Abstract
Reproductive parameters were assessed in 64 male common dolphins (Delphinus delphis) examined post-mortem from strandings and bycatch in New Zealand between 1999 and 2020. The stages of male sexual maturation were assessed using morphological measurements and histological examination of testicular tissue. Age was determined via growth layer groups (GLGs) in teeth. The average age (ASM) and length (LSM) at attainment of sexual maturity were estimated to be 8.8 years and 198.3 cm, respectively. Individual variation in ASM (7.5–10 years) and LSM (190–220 cm) was observed in New Zealand common dolphins. However, on average, sexual maturity was attained at a similar length but at a marginally younger age (< 1 year) in New Zealand compared to populations in the Northern Hemisphere. All testicular variables proved better predictors of sexual maturity compared to demographic variables (age and total body length), with combined testes weight the best outright predictor of sexual maturity. Reproductive seasonality was observed in male common dolphins, with a significant increase in combined testes weight in austral summer. This aligns with most other studied populations, where seasonality in reproduction is typically observed. Given the known anthropogenic impacts on New Zealand common dolphins, we recommend that these findings be used as a baseline from which to monitor population-level changes as part of conservation management efforts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An understanding of male reproductive parameters can provide insights into population structure, intraspecific competition, mating behaviours and systems, group structure, and sexual dimorphism within a population or species (delBarco-Trillo and Ferkin 2004; Dixson and Anderson 2004; Bronson 2009; De Bruyn et al. 2011). Within Cetacea (whales, dolphins, and porpoises), a wide range of reproductive parameters are displayed, including large variation in average age and length at attainment of sexual maturity and gonadal size (Clapham 1996; Ramos et al. 2000; Danilewicz et al. 2004; MacLeod and MacLeod 2009; Nicolosi and Loy 2019).
Spermatogenesis, the production of haploid spermatozoa, is the primary indicator of sexual development in male cetaceans (Plön and Bernard 2007; Katsumata 2010; Kemper et al. 2014). Reproduction is predominantly seasonal in cetaceans, with most males producing sperm only at certain times of the year (Sørensen and Kinze 1994; Neimanis et al. 2000; Vu et al. 2015; Robeck and O'Brien 2018). Sexual features of male cetaceans vary widely, but may include a prominent post-anal hump, large testes size, long penises, and retained bodily scarring caused by intraspecific interactions (Neumann et al. 2002; Dixson and Anderson 2004; Dines et al. 2014, 2015; Betty et al. 2019). In life-history studies, sexual maturity is typically assessed via the gross and histological examination of testes post-mortem (Reddy 1996; Murphy et al. 2005; Kesselring et al. 2019), although hormone analysis of blubber, plasma, and blow exhalant samples are increasingly used for live, free-ranging individuals (Hogg et al. 2009; Kellar et al. 2009; Galligan et al. 2018; Robeck et al. 2019).
Reproductive parameters of male cetaceans have been assessed across many species (Olesiuk et al. 2005; Matkin et al. 2014; Wells 2014; Betty et al. 2019; Plön et al. 2020), with variation often linked to body size and longevity. For example, male harbour porpoises (Phocoena phocoena) in Scottish waters attain sexual maturity at 5 years of age and at 132 cm in length (Learmonth et al. 2014); whereas male bottlenose dolphins (Tursiops aduncus) in the Indo-Pacific attain sexual maturity at c. 12 years of age and between 208 and 220 cm in length (Kemper et al. 2014). Furthermore, there is considerable variation between populations and ecotypes of the same species (Olesiuk et al. 1990; Robeck and Monfort 2006; Chen et al. 2011). For example, male harbour porpoises in the Celtic and Irish seas obtained sexual maturity at a significantly larger body length than those in the North Sea (Murphy et al. 2020). Additionally, individuals in this region experience mixing with the larger, recently proposed sub-species of Iberian harbour porpoises, which would also increase the length at sexual maturity shown by males (Murphy et al. 2020).
