Abstract
Purpose
To systematically assess the evidence of efficacy and safety of the use of ketamine and esketamine for patients with treatment-resistant depression (TRD) with suicidal ideation (SI).
Methods
We independently searched for clinical trials from inception to January 2023 using electronic databases, e.g., PubMed and EMBASE. A systematic review and meta-analysis were performed to assess SI scores of depression rating scales, which were regarded as the outcomes.
Results
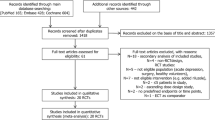
A total of five independent double-blind, placebo controlled randomized clinical trials (RCTs) are eligible for inclusion. Four of the studies used ketamine as an intervention and one used esketamine as an intervention. Three hundred ninety-one patients with TRD were included (the intervention group with ketamine or esketamine is 246, and the control group is 145). No statistically significant interaction between the subscales of suicide ideation (SMD = − 0.66, 95% CI (− 1.61, 0.29); Z = 1.36, P = 0.17) and antidepressant effects (SMD = − 0.99, 95% CI (− 2.33, 0.34); Z = 1.46, P = 0.15) based on the results of ketamine and esketamine, compared with placebo groups.
Conclusion
This meta-analysis suggested that esketamine and ketamine have failed to reduce suicidal ideation in patients with TRD. Further studies are desirable to confirm the effects of ketamine and esketamine in TRD patients.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- Ads:
-
Antidepressants
- CI:
-
Confidence
- MADRS-SI:
-
Montgomery-Åsberg Depression Rating Scale suicidality item
- CRSS-SI:
-
Columbia Suicide Severity Rating Scale suicidality item
- CSSRS:
-
Columbia Suicide Severity Rating Scale
- DB:
-
Double-blind
- DSM-5:
-
Diagnostic and Statistical Manual of Mental Disorders (fifth edition) criteria
- HAM-D:
-
Hamilton Depression Rating Scale
- IN:
-
Intranasal
- IV:
-
Intravenous injection
- MADRS:
-
Montgomery-Åsberg Depression Rating Scale
- MDD:
-
Major depressive disorder
- RCTs:
-
Randomized clinical trials
- SCID:
-
Structured Clinical Interview for DSM-IV Diagnoses
- SI:
-
Suicidality item
- SIcomposite :
-
A composite explicit suicidality index (Beck Scale, MADRS, and Quick Inventory of Depressive Symptomatology-Self Report)
- TRD:
-
Treatment-resistant depression
References
Wang YT, Wang XL, Wang ZZ, Lei L, Hu D, Zhang Y (2022) Antidepressant effects of the traditional Chinese herbal formula Xiao-Yao-San and its bioactive ingredients. Phytomedicine 109:154558. https://doi.org/10.1016/j.phymed.2022.154558
Lei L, Wang Y-T, Hu D, Gai C, Zhang Y (2023) Astroglial connexin 43-mediated gap junctions and hemichannels: potential antidepressant mechanisms and the link to neuroinflammation. Cell Mol Neurobiol 43(8):4023–4040. https://doi.org/10.1007/s10571-023-01426-5
Wang XL, Wang YT, Guo ZY, Zhang NN, Wang YY, Hu D, Wang ZZ, Zhang Y (2022) Efficacy of paeoniflorin on models of depression: a systematic review and meta-analysis of rodent studies. J Ethnopharmacol 290:115067. https://doi.org/10.1016/j.jep.2022.115067
Wang XL, Feng ST, Wang YT, Chen NH, Wang ZZ, Zhang Y (2021) Paeoniflorin: a neuroprotective monoterpenoid glycoside with promising anti-depressive properties. Phytomedicine 90:153669. https://doi.org/10.1016/j.phymed.2021.153669
Wang YT, Wang XL, Feng ST, Chen NH, Wang ZZ, Zhang Y (2021) Novel rapid-acting glutamatergic modulators: targeting the synaptic plasticity in depression. Pharmacol Res 171:105761. https://doi.org/10.1016/j.phrs.2021.105761
