Abstract
Purpose
Investigate inhaled nitric oxide’s influence on mortality rates, mechanical ventilation and cardiopulmonary bypass duration, and length of stay in the intensive care unit and hospital when administered during cardiopulmonary bypass.
Methods
Following the PRISMA guidelines, we searched four electronic databases (PubMed, EMBASE, Cochrane Library, and Web of Science) up to 4th March 2023. The protocol was registered in the PROSPERO database with ID: CRD42023423007. Using Review Manager software, we reported outcomes as risk ratios (RRs) or mean difference (MD) and confidence intervals (CIs).
Results
The meta-analysis included a total of 17 studies with 2897 patients. Overall, there were no significant differences in using nitric oxide over control concerning mortality (RR = 1.03, 95% CI 0.73 to 1.45; P = 0.88) or cardiopulmonary bypass duration (MD = −0.14, 95% CI − 0.96 to 0.69; P = 0.74). The intensive care unit days were significantly lower in the nitric oxide group than control (MD = −0.80, 95% CI − 1.31 to −0.29; P = 0.002). Difference results were obtained in terms of the length of stay in the hospital according to sensitivity analysis (without sensitivity [MD = −0.41, 95% CI − 0.79 to −0.02; P = 0.04] vs. with sensitivity [MD = −0.31, 95% CI − 0.69 to 0.07; P = 0.11]. Subgroup analysis shows that, in children, nitric oxide was favored over control in significantly reducing the duration of mechanical ventilation (MD = −4.58, 95% CI − 5.63 to −3.53; P < 0.001).
Conclusion
Using inhaled nitric oxide during cardiopulmonary bypass reduces the length of stay in the intensive care unit, and for children, it reduces the duration of mechanical ventilation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiopulmonary bypass (CPB) is a procedure that keeps the body’s blood and oxygen supply flowing during surgery by temporarily replacing the heart and lungs with a machine. CPB may be required for patients with coronary artery bypass grafting, aneurysm surgery, heart transplant, heart valve surgery, or lung transplant. Platelets are activated and consumed as blood travels across the synthetic surfaces of the CPB circuit [1]. In addition to necessitating blood product replacement, platelet activation is a factor in the severe inflammatory response observed with CPB procedures [2]. Postoperative problems such as respiratory failure, renal dysfunction, bleeding issues, cognitive dysfunction, altered liver function, cardiac injury, and multiple organ failure may arise due to this inflammatory cascade [3]. Vascular nitric oxide (NO) bioavailability is decreased by CPB, in part due to NO scavenging (through a deoxygenation reaction in the presence of intravascular hemolysis) [4, 5]. NO regulates the endothelial function and microvascular inflammation and is an endogenous anti-inflammatory mediator [6, 7]. When NO binds to platelet intracellular receptors, it has many functions that inhibit platelet activation and aggregation [8]. In primary and secondary pulmonary hypertension from congenital heart disease and patients having cardiac surgery, inhaled NO has been demonstrated to promote pulmonary vasodilation [9,10,11]. Currently, NO is being investigated for its potential protective effects against myocardial damage in patients receiving CPB in several randomized trials [12,13,14]. It is crucial from a therapeutic standpoint to establish the ideal NO dosage range for adult cardiac surgery since many NO-required patients are critically unwell and have failed conventional therapy [15]. In patients susceptible to its effects, prompt therapy with a suitable amount of inhaled NO may eliminate the need for CBP and, in certain circumstances, be life-saving [15]. Currently, several clinical trials on NO have been reported, and results were inconsistent. Furthermore, CPB is utilized for the surgical correction of congenital heart diseases in children [14, 16,17,18]. Low cardiac output syndrome might occur in this operation and cause multiorgan failure [19]. In adults, it finds application in other conditions, such as heart valve replacement or repair surgeries [3, 20,21,22,23,24,25]. However, it is worth mentioning that the reported incidence of low cardiac output syndrome varies from 2 to 27% in the adult population. In the pediatric population, reported incidences are between 17 and 67% [26]. Hence, it would be valuable to underscore the distinctions between its utilization in adults and children.