The reproductive biology of male common dolphins (Delphinus delphis) has been examined in several Northern Hemisphere populations including the western (Westgate and Read 2007) and eastern (Murphy et al. 2005) North Atlantic, the eastern tropical Pacific (Oliver 1973) and the North Pacific (Ferrero and Walker 1995). Less is known for common dolphins in the Southern Hemisphere, with only a single study in the western South Atlantic (Grandi et al. 2022). Such paucity of data has been of concern for Australasia, where common dolphins are subject to several anthropogenic impacts including pollutants (Lavery et al. 2008; Stockin et al. 2021a, b), tourism (Neumann and Orams 2005, 2006; Stockin et al. 2008; Meissner et al. 2015), and fisheries bycatch (Hamer et al. 2008; Stockin et al. 2009; Thompson et al. 2013; Allen et al. 2014; Mackay and Goldsworthy 2017; Barceló et al. 2021; Parra et al. 2021).
As part of the wider Australasian population, New Zealand common dolphins demonstrate significant genetic connectivity with their Australian counterparts (Barceló et al. 2021), which raises concerns about the sustainability of levels of fisheries bycatch on both sides of the Tasman Sea (Du Fresne et al. 2007; Stockin et al. 2009; Thompson et al. 2013; Allen et al. 2014; Mackay and Goldsworthy 2017). However, management units are currently based on Australia and New Zealand having separate populations (Barceló et al. 2021). Given recent declines of common dolphins observed internationally (e.g., in the Mediterranean Sea; Bearzi et al. 2003; Piroddi et al. 2011; Vella et al. 2021), careful management based on a fuller understanding of the reproductive biology of male common dolphins on both sides of the Tasman Sea is required to ensure the long-term viability of the species in this region. As male life history is comparatively less described (Chivers 2009), this study will complement recently published data on female common dolphin reproductive parameters (Palmer et al. 2022), offering population-level insights to common dolphins in New Zealand waters.
Here, we assessed the reproductive biology of male common dolphins in New Zealand waters using histological examination of testicular tissue and a set of testicular measures (combined testes length, combined testes weight, an index of testicular development, and mean seminiferous tubule diameter). Specifically, these testicular parameters were used to examine (1) how testis characteristics change with age, body length, and sexual maturity, (2) the average age and length at attainment of sexual maturity, (3) potential indicators of sexual maturity, and (4) evidence of reproductive seasonality in mature males.
Materials and methods
Sample collection
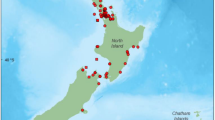
Reproductive data were collected and assessed post-mortem in 64 male common dolphins following Geraci and Lounsbury (2005). The sample size included 56 individuals (54 independent events) that either live-stranded or were found beachcast on the New Zealand coastline between 1999 and 2020. Additionally, six individuals incidentally captured within the commercial fishery for jack mackerel (Trachurus novaezelandiae) off the west coast of the North Island between 2001 and 2003 were included (Fig. 1). The origin of a further 2 males remain unknown.
Testes and associated epididymides were removed and the testes weighed without the associated epididymis to the nearest 0.1 g. Measurements of the length of each testis were taken to the nearest 0.1 cm. Small samples (approx. 1 cm3) were subsequently dissected from each testis and epididymis and were fixed in 10% neutral buffered formalin. Total body length (TBL) was measured to the nearest 0.5 cm. Teeth were carefully extracted for age determination following methods outlined in Murphy et al. (2014). Decomposition state was further noted for each individual (fresh, mild, and moderate) as per Stockin et al. (2007).
Age estimation
Age was estimated by examining decalcified, stained thin sections of tooth from an individual (Murphy et al. 2014). A binocular microscope (10–40 × magnification) was used to examine sections and age was estimated by counting the annual growth layer groups (GLGs) in the dentine as described by Myrick Jr et al. (1983) and Lockyer (1995). Tooth sections were initially read blind (i.e., with no prior biological information known), by at least two of three experienced readers (SM, EB, and EP). Best age estimates or age ranges were subsequently compared, and in the case of any disagreement, further teeth were sectioned and examined again until a final estimate was determined (after Westgate and Read 2007). Individuals that could not be aged reliably (i.e., due to tooth damage or wear) were excluded from further analysis. A neonate was identified if the neonatal line was not present or just forming in the dentine of the tooth.