Ivanova JI, Birnbaum HG, Kidolezi Y, Subramanian G, Khan SA, Stensland MD (2010) Direct and indirect costs of employees with treatment-resistant and non-treatment-resistant major depressive disorder. Curr Med Res Opin 26(10):2475–2484. https://doi.org/10.1185/03007995.2010.517716
Mrazek DA, Hornberger JC, Altar CA, Degtiar I (2014) A review of the clinical, economic, and societal burden of treatment-resistant depression: 1996–2013. Psychiatric services (Washington, DC) 65(8):977–987. https://doi.org/10.1176/appi.ps.201300059
Henter ID, Park LT, Zarate CA Jr (2021) Novel glutamatergic modulators for the treatment of mood disorders: current status. CNS Drugs 35(5):527–543. https://doi.org/10.1007/s40263-021-00816-x
Leon AC, Solomon DA, Li C, Fiedorowicz JG, Coryell WH, Endicott J, Keller MB (2011) Antidepressants and risks of suicide and suicide attempts: a 27-year observational study. J Clin Psychiatry 72(5):580–586. https://doi.org/10.4088/JCP.10m06552
Nobile B, Dubois J, Aouizerate B, Aubin V, Loftus J, Bellivier F, Belzeaux R, Dubertret C, Gard S, Haffen E, Schwan R, Llorca PM, Passerieux C, Roux P, Polosan M, Etain B, Leboyer M, Courtet P, Olié E (2021) Characterization of depressed bipolar patients with current suicidal ideation. Aust N Z J Psychiatry 55(3):289–304. https://doi.org/10.1177/0004867420963744
Lopez-Castroman J, Jaussent I, Gorwood P, Courtet P (2016) Suicidal depressed patients respond less well to antidepressants in the short term. Depress Anxiety 33(6):483–494. https://doi.org/10.1002/da.22473
Wang SM, Kim NY, Na HR, Lim HK, Woo YS, Pae CU, Bahk WM (2021) Rapid onset of intranasal esketamine in patients with treatment resistant depression and major depression with suicide ideation: a meta-analysis. Clin Psychopharmacol Neurosci 19(2):341–354. https://doi.org/10.9758/cpn.2021.19.2.341
Bergfeld IO, Mantione M, Figee M, Schuurman PR, Lok A, Denys D (2018) Treatment-resistant depression and suicidality. J Affect Dis 235:362–367. https://doi.org/10.1016/j.jad.2018.04.016
Gronemann FH, Jørgensen MB, Nordentoft M, Andersen PK, Osler M (2021) Treatment-resistant depression and risk of all-cause mortality and suicidality in Danish patients with major depression. J Psychiatr Res 135:197–202. https://doi.org/10.1016/j.jpsychires.2021.01.014
McIntyre RS, Carvalho IP, Lui LMW, Majeed A, Masand PS, Gill H, Rodrigues NB, Lipsitz O, Coles AC, Lee Y, Tamura JK, Iacobucci M, Phan L, Nasri F, Singhal N, Wong ER, Subramaniapillai M, Mansur R, Ho R, Lam RW, Rosenblat JD (2020) The effect of intravenous, intranasal, and oral ketamine in mood disorders: a meta-analysis. J Affect Dis 276:576–584. https://doi.org/10.1016/j.jad.2020.06.050
Kaur U, Pathak BK, Singh A, Chakrabarti SS (2021) Esketamine: a glimmer of hope in treatment-resistant depression. Eur Arch Psychiatry Clin Neurosci 271(3):417–429. https://doi.org/10.1007/s00406-019-01084-z
Daly EJ, Singh JB, Fedgchin M, Cooper K, Lim P, Shelton RC, Thase ME, Winokur A, Van Nueten L, Manji H, Drevets WC (2018) Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiat 75(2):139–148. https://doi.org/10.1001/jamapsychiatry.2017.3739
Xiong J, Lipsitz O, Chen-Li D, Rosenblat JD, Rodrigues NB, Carvalho I, Lui LMW, Gill H, Narsi F, Mansur RB, Lee Y, McIntyre RS (2021) The acute antisuicidal effects of single-dose intravenous ketamine and intranasal esketamine in individuals with major depression and bipolar disorders: a systematic review and meta-analysis. J Psychiatr Res 134:57–68. https://doi.org/10.1016/j.jpsychires.2020.12.038
Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, McKenzie JE (2021) PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ 372:n160. https://doi.org/10.1136/bmj.n160