Therefore, we conducted a new systematic review and meta-analysis to compare mortality, length of mechanical ventilation, length of stay in the hospital and intensive care unit, and CBP duration between adults and children receiving inhaled NO during CPB and standard care.
Methods
We followed PRISMA statement guidelines when reporting this systemic review and meta-analysis [27]. All steps were done in accordance with the Cochrane Handbook of Systematic Review and Meta-analysis of Interventions (version 5.1.0) [28]. This review methods were established prior to the conduct of the review and no significant deviations from the protocol was observed, and it is registered in the PROSPERO database with ID: CRD42023423007. The thorough PRISMA checklist is shown in supplementary file 1.
Eligibility criteria
We included studies in our review if they satisfied the following criteria:
-
1.
Population: patients undergoing cardiopulmonary bypass.
-
2.
Intervention: nitric oxide administration during cardiopulmonary bypass.
-
3.
Comparator: standard care without nitric oxide administration.
-
4.
Outcome:
-
(i)
Primary outcomes: mortality and length of mechanical ventilation
-
(ii)
Secondary outcomes: the length of stay in the hospital and intensive care unit (ICU) and CBP duration
-
(i)
-
5.
Study design: we included clinical trials and randomized clinical trials.
And we excluded non-published data, reviews, case reports, editorial letters, conference abstracts, study protocols, animal and phantom studies, and patients who had received NO after CBP. No restriction on the language of the study was determined.
Search strategy
We searched the following electronic medical databases: Embase, PubMed, Web of Science, and Cochrane Library from inception till 4th March 2023. The search query is available in (Supplementary File 2).
Screening and data extraction
Citations with abstracts were retrieved from the databases and inserted into the Rayyan database. We resolved the duplications prior to screening. Due to many exported studies, every two authors formed a team, and studies were split equally to assess their relevance. In case of a conflict, a third opinion was made to resolve it, and it was based on reading the original article. Next, the full articles were retrieved, and the entire screen of text started to determine the final eligibility of the study for our meta-analysis. Then, we extracted data in duplicate from studies accordingly in a uniform sheet for primary and secondary outcomes.
Assessment of risk of bias
Two independent authors (Y.R., F.R.) performed the quality assessment of the screened studies using the Risk of Bias 2 (RoB 2) tool [29], and discrepancies were resolved by consensus. The following domains were evaluated individually and graded as “low risk,” “high risk,” or “no information”: randomization processes, deviations from intended intervention, missing outcome data, measuring the outcomes, and selection of reported results.
Data analysis and synthesis
Statistical analyses were conducted using Review Manager v.5.3. We calculated the pooled risk ratios (RR) and 95% confidence intervals (CI) for the binary outcome of mortality using the random-effects model. It accommodates a larger standard error in the pooled estimate, making it suitable for inconsistent or controversial estimates. We estimated the mean difference (MD) and 95% CI for continuous outcomes of the length of mechanical ventilation, ICU and hospital stay, and CBP duration. If continuous variables were expressed as a median and interquartile range, the mean and standard deviation were computed based on the median, interquartile range, and sample size, as described by Hozo et al. [30]. Regarding the heterogeneity, the chi-square test evaluated statistical heterogeneity among studies. Then, the chi-square statistic was used to calculate I-squared. Chi-square with a P value less than 0.1 was considered significant heterogeneity. Also, the I-square value of more than or equal to 50% indicated high heterogeneity [31]. We used pairwise deletion for missing variables. We performed a subgroup analysis to investigate whether the effect of inhaled NO varied between children and adults. We performed sensitivity analyses to determine the robustness of the effect size by removing one study per time to check the strength of the evidence and ensure the overall results were not altered.
Results
Search results and study selection
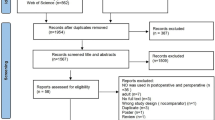
Through the electronic search, 6473 citations were identified. After excluding 1794 duplicates, 4083 studies were chosen for further evaluation. Through reading the titles and abstracts, 4649 ineligible studies were excluded, and 30 studies were identified as potentially eligible for inclusion and were evaluated by reading the full text. We did not retrieve the full text of a Chinese study due to the language barrier; hence, 29 studies went for full-text reading. Twelve studies were excluded due to the absence of a control group, did not report the required outcomes, compared different routes of NO administration, a withdrawn clinical study, or clinical trial protocol. Finally, 17 studies were eligible for systematic review (n = 2897 patients) [3, 13, 14, 16,17,18, 20,21,22,23,24,25, 32,33,34,35,36]. Figure 1 shows the process of literature selection and reasons for study exclusion.
