Abstract
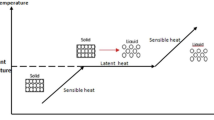
Latent heat storage units are considered as an effective solution for the mismatch problem between energy consumption and energy supply when utilizing solar energy. However, the low thermal conductivity of the current phase change materials (PCMs) is regarded as their main drawback. The present study used dispersing nanoparticles in PCM and installing longitudinal fins on the inner tube simultaneously to enhance the melting rate of PCM in a horizontal shell-and-tube heat exchanger. Different compositions of stearic acid (SA) and titanium dioxide TiO2-NPs were employed as nano-encapsulated PCMs as well. The results demonstrated that installing fins have a significant effect on the melting rate of PCM and can improve the melting rate by 68%. Although adding 0.39 wt% TiO2-NPs to PCM enhanced its thermal conductivity of SA by 7 and 15% in liquid and solid phases, respectively, its effect on improving the melting rate of PCM was less than 4%, which is related to the weakening of the natural convection flows because of the increased viscosity.






















Similar content being viewed by others
Abbreviations
- T :
-
temperature (°C or K)
- f :
-
liquid fraction
- L :
-
latent heat of PCM (J/kg)
- S H :
-
source term in energy equation
- S v :
-
source term in momentum equation
- H :
-
total enthalpy (J/kg)
- h :
-
heat transfer coefficient or enthalpy (W/m2·K or J/kg)
- k :
-
thermal conductivity of PCM (W/m °C)
- t :
-
time (s or min)
- C p :
-
specific heat at constant pressure (J/kg °C)
- l :
-
length of heat exchanger (m)
- r :
-
radius of inner tube (m)
- ∀:
-
total volume of PCM container (m3)
- A :
-
heat transfer surface (m2)
- D h :
-
hydraulic diameter (m)
- V :
-
velocity (m/s)
- H h :
-
hypothetical height of the liquid on the inner tube (m)
- P :
-
pressure (Pa)
- g :
-
gravitational acceleration (m/s2)
- A mushy :
-
mushy zone constant (kg/m3·s)
- \( \overline{h} \) :
-
surface-averaged heat transfer coefficient (W/m2·K)
- \( \overline{Nu} \) :
-
surface-averaged Nusselt number
- M :
-
mass (kg)
- x :
-
characteristic length (m)
- Q :
-
absorbed thermal energy by PCM (j)
- Δt :
-
time step (s)
- \( \overline{\dot{Q}} \) :
-
surface-averaged heat transfer rate (w)
- \( \left\langle \overline{\dot{Q}}\right\rangle \) :
-
time-averaged heat transfer rate (w)
- \( \left\langle \overline{Nu}\right\rangle \) :
-
time -averaged Nusselt number
- Fo :
-
Fourier number \( \alpha t/{D}_h^2 \)
- Ra x :
-
Rayleigh number gβ(THTF − Tm)x3/να
- Ste :
-
Stefan number Cp, l(THTF − Tm)/L
- Nu :
-
Nusselt number hDh/kl
- AR :
-
heat transfer surface Ratio A/Ab
- MR :
-
mass fraction of nanoparticles MNP/MPCM
- ρ :
-
density of PCM (kg/m3)
- μ :
-
dynamic viscosity of PCM (kg/m s)
- α :
-
thermal diffusivity (m2/s)
- ε :
-
overall effectiveness
- η :
-
numerical constant
- β :
-
volumetric expansion coefficient (1/K)
- ϕ :
-
particle volume fraction
- l :
-
liquid phase
- 0:
-
references
- m :
-
melting point
- solidus :
-
solidus of the phase change
- liquidus :
-
liquidus of the phase change
- b :
-
base state
- t :
-
total
- NP :
-
nanoparticles
- PCM :
-
phase change materials
- HTF :
-
heat transfer fluid
- NePCM :
-
nanoparticles enhanced phase change materials
References
Rozanna D, Chuah TG, Salmiah A, Choong TSY, Sa’ari M (2005) Fatty acids as phase change materials (PCMs) for thermal energy storage: a review. Int J Green Energy 1:495–513. https://doi.org/10.1081/GE-200038722
Yuan Y, Zhang N, Tao W, Cao X, He Y (2014) Fatty acids as phase change materials: a review. Renew Sust Energ Rev 29:482–498. https://doi.org/10.1016/j.rser.2013.08.107
Zalba B, Marín JM, Cabeza LF, Mehling H (2003) Review on thermal energy storage with phase change: materials, heat transfer analysis and applications. Appl Therm Eng 23:251–283. https://doi.org/10.1016/S1359-4311(02)00192-8
Al-Abidi AA, Mat S, Sopian K, Sulaiman MY, Mohammad AT (2013) Internal and external fin heat transfer enhancement technique for latent heat thermal energy storage in triplex tube heat exchangers. Appl Therm Eng 53:147–156. https://doi.org/10.1016/j.applthermaleng.2013.01.011
