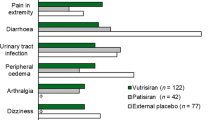
Abstract
Purpose
The transthyretin kinetic stabilizer tafamidis, used as a first-line therapy of amyloidosis patients, binds selectively to the transthyretin protein structure and thus prevents its dissociation. The limited information regarding tafamidis application in Glu89Gln amyloidosis patients imposed our research team to determine and evaluate its individual mean plasma levels and their biological variation.
Methods
The present cohort study investigated Bulgarian amyloidosis patients, grouped by gender, age, and therapy duration. A total of sixty patients aged 40–75 years and therapy duration up to 9 years were included. A precise and accurate high-performance liquid chromatography method with ultraviolet detection was used for plasma concentration measurement.
Results
Mean plasma concentrations were 5.13 ± 2.64 µmol/L and showed low intra-individual (18.50%) and high inter-individual variability (51.43%). No significant difference was observed between tafamidis plasma levels and therapy duration with p = 0.5941 (p < 0.05 considered significant), but a significant positive correlation was found between plasma concentration, gender, and age with obtained results about p-value 0.0001 and 0.0235, respectively.
Conclusion
The summary results of the study showed differences that may be based on some specific clinical features of the Glu89Gln mutation.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Müller ML, Butler J, Heidecker B (2020) Emerging therapies in transthyretin amyloidosis – a new wave of hope after years of stagnancy? Eur J Heart Fail 22(1):39–53
Burton A, Castaño A, Bruno M et al (2021) Drug discovery and development in rare diseases: taking a closer look at the tafamidis story. Drug Des Devel Ther 15:1225–1243
Mundayat R, Stewart M, Alvir J et al (2018) Positive effectiveness of tafamidis in delaying disease progression in transthyretin familial amyloid polyneuropathy up to 2 years: an analysis from the transthyretin amyloidosis outcomes survey (THAOS). Neurol Ther 7(1):87–101
Ando Y, Coelho T, Berk JL et al (2013) Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J Rare Dis 8(1):1–18
Park J, Egolum U, Parker S et al (2020) Tafamidis: a first-in-class transthyretin stabilizer for transthyretin amyloid cardiomyopathy. Ann Pharmacother 54(5):470–477
Rapezzi C, Elliott P, Damy T et al (2021) Efficacy of Tafamidis in patients with hereditary and wild-type transthyretin amyloid cardiomyopathy: further analyses from ATTR-ACT. JACC Hear Fail 9(2):115–123
Coelho T, Merlini G, Bulawa CE et al (2016) Mechanism of action and clinical application of tafamidis in hereditary transthyretin amyloidosis. Neurol Ther 5(1):1–25
Lockwood PA, Le VH, O’Gorman MT et al (2020) The bioequivalence of tafamidis 61-mg free acid capsules and Tafamidis meglumine 4 × 20-mg capsules in healthy volunteers. Clin Pharmacol Drug Dev 9(7):849–854
Monteiro C, Mesgazardeh JS, Anselmo J et al (2019) Predictive model of response to tafamidis in hereditary ATTR polyneuropathy. JCI Insight 4(12):1–17
Gentile L, Tournev I, Amass L et al (2021) Phenotypic differences of Glu89Gln genotype in ATTR amyloidosis from endemic loci: update from THAOS. Cardiol Ther 10(2):481–490
Rapezzi C, Quarta CC, Obici L et al (2013) Disease profile and differential diagnosis of hereditary transthyretin-related amyloidosis with exclusively cardiac phenotype: an Italian perspective. Eur Heart J 34(7):520–528
Cortese A, Vita G, Luigetti M et al (2016) Monitoring effectiveness and safety of tafamidis in transthyretin amyloidosis in Italy: a longitudinal multicenter study in a non-endemic area. J Neurol 263(5):916–924
Gagliardi C, Gospodinova M, Longhi S et al (2015) Glu89Gln transthyretin-related amyloidosis in Italy and Bulgaria: does geographic area influence phenotype beyond the shared mutation? Orphanet J Rare Dis 10(S1):P23. Proceedings of the First European Congress on Hereditary ATTR amyloidosis; November 2–3, 2015; Paris, France
Milanov I, Tournev I (2019) National consensus for diagnosis, treatment, follow-up and prevention of hereditary transthyretin amyloidosis. Bulgarian Neurology 20:1–30 (Bulgarian)
Gospodinova M, Sarafov S, Guergueltcheva V et al (2015) Cardiomyopathy and peripheral polyneuropathy severity in patients with Glu89Gln mutation at the time of diagnosis. Orphanet J Rare Dis 10(S1):P59. Proceedings of the First European Congress on Hereditary ATTR amyloidosis; November 2–3, 2015; Paris, France
Gospodinova M, Sarafov S, Guergueltcheva V et al (2015) Cardiac involvement and clinical follow up of patients with hereditary transthyretin related amyloidosis associated with Glu89Gln mutation. Orphanet J Rare Dis 10 (S1):P54. Proceedings of the First European Congress on Hereditary ATTR amyloidosis; November 2–3, 2015; Paris, France
Gospodinova M, Sarafov S, Chamova T et al (2020) Cardiac involvement, morbidity and mortality in hereditary transthyretin amyloidosis because of p.Glu89Gln mutation. J Cardiovasc Med (Hagerstown) 21:688–695
Nakov R, Sarafov S, Nakov V et al (2019) Gastrointestinal manifestations in hereditary transthyretin amyloidosis associated with Glu89Gln mutation. J Gastrointest Liver Dis 28(4):421–426
Smerikarova M, Bozhanov S, Maslarska V et al (2022) Determination of tafamidis plasma concentrations in amyloidosis patients with Glu89Gln mutation by HPLC-UV detection. J Chrom Sci 60(9):840–847
Sarafov S (2020) Clinical-genetic and epidemiological studies in transthyretin familial amyloid polyneuropathy and in hereditary and familial amyotrophic lateral sclerosis in Bulgaria. Dissertation, Medical University-Sofia
Acknowledgements
The authors are thankful to UMBAL Alexandrovska Hospital, Department of Neurology, for providing human plasma samples for conducting the experiments.
Funding
This work was kindly supported by Medical University-Sofia, Medical Science Council–Grant No. 121/24.06.2020.
Author information
Authors and Affiliations
Contributions
Vania Maslarska and Ivailo Tournev contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Miglena Smerikarova and Stanislav Bozhanov. The first draft of the manuscript was written by Miglena Smerikarova, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Research Ethics Committee at the Medical University of Sofia (RECMUS) with a certification no.1480/14.05.2020.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Smerikarova, M., Bozhanov, S., Tournev, I. et al. Variation of tafamidis plasma levels during the treatment of TTR amyloidosis patients with Glu89Gln mutation. Eur J Clin Pharmacol 79, 1657–1664 (2023). https://doi.org/10.1007/s00228-023-03576-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-023-03576-3