PRISMA chart of the reported studies showing the search selection strategy and exclusion criteria [27]
Characteristics of individual studies
The characteristics of the included studies and outcomes are summarized in Tables 1 and 2. These studies vary in the condition of the patients and NO doses. We also split the studies according to the age of the patients. Adults were involved in 10 studies [3, 20,21,22,23,24,25, 33,34,35], while seven studies included children, infants, and neonates [13, 14, 16,17,18, 32, 36].
Risk of bias and quality assessment
Most of our included studies (13 out of 17) showed a low risk of overall bias, and only four articles showed a high risk of bias. The quality evaluation is shown in Fig. 2.
Risk of bias assessment is represented in traffic light plot and summary plot according to the Cochrane risk-of-bias tool, created using robvis [41]
Outcomes
Mortality
The overall effect of the analysis of seven studies [17, 20, 23, 24, 33, 35, 36] showed an insignificant risk ratio between the NO group and the experimental (RR = 1.03, 95% CI 0.73 to 1.45; P = 0.88), with no heterogeneity (I2 = 0%, P = 0.44). Subgroup analysis also showed no significant differences in adult (RR = 0.87, 95% CI 0.50 to 1.49; P = 0.60) or children’s groups (RR = 1.61, 95% CI 0.40 to 6.46; P = 0.50) (Fig. 3a).
Duration of mechanical ventilation (hours)
In eight studies [13, 14, 16, 20, 22, 24, 35, 36], significant differences were observed with regard to the duration of mechanical ventilation between the two groups (MD = −3.04, 95% CI − 5.42 to −0.67; P = 0.01), with high heterogeneity (I2 = 90%, P < 0.001) (Fig. 3b). The heterogeneity was not resolved. Subgroup analysis also showed no significant differences in adults (MD = −0.72, 95% CI − 2.28 to 0.85; P = 0.37), with heterogeneity (I2 = 62%, P = 0.05). Even though the heterogeneity reduced to I2 = 48% and P = 0.15 after excluding Prendergast et al. [22], the results did not change, even for the overall results. However, the children group favored NO over the control (MD = −4.58, 95% CI − 5.63 to −3.53; P < 0.001), with no heterogeneity (I2 = 0%, P = 0.64) (Fig. 3b).
ICU time (days)
The overall effect of ten studies [13, 14, 16, 20, 22,23,24, 32, 34, 35] showed that the number of days in the ICU was significantly lower in the NO group than control (MD = −0.80, 95% CI − 1.31 to −0.29; P = 0.002), with high heterogeneity (I2 = 95%, P < 0.001) (Fig. 4a). In adults, the results were in the trending line of significance to favor NO as well (MD = −0.79, 95% CI − 1.59 to 0.01; P = 0.05) with high heterogeneity (I2 = 93%, P < 0.001) (Fig. 4a). However, after excluding Kamenshchikov et al. [35], the results became insignificant (MD = −0.37, 95% CI − 0.78 to 0.03; P = 0.07), and the heterogeneity reduced to I2 = 66% and P = 0.02) (Fig. 4b). In children, the results were insignificant (MD = −0.83, 95% CI − 1.74 to 0.07; P = 0.07) with high heterogeneity (I2 = 97%, P < 0.001) (Fig. 4a); however, after excluding Schlapbach et al. [32] (the highest number of patients), the results significantly favored NO (MD = −1.29, 95% CI − 1.53 to −1.04; P < 0.001) with no heterogeneity (I2 = 0%, P = 0.38) (Fig. 4c).