A. Pizzolato, A. Sharma, K. Maute, A. Sciacovelli, V. Verda (2017) Design of e ffective fins for fast PCM melting and solidification in shell-and-tube latent heat thermal energy storage through topology optimization, Appl Energy 1–18. doi:https://doi.org/10.1016/j.apenergy.2017.10.050
Jamekhorshid A, Sadrameli SM, Farid M (2014) A review of microencapsulation methods of phase change materials (PCMs) as a thermal energy storage (TES) medium. Renew Sust Energ Rev 31:531–542. https://doi.org/10.1016/j.rser.2013.12.033
Salunkhe PB, Shembekar PS (2012) A review on effect of phase change material encapsulation on the thermal performance of a system. Renew Sust Energ Rev 16:5603–5616. https://doi.org/10.1016/j.rser.2012.05.037
MacPhee D, Dincer I (2009) Thermal modeling of a packed bed thermal energy storage system during charging. Appl Therm Eng 29:695–705. https://doi.org/10.1016/j.applthermaleng.2008.03.041
Wu M, Xu C, He YL (2014) Dynamic thermal performance analysis of a molten-salt packed-bed thermal energy storage system using PCM capsules. Appl Energy 121:184–195. https://doi.org/10.1016/j.apenergy.2014.01.085
Abdollahzadeh M, Esmaeilpour M (2015) Enhancement of phase change material (PCM) based latent heat storage system with nano fluid and wavy surface. Int J Heat Mass Transf 80:376–385. https://doi.org/10.1016/j.ijheatmasstransfer.2014.09.007
Canseco V, Anguy Y, Josep J, Palomo E (2014) Structural and mechanical characterization of graphite foam / phase change material composites. Carbon N Y 74:266–281. https://doi.org/10.1016/j.carbon.2014.03.031
Zhou D, Zhao CY (2011) Experimental investigations on heat transfer in phase change materials (PCMs) embedded in porous materials. Appl Therm Eng 31:970–977. https://doi.org/10.1016/j.applthermaleng.2010.11.022
Velraj R, Seeniraj RV, Hafner B, Faber C, Schwarzer K (1999) Heat transfer enhancement in a latent heat storage system. Sol Energy 65:171–180. https://doi.org/10.1016/S0038-092X(98)00128-5
Zhang P, Meng ZN, Zhu H, Wang YL, Peng SP (2015) Melting heat transfer characteristics of a composite phase change material fabricated by paraffin and metal foam q. Appl Energy. https://doi.org/10.1016/j.apenergy.2015.10.075
Frusteri F, Leonardi V, Vasta S, Restuccia G (2005) TC measurement of a PCM based storage system containing carbon fibers. Appl Therm Eng 25:1623–1633. https://doi.org/10.1016/j.applthermaleng.2004.10.007
Fukai J, Hamada Y, Morozumi Y, Miyatake O (2002) Effect of carbon-fiber brushes on conductive heat transfer in phase change materials. Int J Heat Mass Transf 45:4781–4792. https://doi.org/10.1016/S0017-9310(02)00179-5
Ranjbar S, Masoumi H, Haghighi Khoshkho R, Mojtaba K (2019) Experimental investigation of stability and TC of phase change materials containing pristine and functionalized multi - walled carbon nanotubes. J Therm Anal Calorim. https://doi.org/10.1007/s10973-019-09005-x
Leong KY, Rosdzimin M, Rahman A, Gurunathan BA (2019) Nano-enhanced phase change materials : a review of thermo-physical properties, applications and challenges. J Energy Storage 21:18–31. https://doi.org/10.1016/j.est.2018.11.008
Khodadadi JM, Hosseinizadeh SF (2007) Nanoparticle-enhanced phase change materials (NEPCM) with great potential for improved thermal energy storage. Int Commun Heat Mass Transf 34:534–543. https://doi.org/10.1016/j.icheatmasstransfer.2007.02.005
Dhaidan NS, Khodadadi JM, Al-hattab TA, Al-mashat SM (2013) Experimental and numerical investigation of melting of NePCM inside an annular container under a constant heat flux including the effect of eccentricity. Int J Heat Mass Transf 67:455–468. https://doi.org/10.1016/j.ijheatmasstransfer.2013.08.002
Sharma RK, Ganesan P, Tyagi VV, Metselaar HSC, Sandaran SC (2016) Thermal properties and heat storage analysis of palmitic acid-TiO2composite as nano-enhanced organic phase change material (NEOPCM). Appl Therm Eng 99:1254–1260. https://doi.org/10.1016/j.applthermaleng.2016.01.130
