Abstract
Aims
Voriconazole (VCZ) displays highly variable pharmacokinetics affecting treatment efficacy and safety. We aimed to identify the factors affecting VCZ steady-state trough concentration (Cssmin) to provide evidence for optimizing VCZ treatment regimens.
Methods
A total of 510 Cssmin of 172 patients with hematopoietic stem cell transplantation and hematologic malignancies and their clinical characteristics and genotypes of FMO, POR, and PXR were included in this study.
Results
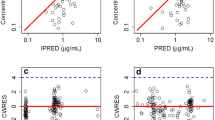
In univariate analysis, the standard loading dose of VCZ significantly increased the Cssmin of VCZ (P < 0.001). The Cssmin of VCZ was significantly correlated with patients’ total bilirubin (TB) (P < 0.001) and procalcitonin (PCT) (P < 0.001). FMO3 rs2266780 (P = 0.025), POR rs10954732 (P = 0.015), PXR rs2461817 (P = 0.010), PXR rs7643645 (P = 0.003), PXR rs3732359 (P = 0.014), PXR rs3814057 (P = 0.005), and PXR rs6785049 (P = 0.013) have a significant effect on Cssmin of VCZ. Loading dose, TB, PCT level, and PXRrs3814057 polymorphism were independent influencing factors of VCZ Cssmin in the analysis of multivariate linear regression. And loading dose, PCT, and PXR rs3814057 had significant effects on the probability of the therapeutic window of VCZ.
Conclusion
The high variability of VCZ Cssmin may be partially explained by loading dose, liver function, inflammation, and PXR polymorphisms. This study suggests the VCZ standard loading dose regimen significantly increased Cssmin and probability of the therapeutic window providing treatment benefits. Patients in the high PCT group may be more likely to exceed 5.5 μg/mL, thus suffering from VCZ toxicity.

Similar content being viewed by others
References
Sheikhbahaei S, Mohammadi A, Sherkat R, Naeini AE, Yaran M, Najafi S (2019) Invasive fungal infection in febrile patients with hematologic malignancies undergoing chemotherapy in Iran. Endocr Metab Immune Disord Drug Targets 19(3):302–307. https://doi.org/10.2174/1871530319666190211163245
Alcazer V, Conrad A, Valour F, Bachy E, Salles G, Huynh A, de Latour RP, Labussiere-Wallet H, Ader F (2019) Early-onset severe infections in allogeneic hematopoietic stem cell transplantation recipients with graft failure. Am J Hematol 94(4):E109–E111. https://doi.org/10.1002/ajh.25406
Sun Y, Meng F, Han M, Zhang X, Yu L, Huang H, Wu D, Ren H, Wang C, Shen Z, Ji Y, Huang X (2015) Epidemiology, management, and outcome of invasive fungal disease in patients undergoing hematopoietic stem cell transplantation in China: a multicenter prospective observational study. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation 21(6):1117–1126. https://doi.org/10.1016/j.bbmt.2015.03.018
Dolton MJ, McLachlan AJ (2014) Voriconazole pharmacokinetics and exposure-response relationships: assessing the links between exposure, efficacy and toxicity. Int J Antimicrob Ag 44(3):183–193. https://doi.org/10.1016/j.ijantimicag.2014.05.019
Perreault S, McManus D, Anderson A, Lin T, Ruggero M, Topal JE (2019) Evaluating a voriconazole dose modification guideline to optimize dosing in patients with hematologic malignancies. Journal of oncology pharmacy practice : official publication of the International Society of Oncology Pharmacy Practitioners 25(6):1305–1311. https://doi.org/10.1177/1078155218786028
Hoenigl M, Duettmann W, Raggam RB, Seeber K, Troppan K, Fruhwald S, Prueller F, Wagner J, Valentin T, Zollner-Schwetz I, Wolfler A, Krause R (2013) Potential factors for inadequate voriconazole plasma concentrations in intensive care unit patients and patients with hematological malignancies. Antimicrob Agents Chemother 57(7):3262–3267. https://doi.org/10.1128/AAC.00251-13
Trifilio S, Ortiz R, Pennick G, Verma A, Pi J, Stosor V, Zembower T, Mehta J (2005) Voriconazole therapeutic drug monitoring in allogeneic hematopoietic stem cell transplant recipients. Bone Marrow Transplant 35(5):509–513. https://doi.org/10.1038/sj.bmt.1704828
Dolton MJ, Ray JE, Chen SC, Ng K, Pont LG, McLachlan AJ (2012) Multicenter study of voriconazole pharmacokinetics and therapeutic drug monitoring. Antimicrob Agents Chemother 56(9):4793–4799. https://doi.org/10.1128/AAC.00626-12
Zeng G, Shi L, Li H, Wang L, Zhu M, Luo J, Zhang Z (2019) Effect of cyclosporine a and polymorphisms in CYP2C19 and ABCC2 on the concentration of voriconazole in patients undergoing allogeneic hematopoietic stem cell transplantation. Xenobiotica; the fate of foreign compounds in biological systems:1–6. https://doi.org/10.1080/00498254.2019.1672907
Amsden JR, Gubbins PO (2017) Pharmacogenomics of triazole antifungal agents: implications for safety, tolerability and efficacy. Expert Opin Drug Metab Toxicol 13(11):1135–1146. https://doi.org/10.1080/17425255.2017.1391213
Phillips IR, Shephard EA (2017) Drug metabolism by flavin-containing monooxygenases of human and mouse. Expert Opin Drug Metab Toxicol 13(2):167–181. https://doi.org/10.1080/17425255.2017.1239718