Hospital stay (days)
Analyzing 11 studies [13, 14, 16, 18, 20, 22,23,24, 32, 35, 36] showed that hospital stay was significantly less in the NO group (MD = −0.41, 95% CI − 0.79 to −0.02; P = 0.04), with high heterogeneity (I2 = 91%, P < 0.001) (Fig. 5a). However, excluding Kamenshchikov et al. [35] from the adult group will show overall insignificant results (MD = −0.31, 95% CI − 0.69 to 0.07; P = 0.11) (Fig. 5b); similarly, if we remove any of these studies from the children group (Christopher et al. [13], Kolcz et al. [16], or Schlapbach et al. [32]), it will result in an insignificant reduction in the hospital stay, and the heterogeneity will remain high. Subgroup analysis also showed similar insignificant results in adults (MD = −0.52, 95% CI − 1.14 to 0.09; P = 0.10) and children (MD = −0.29, 95% CI − 0.90 to 0.33; P = 0.36), with high heterogeneity of I2 = 89% and P < 0.001 and I2 = 93% and P < 0.001, respectively (Fig. 5a).
CBP duration (minutes)
The overall effect of the analysis of nine studies [13, 14, 16, 18, 21,22,23, 25, 36] showed that NO did not significantly influence the changes in CBP duration (MD = −0.14, 95% CI − 0.96 to 0.69; P = 0.74), with high heterogeneity (I2 = 94%, P < 0.001) (Fig. 6a). Subgroup analysis shows that neither adults nor children significantly reported differences in CBP duration in the two groups (MD = −0.68, 95% CI − 1.57 to 0.21; P = 0.13; and MD = 0.30, 95% CI − 0.83 to 1.44; P = 0.60, respectively) (Fig. 6a). Heterogeneity was high in the two groups. Removing Pichugin from the adults’ analysis showed a trending line toward significance to favor NO (MD = −1.00, 95% CI − 2.01 to 0.01; P = 0.05), with high heterogeneity (I2 = 81%, P = 0.006) (Fig. 6b). Sensitivity in the children group was resolved (I2 = 0%, P = 0.59) by excluding Christopher et al. [13] (largest sample size), and the results remained insignificant (MD = −0.12, 95% CI − 0.42 to 0.17; P = 0.41) (Fig. 6c).
Publication bias for included studies
We found asymmetry in the funnel plots assessing studies of mechanical ventilation, ICU stay, hospital stay, and CBP duration, as shown in Fig. 7, indicating the possibility of publication bias due to the insufficient literature and the heterogeneity.
Discussion
This systematic review and meta-analysis revealed a significant difference in the outcomes of patients who received inhaled NO during CBP compared to those who did not. The results indicate that administering inhaled NO is associated with overall lower ICU and hospital stay and lower duration of mechanical ventilation, especially in children. NO did not affect mortality rates or CBP duration. Sensitivity analysis showed insignificant results for the influence of NO on reducing ICU time for children and overall hospital stay. Hence, we suggest further studies on a larger sample size for more robust evidence of the beneficial impact of NO.
In children, NO was introduced to CBP based on two trials involving 16 [14] and 198 children [13], respectively. The former showed its beneficial effect on decreasing ICU time and shorter duration of mechanical ventilation, while the latter reported a lower incidence of low cardiac output syndrome in children < 2 years. However, the results reported by previously published large trials with 1364 children > 2 years showed no significant difference in death within 28 days or ventilator-free days between those who received nitric oxide delivered into CPB and those who received care [32]. According to our study, we have strong evidence that the children group favored NO over control concerning the mortality rate and duration of mechanical ventilation. During the sensitivity analysis, excluding large trials [32] altered the results of the length of stay in the ICU, and sensitivity analysis of hospital stay provided insignificant results.
In adults, in a study by Tylor et al. [33], the authors highlighted the lack of long-term benefits despite initial improvements in oxygenation upon using NO, which was observed in this analysis, and NO did not influence the mortality rate, duration of mechanical ventilation or length of ICU stay, hospital stay (through sensitivity analysis), and CBP duration in this group.
Compared to a previous meta-analysis with five studies and 579 patients [37], the authors reported no association between NO therapy and the hospital or ICU stay length. We obtained similar results during sensitivity analysis. It is worth mentioning that the author’s main aim was to evaluate the relative risk of acute kidney injury after NO therapy, which was not necessarily administered during CPB. Our inclusion criteria determined only two studies eligible for renal replacement therapy analysis; however, we did not see significant differences (RR = 0.51, 95% CI 0.13 to 2.02; P = 0.34).