Sami S, Etesami N (2017) Improving thermal characteristics and stability of phase change material containing TiO2 nanoparticles after thermal cycles for energy storage. Appl Therm Eng. https://doi.org/10.1016/j.applthermaleng.2017.06.023
Harikrishnan S, Magesh S, Kalaiselvam S (2013) Preparation and thermal energy storage behaviour of stearic acid-TiO2 nanofluids as a phase change material for solar heating systems. Thermochim Acta 565:137–145. https://doi.org/10.1016/j.tca.2013.05.001
T. Teng, C. Yu (2012) Characteristics of phase-change materials containing oxide nano-additives for thermal storage 1–10
Babita SK, Sharma SM (2016) Gupta, preparation and evaluation of stable nanofluids for heat transfer application: a review. Exp Thermal Fluid Sci 79:202–212. https://doi.org/10.1016/j.expthermflusci.2016.06.029
E.B. Haghighi, N. Nikkam, M. Saleemi, M. Behi, S.A. Mirmohammadi, H. Poth, R. Khodabandeh, M.S. Toprak, M. Muhammed, B. Palm (2013) Shelf stability of nanofluids and its effect on thermal conductivity and viscosity, Meas. Sci. Technol. 24. doi:https://doi.org/10.1088/0957-0233/24/10/105301
Hosseini MJ, Ranjbar AA, Rahimi M, Bahrampoury R (2015) Experimental and numerical evaluation of longitudinally finned latent heat thermal storage systems. Energy Build. https://doi.org/10.1016/j.enbuild.2015.04.045
Rabienataj Darzi AA, Jourabian M, Farhadi M (2016) Melting and solidification of PCM enhanced by radial conductive fins and nanoparticles in cylindrical annulus. Energy Convers Manag 118:253–263. https://doi.org/10.1016/j.enconman.2016.04.016
Parsazadeh M, Duan X (2017) Numerical and statistical study on melting of nanoparticle enhanced phase change material in a shell-and-tube thermal energy storage system. Appl Therm Eng 111:950–960. https://doi.org/10.1016/j.applthermaleng.2016.09.133
Ranjbar S, Masoumi H, Haghighi Khoshkho R, Mirfendereski SM (2019) Experimental investigation of stability and thermal conductivity of phase change materials containing pristine and functionalized multi-walled carbon nanotubes. J Ther Anal Calorim. https://doi.org/10.1007/s10973-019-09005
Masoumi H, Haghighi Khoshkho R, Mirfendereski SM (2019) Modification of physical and thermal characteristics of stearic acid as a phase change materials using TiO2-nanoparticles. Thermochim Acta 675:9–17. https://doi.org/10.1016/j.tca.2019.02.015
Seddegh S, Wang X, Henderson AD (2016) A comparative study of thermal behaviour of a horizontal and vertical shell-and-tube energy storage using phase change materials. Appl Therm Eng 93:348–358. https://doi.org/10.1016/j.applthermaleng.2015.09.107
Al-Abidi AA, Mat S, Sopian K, Sulaiman MY, Mohammad AT (2013) Numerical study of PCM solidification in a triplex tube heat exchanger with internal and external fins. Int J Heat Mass Transf 61:684–695. https://doi.org/10.1016/j.ijheatmasstransfer.2013.02.030
Kamkari B, Shokouhmand H (2014) Experimental investigation of phase change material melting in rectangular enclosures with horizontal partial fins. Int J Heat Mass Transf 78:839–851. https://doi.org/10.1016/j.ijheatmasstransfer.2014.07.056
Jiang X, Luo R, Peng F, Fang Y, Akiyama T, Wang S (2015) Synthesis, characterization and thermal properties of paraffin microcapsules modified with nano-Al2O3. Appl Energy 137:731–737. https://doi.org/10.1016/j.apenergy.2014.09.028
Jesumathy S, Udayakumar M, Suresh S (2012) Experimental study of enhanced heat transfer by addition of CuO nanoparticle. Heat Mass Transf Und Stoffuebertragung 48:965–978. https://doi.org/10.1007/s00231-011-0945-y
Şahan N, Fois M, Paksoy H (2015) Improving thermal conductivity phase change materials - a study of paraffin nanomagnetite composites. Sol Energy Mater Sol Cells 137:61–67. https://doi.org/10.1016/j.solmat.2015.01.027
Park S, Lee Y, Kim YS, Lee HM, Kim JH, Cheong IW, Koh WG (2014) Magnetic nanoparticle-embedded PCM nanocapsules based on paraffin core and polyurea shell. Colloids Surfaces A Physicochem Eng Asp 450:46–51. https://doi.org/10.1016/j.colsurfa.2014.03.005