Histological assessment of reproductive organs
Stages of sexual maturity were determined by histological examination of testicular tissue (Murphy et al. 2005; Betty et al. 2019). Testicular tissue was processed using standard histological techniques, i.e., by dehydration, clearing, and embedding in paraffin wax. Tissues were sectioned at 5 µm, stained with H&E (haematoxylin and eosin), and mounted on glass slides. Histological slides were examined microscopically (100–400x), and the stage of sexual maturity was determined via assessment of all seminiferous tubules in approximately 1 cm2 section of the testicular tissue. Parameters used to assess maturity included the mean diameter of seminiferous tubules (mean DT), the relative proportion of Sertoli cells, interstitial tissue, germinal cells (spermatogonia, spermatocytes, spermatids, and spermatozoa), the activity in the epididymis, and the presence and proportion of spermatozoa in the epididymis (Murphy 2004; Murphy et al. 2005). Neimanis (1996) suggested that in slightly autolysed tissue, the basement membrane may detach from the seminiferous epithelium in some areas, but this would not significantly change the diameter measurements of the tubules in comparison to fresh tissue. Therefore, a mean diameter of the seminiferous tubules was taken by measuring the basement membranes of ten tubule cross-sections. These measurements were collected from images taken using Axiocam 105 with associated Zeiss software (Zeiss 2013) and ImageJ, an image analysis system (Schneider et al. 2012). Only clear circular tubules were measured to ensure an accurate cross-section of the longitudinal axis were taken, in accordance with other cetacean reproductive studies (Neimanis et al. 2000; O’Hara et al. 2002; Betty et al. 2019). If the tissue was observed to be in a state of moderate or advanced autolysis, measurements were not taken as the basement membrane could not be clearly distinguished.
Males were classified into three maturity stages; immature, pubescent, and mature (Murphy et al. 2005; Kemper et al. 2014). Maturity stages were based on the presence and proportion of cell types in the seminiferous tubules, as follows: immature individuals had only Sertoli cells and spermatogonia present; pubescent individuals had both spermatogonia and spermatocytes present; mature individuals had all stages of spermatogenesis present in the tubules (including spermatids and spermatozoa).
Statistical analysis
Models of maturity status given length, age, and testicular measurements
A dataset of six individual-level variables was compiled for 35 males, including two demographic variables (age and TBL) and four testicular variables (combined testes length, combined testes weight, index of testicular development, and mean seminiferous tubule diameter). The index of testicular development was calculated as the combined testes weight in grams (excluding epididymis) divided by the combined testes length in millimetres (Hohn et al. 1985). Relationships among these six variables were explored using charts and Spearman’s rank correlation coefficients. The index variable was log-transformed as it was found to be a better predictor of sexual maturity than its raw form.
The average age (ASM) and length (LSM) at attainment of sexual maturity were estimated for male common dolphins using two methods: (1) Bayesian modelling as detailed hereafter and (2) the sum-of-fraction of immature (SOFI) method (Hohn 1989, see S1 in Supplementary Material).
Progression through the maturity stages (immature, pubescent, and mature) was modelled with Bayesian cumulative logit models fitted with the ‘brms’ package for R (Bürkner 2017). To compare the utility of each of the individual-level variables to predict maturity stage, each variable was used as the single predictor variable (x) in turn. Maturity stage (Y) was treated as an ordinal variable with three categories (immature, pubescent, and mature) represented as k = {1, 2, 3}, respectively. The probability of a male being in stage k or below (πk = P(Y ≤ k)) was modelled as
for k = 1, 2; π3 = 1 − π2. For all three parameters (α1, α2, and β), weakly informative prior distributions [Student′ s t(3, 0, 10)] were assumed. To estimate x50, the value of x at which 50% of males were classified as mature, the posterior distribution of α2⁄β was used. The posterior distribution was then summarised using the mean and 95% credible intervals (‘CrI’; based on 2.5% and 97.5% percentiles).
