Abstract
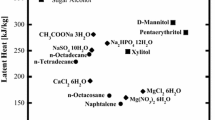
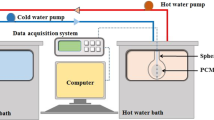
In this study, we investigated the fundamental melting and solidification characteristics of mannitol, erythritol, and their mixture (70 % by mass mannitol: 30 % by mass erythritol) as potential phase-change materials (PCMs) for latent heat thermal energy storage systems, specifically those pertaining to industrial waste heat, having temperatures in the range of 100–250 °C. The melting point of erythritol and mannitol, the melting peak temperature of their mixture, and latent heat were measured using differential scanning calorimetry. The thermal performance of the mannitol mixture was determined during melting and solidification processes, using a heat storage vessel with a pipe heat exchanger. Our results indicated phase-change (fusion) temperatures of 160 °C for mannitol and 113 and 150 °C for the mannitol mixture. Nondimensional correlation equations of the average heat transfer during the solidification process, as well as the temperature and velocity efficiencies of flowing silicon oil in the pipe and the phase-change material (PCM), were derived using several nondimensional parameters.




















Similar content being viewed by others
References
Bo H, Gustafsson EM, Setterwall F (1999) Tetradecane and hexadecane binary mixtures as phase change materials (PCMs) for cool storage in district cooling systems. Energy 24:1015–1028
Choi ES, Cho YI, Lorsch HG (1992) Thermal analysis of the mixture of laboratory and commercial grades hexadecane and tetradecane. Int Commun Heat Mass Transf 19:1–15
Kumano H, Saito A, Okawa S, Takeda K, Okuda A (2005) Study of direct contact melting with hydrocarbon mixtures as the PCM. Int J Heat Mass Transf 48:3212–3220
Webb BW, Viskanta R (1986) An experimental and analytical study of solidification of a binary mixture. Proc Int Heat Transf Conf 4:1739–1744
Nagano K, Ogawa K, Mochida T, Hayashi K, Ogoshi H (2004) Thermal characteristics of magnesium nitrate hexahydrate and magnesium chloride hexahydrate mixture as a phase change material for effective utilization of urban waste heat. Appl Therm Eng 24:221–232
Cedeno FO, Prieto MM, Espina A, Garcia JR (2001) Measurements of temperature and melting heat of some pure fatty acids and their binary and ternary mixtures by differential scanning calorimetry. Thermochim Acta 369:39–50
Sari A, Kaygusuz K (2002) Thermal performance of a eutectic mixture of lauric and stearic acids as PCM encapsulated in the annulus of two concentric pipes. Sol Energy 72:493–504
Hidaka H, Yamazaki M, Yabe M, Kakiuchi H, Ona EP, Kojima Y, Matsuda H (2004) New PCMs prepared from erythritol-polyalcohols mixtures for latent heat storage between 80 and 100 °C. J Chem Eng Jpn 37:1155–1162
Kubota M, Ona EP, Watanabe F, Matsuda H, Hidaka H, Kakiuchi H (2007) Studies on phase change characteristics of binary mixtures of erythritol and MgCl2·6H2O. J Chem Eng Jpn 40:80–84
Yanxia Du, Yanping Yuan, Daiyong Jia, Baoyi Cheng, Jinfeng Mao (2007) Experimental investigation on melting characteristics of ethanolamine-water binary mixture used a PCM. Int Commun Heat Mass Trasnf 34:1056–1063
Nagano K, Ogawa T, Mochida T, Hayashi K, Ogoshi H (2004) Performance of heat charge/discharge of magnesium nitrate hexahydrate and magnesium chloride hexahydrate mixture to a single vertical tube for a latent heat storage system. Appl Therm Eng 24:209–220
Baran G, Sari A (2003) Phase change and heat transfer characteristics of a eutectic mixture of palmitic and stearic acids as PCM in a latent heat storage system. Energy Convers Manag 44:3227–3246
Horibe A, Yu JS, Haruki N, Kaneda A, Machida A, Kato M (2011) Melting characteristic of mixture of two kinds of latent heat storage material. Netsu Bussei 25:136–142
Acknowledgments
This research was performed in cooperation with the New Energy and Industrial Technology Development Organization (NEDO) of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, J., Horibe, A., Haruki, N. et al. Melting and solidification characteristics of a mixture of two types of latent heat storage material in a vessel. Heat Mass Transfer 52, 2563–2574 (2016). https://doi.org/10.1007/s00231-016-1767-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00231-016-1767-8