Feng ST, Wang XL, Wang YT, Yuan YH, Li ZP, Chen NH, Wang ZZ, Zhang Y (2021) Efficacy of traditional Chinese medicine combined with selective serotonin reuptake inhibitors on the treatment for Parkinson’s disease with depression: a systematic review and meta-analysis. Am J Chin Med 49(3):627–643. https://doi.org/10.1142/s0192415x21500282
Zhang Y, Wang ZZ, Sun HM, Li P, Li YF, Chen NH (2014) Systematic review of traditional Chinese medicine for depression in Parkinson’s disease. Am J Chin Med 42(5):1035–1051. https://doi.org/10.1142/s0192415x14500657
Wang XL, Feng ST, Chen B, Hu D, Wang ZZ, Zhang Y (2022) Efficacy and safety of istradefylline for Parkinson’s disease: a systematic review and meta-analysis. Neurosci Lett 774:136515. https://doi.org/10.1016/j.neulet.2022.136515
Marcantoni WS, Akoumba BS, Wassef M, Mayrand J, Lai H, Richard-Devantoy S, Beauchamp S (2020) A systematic review and meta-analysis of the efficacy of intravenous ketamine infusion for treatment resistant depression: January 2009 - January 2019. J Affect Dis 277:831–841. https://doi.org/10.1016/j.jad.2020.09.007
Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, Thomas J (2019) Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev 10:Ed000142. https://doi.org/10.1002/14651858.Ed000142
Chen MH, Wu HJ, Li CT, Lin WC, Tsai SJ, Hong CJ, Tu PC, Bai YM, Mao WC, Su TP (2022) Low-dose ketamine infusion in treatment-resistant double depression: revisiting the adjunctive ketamine study of Taiwanese patients with treatment-resistant depression. Hum Psychopharmacol 37(2). https://doi.org/10.1002/hup.2820
Takahashi N, Yamada A, Shiraishi A, Shimizu H, Goto R, Tominaga Y (2021) Efficacy and safety of fixed doses of intranasal esketamine as an add-on therapy to oral antidepressants in Japanese patients with treatment-resistant depression: a phase 2b randomized clinical study. BMC Psychiatry 21(1):526. https://doi.org/10.1186/s12888-021-03538-y
Fava M, Freeman MP, Flynn M, Judge H, Hoeppner BB, Cusin C, Ionescu DF, Mathew SJ, Chang LC, Iosifescu DV, Murrough J, Debattista C, Schatzberg AF, Trivedi MH, Jha MK, Sanacora G, Wilkinson ST, Papakostas GI (2020) Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry 25(7):1592–1603. https://doi.org/10.1038/s41380-018-0256-5
Chen MH, Lin WC, Tu PC, Li CT, Bai YM, Tsai SJ, Su TP (2019) Antidepressant and antisuicidal effects of ketamine on the functional connectivity of prefrontal cortex-related circuits in treatment-resistant depression: a double-blind, placebo-controlled, randomized, longitudinal resting fMRI study. J Affect Disord 259:15–20. https://doi.org/10.1016/j.jad.2019.08.022
Su TP, Chen MH, Li CT, Lin WC, Hong CJ, Gueorguieva R, Tu PC, Bai YM, Cheng CM, Krystal JH (2017) Dose-related effects of adjunctive ketamine in Taiwanese patients with treatment-resistant depression. Neuropsychopharmacology 42(13): 2482–2492. https://doi.org/10.1038/npp.2017.94
Li CT, Chen MH, Lin WC, Hong CJ, Yang BH, Liu RS, Tu PC, Su TP (2016) The effects of low-dose ketamine on the prefrontal cortex and amygdala in treatment-resistant depression: a randomized controlled study. Hum Brain Mapp 37(3):1080–1090. https://doi.org/10.1002/hbm.23085
Price R, Iosifescu DV, Murrough JW, Chang LC, Al Jurdi RK, Charney DS, Foulkes AL, Mathew SJ (2013) Effects of intravenous ketamine on explicit and implicit suicidal cognition: a randomized controlled trial in treatment-resistant depression. Biol Psychiatry 73(9):142S-143S