Furthermore, a recent meta-analysis by Elnaiem et al. [38] investigated six studies. It included a total of 1666 children who were undergoing cardiac surgery and showed comparative results to our study in terms of reduced time on mechanical ventilation when using NO during CBP. In addition, they showed a reduction in postoperative levels of IL-6 and tumor necrosis factor-alpha (P < 0.001 and P = 0.05, respectively). The agreement of the results between the studies suggests the beneficial use of NO, particularly for children.
A potential rationale was previously presented by Lincoln et al. [39]. The authors have linked the disparities in acute respiratory distress syndrome between children and adults to the continuing progression of postnatal lung development until reaching adult stature. This process involves an intersection of factors that govern the regulation of postnatal lung maturation, as well as mechanisms related to inflammation, apoptosis, alveolar fluid clearance, and tissue repair. Consequently, a distinct underlying framework of genetic and protein expression exists in children when compared to adults. Building upon this premise, Hunt et al. [40] proposed a hypothesis. Recognizing that the disease state is not uniform across all age groups, the authors proposed that the response to therapies, specifically inhaled NO, might exhibit variations.
Strength and limitations
One of the main strengths of this study is the large sample size, which increases the reliability of the findings. Additionally, the use of meta-analytic techniques allowed us to pool data from multiple studies and synthesize the results statistically rigorously. This helped increase the study’s power and reduce the risk of type I errors. However, some limitations to this study should be noted. First, some analyses had mild to high heterogeneity, which could limit the generalizability of the findings even when using the random model effect. This heterogeneity could be due to differences in patient populations, clinical conditions, surgical techniques, duration, and postoperative management strategies. Some included studies showed a high risk of bias. Finally, the present study did not report some competing endpoints, such as renal replacement therapy, the impact of different doses—most doses were 20 PPM—or the duration of NO.
Conclusion
This systematic review and meta-analysis showed that inhaled nitric oxide during cardiopulmonary bypass lowered the overall length of stay in the intensive care unit and might lower the duration of mechanical ventilation in children. Nitric oxide did not influence the mortality rates, hospital stay, or cardiopulmonary bypass duration, suggesting additional high-quality studies to validate these results.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information file.
References
Despotis GJ, Avidan MS, Hogue CW (2001) Mechanisms and attenuation of hemostatic activation during extracorporeal circulation. Ann Thorac Surg 72:S1821-1831
Rinder CS, Bonan JL, Rinder HM, Mathew J, Hines R, Smith BR (1992) Cardiopulmonary bypass induces leukocyte-platelet adhesion. Blood 79:1201–1205
Gianetti J, Del Sarto P, Bevilacqua S, Vassalle C, De Filippis R, Kacila M et al (2004) Supplemental nitric oxide and its effect on myocardial injury and function in patients undergoing cardiac surgery with extracorporeal circulation. J Thorac Cardiovasc Surg 127:44–50
Vermeulen Windsant IC, de Wit NCJ, Sertorio JTC, van Bijnen AA, Ganushchak YM, Heijmans JH et al (2014) Hemolysis during cardiac surgery is associated with increased intravascular nitric oxide consumption and perioperative kidney and intestinal tissue damage. Front Physiol 5:340
Vermeulen Windsant IC, Hanssen SJ, Buurman WA, Jacobs MJ (2011) Cardiovascular surgery and organ damage: time to reconsider the role of hemolysis. J Thorac Cardiovasc Surg 142:1–11