Comparison of models
The relative utility of the demographic and testicular variables as indicators of maturity in the cumulative logic models was compared using the Bayesian LOO (Leave-One-Out) estimate of the expected log pointwise predictive density (using the ‘elpd_loo’ package for R; Vehtari et al. (2017)). ELPD-LOO is a criterion used to estimate out-of-sample predictive accuracy—that is, how accurately a model will predict new data that were not used in the fitting of the model (Vehtari et al. 2017). ELPD-LOO scores are used to compare models fit to the same dataset (Vehtari et al. 2017). In our case, not all individuals had data for all six variables available (due to tissue quality) and so only 35 complete cases were available for comparing all the fitted models with ELPD-LOO. There was only one case of a ‘pubescent’ male in the ‘complete’ dataset, so this case was excluded from the comparison of the models. Thus, a dataset of 34 complete cases was used for comparisons and the pubescent category was omitted. For the more specific comparison of the models with age and TBL as predictor variables, we used a larger data set (n = 52) with all the complete cases for these two predictor variables. Two models were fitted to each of the four testicular variables (combined testes weight, combined testes length, index of testicular development, and mean diameter of the seminiferous tubules) as these variables had skewed distributions. One of the models used the raw values (x) and the other used the log-transformed values (log x). These models were compared based on the ELPD-LOO criterion, which indicated that the log-transformed variables were a better fit. Therefore, we present the models that use the log-transformed testicular variables.
Quantifying reproductive seasonality
To assess potential reproductive seasonality in mature males, the variation in mean diameter of seminiferous tubules and combined testes weight between seasons, and across the year (using Julian dates) was tested using Kruskal–Wallis and randomisation tests. Due to the small sample size, males were grouped into austral seasons instead of months following Murphy et al. (2005) and Westgate and Read (2007). Austral seasons were defined as summer (December–February), autumn (March–May), winter (June–August), and spring (September–November). Differences between combined testes weight and mean seminiferous tubule diameter among seasons were assessed with t tests. The testicular and seasonality data were first tested for normality using Shapiro–Wilk tests.
All statistical analyses were conducted using R version 2021.4.1.1 (R Development Core Team 2021).
Results
Stages of sexual maturation
Of the 64 males assessed, 37 (58%) and 24 (38%) were classified as immature and mature, respectively. A further 3 (5%) were classified as pubescent (Table 1). Of the 59 males that had decomposition state recorded, 18 (31%) were classified as fresh, 27 (46%) as mild, and 14 (23%) as moderate.
From the sample set, 18 individuals had missing data and/or testicular tissue too autolysed to assess testicular features such as seminiferous tubule measurements. Histological appearances of immature, pubescent, and mature testis and epididymis are shown in Fig. 2 and described herein.
Histological appearance of immature, pubescent, and mature common dolphin (Delphinus delphis) testes examined from New Zealand waters (1999–2020). Shown are two individuals for each maturity stage and the scale bar is 100 µm. a KS14-63Dd; immature, TBL 95 cm, combined testes weight 3.1 g, mean seminiferous tubule diameter 18.95 µm, and b KS20-30Dd; immature, TBL 163 cm, combined testes weight 10 g, mean seminiferous tubule diameter 27.93 µm. c KS19-12Dd; pubescent, TBL 201 cm, combined testes weight 429 g, mean seminiferous tubule diameter 52.68 µm, and d KS10-78Dd; pubescent, TBL 201 cm, mean seminiferous tubule diameter 62.45 µm. e KS20-05Dd; adult, mature, TBL 210 cm, combined testes weight 1505 g, mean seminiferous tubule diameter 109.18 µm, and f KS17-01Dd; adult, mature, TBL 208 cm, combined testes weight 2430 g, mean seminiferous tubule diameter 165.53 µm
Immature testes (Fig. 2a, b, n = 37) had seminiferous tubules that were narrow (x̄ = 17.3 ± 0.9 µm; range 10.5–27.9), tightly arranged, and embedded in abundant interstitial tissue. Enclosed by the basement membrane were one or two layers of two types of cells: the supportive Sertoli cells, and spermatogonia (germinal cells). These cells were undergoing mitosis and neatly aligned the edges of the tubules. The epididymis was empty and exhibited a resting epithelium, indicating that it was undeveloped. In immature testes (and epididymides), no spermatocytes, spermatids, or spermatozoa were observed.