Chen MH, Lin WC, Wu HJ, Cheng CM, Li CT, Hong CJ, Tu PC, Bai YM, Tsai SJ, Su TP (2019) Antisuicidal effect, BDNF Val66Met polymorphism, and low-dose ketamine infusion: reanalysis of adjunctive ketamine study of Taiwanese patients with treatment-resistant depression (AKSTP-TRD). J Affect Disord 251:162–169. https://doi.org/10.1016/j.jad.2019.03.075
Feeney A, Hock RS, Freeman MP, Flynn M, Hoeppner B, Iosifescu DV, Trivedi MH, Sanacora G, Mathew SJ, Debattista C, Ionescu DF, Fava M, Papakostas GI (2021) The effect of single administration of intravenous ketamine augmentation on suicidal ideation in treatment-resistant unipolar depression: results from a randomized double-blind study. Eur Neuropsychopharmacol 49:122–132. https://doi.org/10.1016/j.euroneuro.2021.04.024
Ohnishi T, Wakamatsu A, Kobayashi H (2022) Different symptomatic improvement pattern revealed by factor analysis between placebo response and response to esketamine in treatment resistant depression. Psychiatry Clin Neurosci 76(8):377–383. https://doi.org/10.1111/pcn.13379
Price RB, Iosifescu DV, Murrough JW, Chang LC, Al Jurdi RK, Iqbal SZ, Soleimani L, Charney DS, Foulkes AL, Mathew SJ (2014) Effects of ketamine on explicit and implicit suicidal cognition: a randomized controlled trial in treatment-resistant depression. Depress Anxiety 31(4):335–343. https://doi.org/10.1002/da.22253
Ionescu DF, Bentley KH, Eikermann M, Taylor N, Johnson-Akeju O, Swee MB, Pavone KJ, Petrie SR, Dording C, Mischoulon D, Alpert JE, Brown EN, Baer L, Nock MK, Fava M, Cusin C (2019) Repeat-dose ketamine augmentation for treatment-resistant depression with chronic suicidal ideation: a randomized, double blind, placebo controlled trial. J Affect Disord 243:516–524. https://doi.org/10.1016/j.jad.2018.09.037
Berman RM, Cappiello A, Anand A, Oren DA, Heninger GR, Charney DS, Krystal JH (2000) Antidepressant effects of ketamine in depressed patients. Biol Psychiatry 47(4):351–354. https://doi.org/10.1016/s0006-3223(99)00230-9
Borbély É, Simon M, Fuchs E, Wiborg O, Czéh B, Helyes Z (2022) Novel drug developmental strategies for treatment-resistant depression. Br J Pharmacol 179(6):1146–1186. https://doi.org/10.1111/bph.15753
Zheng W, Gu LM, Yang XH, Zhou YL, Wang CY, Lan XF, Zhang B, Ning YP (2022) Association of anhedonia and suicidal ideation in patients with treatment-refractory depression after intravenous ketamine infusions. Int J Psychiatry Clin Pract. https://doi.org/10.1080/13651501.2022.2138444
Fancy F, Rodrigues NB, Di Vincenzo JD, Chau EH, Sethi R, Husain MI, Gill H, Tabassum A, McKenzie A, Phan L, McIntyre RS, Rosenblat JD (2022) Real-world effectiveness of repeated ketamine infusions for treatment-resistant bipolar depression. Bipolar Disord. https://doi.org/10.1111/bdi.13284
Singh JB, Fedgchin M, Daly EJ, De Boer P, Cooper K, Lim P, Pinter C, Murrough JW, Sanacora G, Shelton RC, Kurian B, Winokur A, Fava M, Manji H, Drevets WC, Van Nueten L (2016) A double-blind, randomized, placebo-controlled, dose-frequency study of intravenous ketamine in patients with treatment-resistant depression. Am J Psychiatry 173(8):816–826. https://doi.org/10.1176/appi.ajp.2016.16010037
Murrough JW, Iosifescu DV, Chang LC, Al Jurdi RK, Green CM, Iqbal S, Pillemer S, Perez AM, Foulkes A, Shah A et al (2012) Antidepressant efficacy of ketamine in treatment-resistant major depression: a two-site, randomized, parallel-arm, midazolam-controlled, clinical trial. Neuropsychopharmacology 38:S151‐S152. https://doi.org/10.1038/npp.2012.219
Ballard ED, Luckenbaugh DA, Richards EM, Walls TL, Brutsché NE, Ameli R, Niciu MJ, Vande Voort JL, Zarate CA (2015) Assessing measures of suicidal ideation in clinical trials with a rapid-acting antidepressant. J Psychiatr Res 68:68–73. https://doi.org/10.1016/j.jpsychires.2015.06.003
