283
Martin M, Patterson M, Garg S, Fischer SO, Pisanti N, Klau G, Schöenhuth A, Marschall T (2016) Whatshap: fast and accurate read-based phasing. bioRxiv P. 085050. https://www.biorxiv.org/content/10.1101/085050v2
Mason AJ, Holding ML, Rautsaw RM, Rokyta DR, Parkinson CL, Gibbs HL (2022) Venom gene sequence diversity and expression jointly shape diet adaptation in pitvipers. Mol Biol Evol 39:1–14
Mason AJ, Margres MJ, Strickland JL, Rokyta DR, Sasa M, Parkinson CL (2020) Trait differentiation and modular toxin expression in palm-pitvipers. BMC Genom 21:147
Murrell B, Weaver S, Smith MD, Wertheim JO, Murrell S, Aylward A, Eren K, Pollner T, Martin DP, Smith DM, Scheffler K, Pond SLK (2015) Gene-wide identification of episodic selection. Mol Biol Evol 32:1365–1371
Nachtigall PG, Rautsaw RM, Ellsworth SA, Mason AJ, Rokyta DR, Parkinson CL, de Azevedo ILMJ (2021) Toxcodan: a new toxin annotator and guide to venom gland transcriptomics. Brief Bioinfo. https://doi.org/10.1093/bib/bbab095/6235957
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci 76:5269–5273
Neri-Castro E, Bénard-Valle M, Gil G, Borja M, de León JL, Alagón A (2020) Venomous snakes in Mexico: a review of the study of venoms, antivenom and epidemiology. Revista Latinoamericana de Herpetologia 3:05–22
Neri-Castro E, Hernández-Dávila A, Olvera-Rodríguez A, Cardoso-Torres H, Bénard-Valle M, Bastiaans E, López-Gutierrez O, Alagón A (2019) Detection and quantification of a \(\beta\)-neurotoxin (crotoxin homologs) in the venom of the rattlesnakes Crotalus simus, C. culminatus and C. tzabcan from Mexico. Toxicon: X 2:100007. https://doi.org/10.1016/j.toxcx.2019.100007
Neri-Castro E, Sanz L, Olvera-Rodríguez A, Bénard-Valle M, Alagón A, Calvete JJ (2020) Venomics and biochemical analysis of the black-tailed horned pitviper, Mixcoatlus melanurus, and characterization of Melanurutoxin, a novel crotoxin homolog. J Proteom 225:103865
Nguyen LT, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–274
Nielsen R (2005) Molecular signatures of natural selection. Annu Rev Genet 39:197–218
Ochoa A, Broe M, Moriarty Lemmon E, Lemmon AR, Rokyta DR, Gibbs HL (2020) Drift, selection and adaptive variation in small populations of a threatened rattlesnake. Mol Ecol 29:2612–2625
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528
Petersen TN, Brunak S, Von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786
Pond SLK, Poon AFY, Velazquez R, Weaver S, Hepler NL, Murrell B, Shank SD, Magalis BR, Bouvier D, Nekrutenko A, Wisotsky S, Spielman SJ, Frost SDW, Muse SV (2020) Hyphy 2.5–a customizable platform for evolutionary hypothesis testing using phylogenies. Mol Biol Evol 37:295–299
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Radvanyi FR, Bon C (1982) Catalytic activity and reactivity with p-bromophenacyl bromide of the phospholipase subunit of crotoxin. influence of dimerization and association with the noncatalytic subunit. J Biol Chem 257:12616–12623
Rautsaw RM, Hofmann EP, Margres MJ, Holding ML, Strickland JL, Mason AJ, Rokyta DR, Parkinson CL (2019) Intraspecific sequence and gene expression variation contribute little to venom diversity in sidewinder rattlesnakes (Crotalus cerastes). Proc R Soc B: Biol Sci 286:20190810
Rautsaw RM, Jiménez-Velázquez G, Hofmann EP, Alencar LRV, Grünwald CI, Martins M, Carrasco P, Doan TM, Parkinson CL (2022) VenomMaps: Updated species distribution maps and models for New World pitvipers (Viperidae: Crotalinae). Sci Data 9:232
Robinson MD, McCarthy DJ, Smyth GK (2010) Edger: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140. https://doi.org/10.1093/bioinformatics/btp616
Rokyta DR, Lemmon AR, Margres MJ, Aronow K (2012) The venom-gland transcriptome of the eastern diamondback rattlesnake (Crotalus adamanteus). BMC Genom 13:312
Rokyta DR, Margres MJ, Calvin K (2015) Post-transcriptional mechanisms contribute little to phenotypic variation in snake venoms. G3 Genes, Genomes, Genet 5:2375–2382
Rokyta DR, Wray KP, Lemmon AR, Lemmon EM, Caudle SB (2011) A high-throughput venom-gland transcriptome for the Eastern Diamondback Rattlesnake (Crotalus adamanteus) and evidence for pervasive positive selection across toxin classes. Toxicon 57:657–671