Hataishi R, Rodrigues AC, Neilan TG, Morgan JG, Buys E, Shiva S et al (2006) Inhaled nitric oxide decreases infarction size and improves left ventricular function in a murine model of myocardial ischemia-reperfusion injury. Am J Physiol-Heart Circ Physiol 291:H379–H384
Chello M, Mastroroberto P, Perticone F, Celi V, Colonna A (1998) Nitric oxide modulation of neutrophil-endothelium interaction: difference between arterial and venous coronary bypass grafts. J Am Coll Cardiol 31:823–826
Naseem KM, Roberts W (2011) Nitric oxide at a glance. Platelets 22:148–152
Snow DJ, Gray SJ, Ghosh S, Foubert L, Oduro A, Higenbottam TW et al (1994) Inhaled nitric oxide in patients with normal and increased pulmonary vascular resistance after cardiac surgery. Br J Anaesth 72:185–189
Roberts JD, Fineman JR, Morin FC, Shaul PW, Rimar S, Schreiber MD et al (1997) Inhaled nitric oxide and persistent pulmonary hypertension of the newborn. N Engl J Med 336:605–610
Kulik TJ (1996) Inhaled nitric oxide in the management of congenital heart disease. Curr Opin Cardiol 11:75–80
Rajek A, Pernerstorfer T, Kastner J, Mares P, Grabenwöger M, Sessler DI et al (2000) Inhaled nitric oxide reduces pulmonary vascular resistance more than prostaglandin E(1) during heart transplantation. Anesth Analg 90:523–530
James C, Millar J, Horton S, Brizard C, Molesworth C, Butt W (2016) Nitric oxide administration during paediatric cardiopulmonary bypass: a randomised controlled trial. Intensive Care Med 42:1744–1752
Checchia PA, Bronicki RA, Muenzer JT, Dixon D, Raithel S, Gandhi SK et al (2013) Nitric oxide delivery during cardiopulmonary bypass reduces postoperative morbidity in children–a randomized trial. J Thorac Cardiovasc Surg 146:530–536
Solina AR, Ginsberg SH, Papp D, Grubb WR, Scholz PM, Pantin EJ et al (2001) Dose response to nitric oxide in adult cardiac surgery patients. J Clin Anesth 13:281–286
Kolcz J, Karnas E, Madeja Z, Zuba-Surma EK (2022) The cardioprotective and anti-inflammatory effect of inhaled nitric oxide during Fontan surgery in patients with single ventricle congenital heart defects: a prospective randomized study. J Intensive Care 10:48
Miller OI, Tang SF, Keech A, Pigott NB, Beller E, Celermajer DS (2000) Inhaled nitric oxide and prevention of pulmonary hypertension after congenital heart surgery: a randomised double-blind study. Lancet 356:1464–1469
Elzein C, Urbas C, Hughes B, Li Y, Lefaiver C, Ilbawi M et al (2020) Efficacy of nitric oxide administration in attenuating ischemia/reperfusion injury during neonatal cardiopulmonary bypass. World J Pediatr Congenit Heart Surg 11:417–423
Hoffman TM, Wernovsky G, Atz AM, Kulik TJ, Nelson DP, Chang AC et al (2003) Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation 107:996–1002
Nakane T, Esaki J, Ueda R, Honda M, Okabayashi H (2021) Inhaled nitric oxide improves pulmonary hypertension and organ functions after adult heart valve surgeries. Gen Thorac Cardiovasc Surg 69:1519–1526
Mellgren K, Mellgren G, Lundin S, Wennmalm A, Wadenvik H (1998) Effect of nitric oxide gas on platelets during open heart operations. Ann Thorac Surg 65:1335–1341
Prendergast B, Scott DH, Mankad PS (1998) Beneficial effects of inhaled nitric oxide in hypoxaemic patients after coronary artery bypass surgery. Eur J Cardio-Thorac Surg Off J Eur Assoc Cardio-Thorac Surg 14:488–493
Fattouch K, Sbraga F, Sampognaro R, Bianco G, Gucciardo M, Lavalle C et al (2006) Treatment of pulmonary hypertension in patients undergoing cardiac surgery with cardiopulmonary bypass: a randomized, prospective, double-blind study. J Cardiovasc Med Hagerstown Md 7:119–123
Lei C, Berra L, Rezoagli E, Yu B, Dong H, Yu S et al (2018) Nitric oxide decreases acute kidney injury and stage 3 chronic kidney disease after cardiac surgery. Am J Respir Crit Care Med 198:1279–1287
Pichugin VV, Seyfetdinov IR, Ryazanov MV, Domnin SE, Gamzaev AB, Chiginev VA et al (2020) New technology for the use of inhaled nitric oxide to protect the heart and lungs during operations with cardiopulmonary bypass. Sovrem Tehnol V Med 12:28