Witt K, Potts J, Hubers A, Grunebaum MF, Murrough JW, Loo C, Cipriani A, Hawton K (2020) Ketamine for suicidal ideation in adults with psychiatric disorders: a systematic review and meta-analysis of treatment trials. Aust N Z J Psychiatry 54(1):29–45. https://doi.org/10.1177/0004867419883341
Vieira F, Correia-Melo FS, Santos-Lima C, Souza-Marques B, Leal GC, Jesus-Nunes AP, Mello RP, Caliman-Fontes AT, Bandeira ID, Marback RF, Telles M, Argolo FC, Lins-Silva DH, Echegaray MVF, Beanes G, Araújo-de-Freitas L, Silva SS, Cardoso TA, Kapczinski F, Turecki G, Lacerda ALT, Quarantini LC (2021) Ketamine and esketamine augmentation for suicidal ideation: a randomized, double-blinded clinical trial. Gen Hosp Psychiatry 68:97–99. https://doi.org/10.1016/j.genhosppsych.2020.12.011
Grunebaum MF, Ellis SP, Keilp JG, Moitra VK, Cooper TB, Marver JE, Burke AK, Milak MS, Sublette ME, Oquendo MA et al (2017) Ketamine versus midazolam in bipolar depression with suicidal thoughts: a pilot midazolam-controlled randomized clinical trial. Bipolar Disord 19(3):176–183. https://doi.org/10.1111/bdi.12487
Ionescu DF, Swee MB, Pavone KJ, Taylor N, Akeju O, Baer L, Nyer M, Cassano P, Mischoulon D, Alpert JE, Brown EN, Nock MK, Fava M, Cusin C (2016) Rapid and sustained reductions in current suicidal ideation following repeated doses of intravenous ketamine: secondary analysis of an open-label study. J Clin Psychiatry 77(6):e719–e725. https://doi.org/10.4088/JCP.15m10056
Murrough JW, Soleimani L, DeWilde KE, Collins KA, Lapidus KA, Iacoviello BM, Lener M, Kautz M, Kim J, Stern JB, Price RB, Perez AM, Brallier JW, Rodriguez GJ, Goodman WK, Iosifescu DV, Charney DS (2015) Ketamine for rapid reduction of suicidal ideation: a randomized controlled trial. Psychol Med 45(16):3571–3580. https://doi.org/10.1017/S0033291715001506
Kollmar R, Markovic K, Thürauf N, Schmitt H, Kornhuber J (2008) Ketamine followed by memantine for the treatment of major depression. Aust N Z J Psychiatry 42(2):170. https://doi.org/10.1080/00048670701787628
López-Díaz Á, Fernández-González JL, Luján-Jiménez JE, Galiano-Rus S, Gutiérrez-Rojas L (2017) Use of repeated intravenous ketamine therapy in treatment-resistant bipolar depression with suicidal behaviour: a case report from Spain. Ther Adv Psychopharmacol 7(4):137–140. https://doi.org/10.1177/2045125316675578
Vande Voort JL, Morgan RJ, Kung S, Rasmussen KG, Rico J, Palmer BA, Schak KM, Tye SJ, Ritter MJ, Frye MA, Bobo WV (2016) Continuation phase intravenous ketamine in adults with treatment-resistant depression. J Affect Disord 206:300–304. https://doi.org/10.1016/j.jad.2016.09.008
Funding
This work was supported by grants from the National Natural Science Foundation of China (Nos. 82274127 and 82104644).
Author information
Authors and Affiliations
Contributions
Y-TW: investigation, methodology, formal analysis, writing—original draft. X-LW: investigation, methodology, formal analysis. LL: investigation. Z-YG: investigation. F-FK: investigation. DH: investigation. CG: investigation. YZ: conceptualization, methodology, supervision, writing—review and editing. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, YT., Wang, XL., Lei, L. et al. A systematic review and meta-analysis of the efficacy of ketamine and esketamine on suicidal ideation in treatment-resistant depression. Eur J Clin Pharmacol 80, 287–296 (2024). https://doi.org/10.1007/s00228-023-03605-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-023-03605-1