Rokyta DR, Wray KP, Margres MJ (2013) The genesis of an exceptionally lethal venom in the timber rattlesnake (Crotalus horridus) revealed through comparative venom-gland transcriptomics. BMC Genom 14:394
Rokyta DR, Wray KP, McGivern JJ, Margres MJ (2015) The transcriptomic and proteomic basis for the evolution of a novel venom phenotype within the timber rattlesnake (Crotalus horridus). Toxicon 98:34–48. https://doi.org/10.1016/j.toxicon.2015.02.015
Rotenberg D, Bamberger ES, Kochva E (1971) Studies on ribonucleic acid synthesis in the venom glands of Vipera palaestinae (Ophidia, Reptilia). Biochem J 121:609–612
Rübsamen K, Breithaupt H, Habermann B (1971) Biochemistry and pharmacology of the crotoxin complex. Naunyn-Schmiedebergs Arch. Pharmak 270:274–288. https://doi.org/10.1007/BF00997027
Sasa M (1997) Cerrophidion godmani in Costa Rica: a case of extremely low allozyme variation? J Herpetol 31:569
Saviola AJ, Pla D, Sanz L, Castoe TA, Calvete JJ, Mackessy SP (2015) Comparative venomics of the Prairie Rattlesnake (Crotalus viridis viridis) from Colorado: Identification of a novel pattern of ontogenetic changes in venom composition and assessment of the immunoreactivity of the commercial antivenom CroFab®. J Proteom 121:28–43
Schield DR, Perry BW, Adams RH, Holding ML, Nikolakis ZL, Gopalan SS, Smith CF, Parker JM, Meik JM, DeGiorgio M, Mackessy SP, Castoe TA (2022) The roles of balancing selection and recombination in the evolution of rattlesnake venom. Nat Ecol Evol. https://www.nature.com/articles/s41559-022-01829-5
Schramer TD, de la Torre-Loranca MA, Salazar-Saavedra M, Kalki Y, Wylie DB (2018) Addiotional diet information for Cerrophidion godmani (Günther, 1863) and a dietary synopsis of the genus Cerrophidion Campbell and Lamar, 1992. Revista Latinoamericana de Herpetología 1:35
Sievers F, Higgins DG (2018) Clustal Omega for making accurate alignments of many protein sequences. Protein Sci 27:135–145
Simão FA, Waterhouse RM, Ioannidis P, Kriventseva EV, Zdobnov EM (2015) BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31:3210–3212. https://doi.org/10.1093/bioinformatics/btv351
Strickland JL, Mason AJ, Rokyta DR, Parkinson CL (2018) Phenotypic variation in Mojave rattlesnake (Crotalus scutulatus) venom is driven by four toxin families. Toxins 10:1–23
Strickland JL, Smith CF, Mason AJ, Schield DR, Borja M, Castañeda-Gaytán G, Spencer CL, Smith LL, Trápaga A, Bouzid NM, Campillo-García G, Flores-Villela OA, Antonio-Rangel D, Mackessy SP, Castoe TA, Rokyta DR, Parkinson CL (2018) Evidence for divergent patterns of local selection driving venom variation in Mojave Rattlesnakes (Crotalus scutulatus). Sci Rep 8:17622
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Tange O (2020) GNU Parallel 20201122 (’Biden’). GNU Parallel is a general parallelizer to run multiple serial command line programs in parallel without changing them. https://doi.org/10.5281/zenodo.4284075
Tasoulis T, Isbister G (2017) A review and database of snake venom proteomes. Toxins 9:290
Torre-Loranca MAL, Grünwald CI, Valdenegro-Brito AE, Cervantes-Burgos RI, García-Vázquez UO (2019) New distributional records for the rare Mexican pitviper Cerrophidion petlalcalensis (Squamata: Viperidae) from Veracruz and Oaxaca, Mexico, with comments on distribution and the natural history. Revista Latinoamericana de Herpetologia 2:71–77
Tumescheit C, Firth AE, Brown K (2020) Cialign—a highly customisable command line tool to clean, interpret and visualise multiple sequence alignments. bioRxiv. https://doi.org/10.1101/2020.09.14.291484
Whittington AC, Mason AJ, Rokyta DR (2018) A single mutation unlocks cascading exaptations in the origin of a potent pitviper neurotoxin. Mol Biol Evol 35:887–898
Wooldridge BJ, Pineda G, Banuelas-Ornelas JJ, Dagda RK, Gasanov SE, Rael ED, Lieb CS (2001) Mojave rattlesnakes (Crotalus scutulatus scutulatus) lacking the acidic subunit DNA sequence lack Mojave toxin in their venom. Compar Biochem Physiol B Biochem Mol Biol 130:169–179