Schoonen A, van Klei WA, van Wolfswinkel L, van Loon K (2022) Definitions of low cardiac output syndrome after cardiac surgery and their effect on the incidence of intraoperative LCOS: a literature review and cohort study. Front Cardiovasc Med 9:926957
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, The PRISMA et al (2020) statement: an updated guideline for reporting systematic reviews. BMJ 2021:372
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ et al (2019) Cochrane handbook for systematic reviews of interventions. Cochrane Handb Syst Rev Interv 1–694
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5:13
Identifying and quantifying Heterogeneity (2009) In: Introduction to meta-analysis. John Wiley & Sons, Ltd, pp 107–125
Schlapbach LJ, Gibbons KS, Horton SB, Johnson K, Long DA, Buckley DHF et al (2022) Effect of nitric oxide via cardiopulmonary bypass on ventilator-free days in young children undergoing congenital heart disease surgery: the NITRIC Randomized Clinical Trial. JAMA 328:38–47
Taylor RW, Zimmerman JL, Dellinger RP, Straube RC, Criner GJ, Davis K et al (2004) Low-dose inhaled nitric oxide in patients with acute lung injury: a randomized controlled trial. JAMA 291:1603–1609
Chung A, Wildhirt SM, Wang S, Koshal A, Radomski MW (2005) Combined administration of nitric oxide gas and iloprost during cardiopulmonary bypass reduces platelet dysfunction: a pilot clinical study. J Thorac Cardiovasc Surg 129:782–790
Kamenshchikov NO, Anfinogenova YJ, Kozlov BN, Svirko YS, Pekarskiy SE, Evtushenko VV et al (2022) Nitric oxide delivery during cardiopulmonary bypass reduces acute kidney injury: a randomized trial. J Thorac Cardiovasc Surg 163:1393-1403.e9
Niebler RA, Chiang-Ching H, Daley K, Janecke R, Jobe SM, Mitchell ME et al (2021) Nitric oxide added to the sweep gas of the oxygenator during cardiopulmonary bypass in infants: a pilot randomized controlled trial. Artif Organs 45:22–28
Hu J, Spina S, Zadek F, Kamenshchikov NO, Bittner EA, Pedemonte J et al (2019) Effect of nitric oxide on postoperative acute kidney injury in patients who underwent cardiopulmonary bypass: a systematic review and meta-analysis with trial sequential analysis. Ann Intensive Care 9:129
Elnaiem W, Mohamed Elnour A, Koko AEA, Madany M, Hemmeda L (2012) Efficacy and safety of inhaled nitric oxide administered during cardiopulmonary bypass for pediatric cardiac surgery: a systematic review and meta-analysis. Ann Med Surg 2023(85):2865–2874
Smith LS, Zimmerman JJ, Martin TR (2013) Mechanisms of acute respiratory distress syndrome in children and adults: a review and suggestions for future research. Pediatr Crit Care Med 14:631
Hunt JL, Bronicki RA, Anas N (2016) Role of inhaled nitric oxide in the management of severe acute respiratory distress syndrome. Front Pediatr 4
McGuinness LA, Higgins JPT (2021) Risk-of-bias VISualization (robvis): an R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods 12:55–61
Acknowledgements
Mohamed Abouzid is a participant of STER Internationalization of Doctoral Schools Program from NAWA Polish National Agency for Academic Exchange No. PPI/STE/2020/1/00014/DEC/02.
Author information
Authors and Affiliations
Contributions
AR and MA: conception and design. MA, YR, JM, FMR, AAAA, MH, MT, ME, and ND: conducted the search and extracted data. MA and AR: data analysis. MA and MT: wrote the manuscript. MA: editing and supervising the work. All authors read and approved the final content.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abouzid, M., Roshdy, Y., Daniel, J.M. et al. The beneficial use of nitric oxide during cardiopulmonary bypass on postoperative outcomes in children and adult patients: a systematic review and meta-analysis of 2897 patients. Eur J Clin Pharmacol 79, 1425–1442 (2023). https://doi.org/10.1007/s00228-023-03554-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-023-03554-9