Harikrishnan S, Deenadhayalan M, Kalaiselvam S (2014) Experimental investigation of solidification and melting characteristics of composite PCMs for building heating application. Energy Convers Manag 86:864–872. https://doi.org/10.1016/j.enconman.2014.06.042
Motahar S, Nikkam N, Alemrajabi AA, Khodabandeh R, Toprak MS, Muhammed M (2014) Experimental investigation on thermal and rheological properties of n-octadecane with dispersed TiO2nanoparticles. Int Commun Heat Mass Transf 59:68–74. https://doi.org/10.1016/j.icheatmasstransfer.2014.10.016
Babapoor A, Karimi G (2015) Thermal properties measurement and heat storage analysis of paraffin-nanoparticles composites phase change material: comparison and optimization. Appl Therm Eng. https://doi.org/10.1016/j.applthermaleng.2015.07.083
Wang J, Xie H, Li Y, Xin Z (2010) PW based phase change nanocomposites containing γ-Al 2O3. J Therm Anal Calorim 102:709–713. https://doi.org/10.1007/s10973-010-0850-5
Ho CJ, Gao JY (2009) Preparation and thermophysical properties of nanoparticle-in-paraffin emulsion as phase change material. Int Commun Heat Mass Transf 36:467–470. https://doi.org/10.1016/j.icheatmasstransfer.2009.01.015
Fan L, Khodadadi JM (2012) An experimental investigation of enhanced thermal conductivity and expedited unidirectional freezing of cyclohexane-based nanoparticle suspensions utilized as nano-enhanced phase change materials (NePCM). Int J Therm Sci 62:120–126. https://doi.org/10.1016/j.ijthermalsci.2011.11.005
Krishna J, Kishore PS, Solomon AB (2017) Heat pipe with nano enhanced-PCM for electronic cooling application. Exp Thermal Fluid Sci 81:84–92. https://doi.org/10.1016/j.expthermflusci.2016.10.014
Nourani M, Hamdami N, Keramat J, Moheb A, Shahedi M (2016) Thermal behavior of paraffin-nano-Al2O3stabilized by sodium stearoyl lactylate as a stable phase change material with high thermal conductivity. Renew Energy 88:474–482. https://doi.org/10.1016/j.renene.2015.11.043
Colla L, Fedele L, Mancin S, Danza L, Manca O (2017) Nano-PCMs for enhanced energy storage and passive cooling applications. Appl Therm Eng 110:584–589. https://doi.org/10.1016/j.applthermaleng.2016.03.161
H. C. Brinkman (1952) “The Viscosity of Concentrated Suspensions and Solutions,” 571:1–2. https://doi.org/10.1063/1.1700493
Fan L, Zhu Z, Zeng Y, Ding Q, Liu M (2016) Unconstrained melting heat transfer in a spherical container revisited in the presence of nano-enhanced phase change materials (NePCM). Int J Heat Mass Transf 95:1057–1069. https://doi.org/10.1016/j.ijheatmasstransfer.2016.01.013
E. Assis, L. Katsman, G. Ziskind, R. Letan (2007) Numerical and experimental study of melting in a spherical shell, 50: 1790–1804. https://doi.org/10.1016/j.ijheatmasstransfer.2006.10.007
Jones D, Smith F (1970) Optimum arrangement of rectangular fins on horizontal surfaces for free convection heat transfer. J Heat Transf 92:6–10
Wong S, Huang G (2013) International journal of heat and mass transfer parametric study on the dynamic behavior of natural convection from horizontal rectangular fin arrays. Int J Heat Mass Transf 60:334–342. https://doi.org/10.1016/j.ijheatmasstransfer.2013.01.019
Kline S, Mcclintock F (1953) Describing uncertainties in single-sample experiments. Mech Eng 75:3–8
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Simultaneously are studied effects of fins and nanoparticles on melting of PCM.
• Different composites of TiO2 nanoparticles and stearic acid are studied as NePCM.
• Thermal behavior of a shell-and-tube energy storage system using PCM is studied.
• Effect of increasing the HTF temperature on melting PCM is examined.
• Installing fins is more effective than adding nanoparticles on PCM melting rates.
Rights and permissions
About this article
Cite this article
Masoumi, H., Haghighi Khoshkhoo, R. Investigation of melting of nanoparticle-enhanced phase change materials (NePCMs) in a shell-and-tube heat exchanger with longitudinal fins. Heat Mass Transfer 57, 681–701 (2021). https://doi.org/10.1007/s00231-020-02983-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00231-020-02983-x