Wright S (1949) The genetic structure of populations. Ann Eugen 15:323–354
Yamazaki Y, Matsunaga Y, Tokunaga Y, Obayashi S, Saito M, Morita T (2009) Snake venom vascular endothelial growth factors (VEGF-Fs) exclusively vary their structures and functions among species. J Biol Chem 284:9885–9891
Zancolli G, Calvete JJ, Cardwell MD, Greene HW, Hayes WK, Hegarty MJ, Herrmann HW, Holycross AT, Lannutti DI, Mulley JF, Sanz L, Travis ZD, Whorley JR, Wüster CE, Wüster W (2019) When one phenotype is not enough: divergent evolutionary trajectories govern venom variation in a widespread rattlesnake species. Proc R Soc B Biol Sci. https://doi.org/10.1098/rspb.2018.2735
Zelanis A, Andrade-Silva D, Rocha MM, Furtado MF, Serrano SMT, Junqueira-de Azevedo ILM, Ho PL (2012) A transcriptomic view of the proteome variability of newborn and adult Bothrops jararaca snake venoms. PLoS Neglected Trop Dis 6:e1554
Zelanis A, Menezes MC, Kitano ES, Liberato T, Tashima AK, Pinto AF, Sherman NE, Ho PL, Fox JW, Serrano SM (2016) Proteomic identification of gender molecular markers in Bothrops jararaca venom. J Proteom 139:26–37. https://doi.org/10.1016/j.jprot.2016.02.030
Zhang C, Rabiee M, Sayyari E, Mirarab S (2018) Astral-III: polynomial time species tree reconstruction from partially resolved gene trees. BMC Bioinfo 19:15–30. https://doi.org/10.1186/s12859-018-2129-y
Zhang J, Kobert K, Flouri T, Stamatakis A (2014) PEAR: a fast and accurate illumina paired-end reAd mergeR. Bioinformatics 30:614–620
Acknowledgements
We want to thank Jason Jones, Leo Badillo, and Cristobal Moreno for help in the field. We greatly appreciate Tristan Schramer’s assistance with illustrations, and digital photography. Additionally, he and N. Jade Mellor are thanked for the many great discussions and comments on the manuscript. We greatly appreciate Carl Whittington for his comments on the PLA\(_2\) model analysis. Parallel computing resources were provided by the Clemson Palmetto High-Performance Computing Cluster and in part, with support from the Clemson University Genomics and Bioinformatics Facility, which receives support from two Institutional Development Awards (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under grant numbers P20GM109094 and P20GM139767.
Funding
This work was supported by the National Science Foundation Grants DUE 1161228, DEB 1638879, and DEB 1822417 to C.L.P., DEB 1638902 to D.R.R, and a Fulbright García Robles graduate fellowship to R.A.R.G.
Author information
Authors and Affiliations
Contributions
RARG, EPH, and CLP conceived the study. CIG, HFC, ITAC, RRC, MADL, MB, GCG, MS, MLH, and RMR collected samples used in this study. RMR, EPH, MLH, JLS, AJM, and RARG generated data, RARG, RMR, and EPH processed data and performed analyses. RARG drafted the manuscript. JLS, AJM, DRR, EAM, MLH, and CLP provided analytical and conceptual input. RMR, EAM, EPH, AJM, JLS, MLH, and CLP reviewed and edited the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent for Publication
Not applicable
Ethics Approval
All protocols involving live snakes followed the ASIH guidelines and were approved by the University of Central Florida and Clemson University Animal Care and Use Committees (16-17W: UCF and 2017-067: CU). The sample (LIAP244) was collected in and exported from Costa Rica under Investigation Permit SINAC-ACC-OSJ-re-428-2021. Samples (CHFCB-0271, CHFCB-0272, CHFCB-0274, CHFCB-0290, CHFCB-0291, CHFCB-0300, CHFCB-0236, CHFCB-0238, CHFCB-0239, CHFCB-0276, CHFCB-027, CHFCB-0296, CHFCB-0471) were collected in and exported from Mexico under SEMARNAT:SGPA/DGVS/01090/17;SGPA/DGVS/002288/18;SGPA/DGVS/08831/20.
Additional information
Handling editor: Keith Crandall.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rosales-García, R.A., Rautsaw, R.M., Hofmann, E.P. et al. Sequence Divergence in Venom Genes Within and Between Montane Pitviper (Viperidae: Crotalinae: Cerrophidion) Species is Driven by Mutation–Drift Equilibrium. J Mol Evol 91, 514–535 (2023). https://doi.org/10.1007/s00239-023-10115-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-023-10115-2