Shirasaka Y, Chaudhry AS, McDonald M, Prasad B, Wong T, Calamia JC, Fohner A, Thornton TA, Isoherranen N, Unadkat JD, Rettie AE, Schuetz EG, Thummel KE (2016) Interindividual variability of CYP2C19-catalyzed drug metabolism due to differences in gene diplotypes and cytochrome P450 oxidoreductase content. Pharm J 16(4):375–387. https://doi.org/10.1038/tpj.2015.58
Agrawal V, Huang N, Miller WL (2008) Pharmacogenetics of P450 oxidoreductase: effect of sequence variants on activities of CYP1A2 and CYP2C19. Pharmacogenet Genom 18(7):569–576. https://doi.org/10.1097/Fpc.0b013e32830054ac
Harmsen S, Meijerman I, Beijnen JH, Schellens JHM (2007) The role of nuclear receptors in pharmacokinetic drug-drug interactions in oncology. Cancer Treat Rev 33(4):369–380. https://doi.org/10.1016/j.ctrv.2007.01.003
Dapia I, Garcia I, Martinez JC, Arias P, Guerra P, Diaz L, Garcia A, Ochoa D, Tenorio J, Ramirez E, Roman M, Gordo G, Saiz-Rodriguez M, Frias J, Abad-Santos F, Lapunzina P, Carcas AJ, Borobia AM (2019) Prediction models for voriconazole pharmacokinetics based on pharmacogenetics: AN exploratory study in a Spanish population. Int J Antimicrob Agents 54(4):463–470. https://doi.org/10.1016/j.ijantimicag.2019.06.026
Veringa A, ter Avest M, Span LFR, van den Heuvel ER, Touw DJ, Zijlstra JG, Kosterink JGW, van der Werf TS, Alffenaar JWC (2017) Voriconazole metabolism is influenced by severe inflammation: a prospective study. J Antimicrob Chemother 72(1):261–267. https://doi.org/10.1093/jac/dkw349
Jourdil JF, Tonini J, Stanke-Labesque F (2013) Simultaneous quantitation of azole antifungals, antibiotics, imatinib, and raltegravir in human plasma by two-dimensional high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B 919:1–9. https://doi.org/10.1016/j.jchromb.2012.12.028
Dote S, Sawai M, Nozaki A, Naruhashi K, Kobayashi Y, Nakanishi H (2016) A retrospective analysis of patient-specific factors on voriconazole clearance. J Pharm Health Care Sci 2:10–16. https://doi.org/10.1186/s40780-016-0044-9
Li ZW, Peng FH, Yan M, Liang W, Liu XL, Wu YQ, Lin XB, Tan SL, Wang F, Xu P, Fang PF, Liu YP, Xiang DX, Zhang BK (2017) Impact of CYP2C19 genotype and liver function on voriconazole pharmacokinetics in renal transplant recipients. Ther Drug Monit 39(4):422–428
Morgan ET (2009) Impact of infectious and inflammatory disease on cytochrome P450-mediated drug metabolism and pharmacokinetics. Clin Pharmacol Ther 85(4):434–438. https://doi.org/10.1038/clpt.2008.302
Redl H, Schiesser A, Togel E, Assicot M, Bohuon C (2001) Possible role of TNF on procalcitonin release in a baboon model of sepsis. Shock 16(1):25–27. https://doi.org/10.1097/00024382-200116010-00005
Shah RR, Smith RL (2015) Inflammation-induced phenoconversion of polymorphic drug metabolizing enzymes: hypothesis with implications for personalized medicine. Drug Metab Dispos 43(3):400–410. https://doi.org/10.1124/dmd.114.061093
Yanni SB, Annaert PP, Augustijns P, Ibrahim JG, Benjamin DK, Thakker DR (2010) In vitro hepatic metabolism explains higher clearance of Voriconazole in children versus adults: role of CYP2C19 and flavin-containing monooxygenase 3. Drug Metab Dispos 38(1):25–31. https://doi.org/10.1124/dmd.109.029769
Zhang HF, Li ZH, Liu JY, Liu TT, Wang P, Fang Y, Zhou J, Cui MZ, Gao N, Tian X, Gao J, Wen Q, Jia LJ, Qiao HL (2016) Correlation of cytochrome P450 oxidoreductase expression with the expression of 10 isoforms of cytochrome P450 in human liver. Drug Metab Dispos 44(8):1193–1200. https://doi.org/10.1124/dmd.116.069849
Mbatchi LC, Brouillet JP, Evrard A (2018) Genetic variations of the xenoreceptors NR1I2 and NR1I3 and their effect on drug disposition and response variability. Pharmacogenomics 19(1):61–77. https://doi.org/10.2217/pgs-2017-0121
Prakash C, Zuniga B, Song CS, Jiang S, Cropper J, Park S, Chatterjee B (2015) Nuclear receptors in drug metabolism, drug response and drug interactions. Nuclear Receptor Res 2. https://doi.org/10.11131/2015/101178
Acknowledgments
We thank all patients who contributed to this work. We thank Professor Shusen Sun from College of Pharmacy and Health Science at Western New England University, USA, for his valuable advice.
Funding
This work was supported by a grant from the Natural Science Foundation of Hunan Province (No.2017JJ2398).
Author information
Authors and Affiliations
Contributions
Zanling Zhang and Jia Luo designed the study. Guangting Zeng and Linlin Wang performed the date analysis and wrote the manuscript. Lihong Shi and Huilan Li recuited patients. Miaomiao Zhu extracted the DNA.
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 82 kb)
Rights and permissions
About this article
Cite this article
Zeng, G., Wang, L., Shi, L. et al. Variability of voriconazole concentrations in patients with hematopoietic stem cell transplantation and hematological malignancies: influence of loading dose, procalcitonin, and pregnane X receptor polymorphisms. Eur J Clin Pharmacol 76, 515–523 (2020). https://doi.org/10.1007/s00228-020-02831-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-020-02831-1