Pubescent testes (Fig. 2c, d, n = 3) contained medium-sized seminiferous tubules (x̄ = 57.8 ± 4.9 µm; range 52.7–62.5), with spermatogonia and spermatocytes present. A reduction in interstitial tissue and Sertoli cells was noted. Zonation of the spermatogonia and spermatocytes was evident as spermatogonia were undergoing meiosis to produce spermatocytes. No spermatozoa present in the epididymis.
Mature testes (Fig. 2e, f, n = 24) contained large seminiferous tubules with a mean diameter of 92.0 ± 8.2 µm (range 51.1–165.5). All cell types involved in spermatogenesis were present, with spermatocytes, spermatids, and spermatozoa observed in high proportions. Low proportions of interstitial tissue, Sertoli cells, and spermatogonia were noted. Spermatozoa were present in the lumen of the tubules and the epididymis, and the epididymis was enlarged.
Models of maturity status given with demographic variables
Males ranged from 89 to 241 cm in TBL (n = 62; two individuals were excluded due to missing TBL data), with a modal size class of 211–220 cm (median = 178 cm; Fig. 3a). The estimated age of male common dolphins ranged from 0 to 26 years (n = 55), with two males (KS08-11Dd and KS18-01Dd) only having minimum ages estimated. Of the males that had sexual maturity stage determined, 82% (n = 40) of individuals were aged less than 15 years. Immature males ranged from 89 to 200 cm in length and 0 to 9 years in age. Pubescent males ranged from 201 to 220 cm in length and 9 to 10 years in age. Mature males ranged from 190 to 241 cm in length and 7.5 to 26 years in age. Age and TBL increased with maturity stage (Table 1 and Fig. 3), though some overlap between age 8 and 9 years, and 195 cm and 199 cm were evident.
Bayesian modelling estimated the ASM and LSM to be 8.8 years (95% CrI = 7.8–9.8, n = 51) and 198.3 cm (95% CrI = 191.4–204.9, n = 61; Table 2 and Fig. 5), respectively. Model comparison based on ELPD_LOO scores for the dataset with both age and length (n = 34) provided little statistical support for a difference between the utility of age and TBL as indicators of maturity stage, given the difference in ELPD_LOO scores (1.31) was less than the respective standard errors (SE = 3.68 and 3.19). Using the larger ‘age vs TBL’ dataset (n = 52), the ELPD_LOO score for the model with age was 2.2 units greater than that of the model using TBL, but the large standard error (SE = 4.1) once again indicated no evidence of one being a better predictor than the other. Applying the SOFI method for comparison across previously studied populations (Ferrero and Walker 1995; Murphy et al. 2005; Westgate and Read 2007), the ASM and LSM were estimated as 8.8 years (SE = 0.16, n = 11) and 200 cm (SE = 0.15, n = 13), respectively, where n is the number of individuals in the indeterminate age or length classes (i.e., age or length classes in which both immature and mature individuals occur in the sample).
Models of maturity status given with testicular variables
Testicular variables (combined testes weight, combined testes length, index of testicular development, and mean seminiferous tubule diameter) for immature, pubescent, and mature male common dolphins are summarised in Table 2. The testicular and demographic variables were all highly correlated with each other (Fig. 5). A general pattern of increasing size with maturity stages was observed with testicular variables, though some overlap between the stages was evident (Table 1, Fig. 4). A significant difference (p < 0.001) between the combined testes weight of immature (x̄ ± SE: 15.3 ± 2.6 g) versus mature (x̄ ± SE: 1921.2 ± 294.4 g) individuals was detected. A similar difference (p < 0.001) was further observed in the mean seminiferous tubule diameter between immature (x̄ ± SE: 17.3 ± 0.9 µm) and mature (x̄ ± SE: 92.0 ± 8.2 µm) males.
Demographic variables (age and TBL) versus testicular variables (combined testes length [age: n = 52, TBL: n = 59], combined testes weight [age: n = 52, TBL; n = 60], log of index of testicular development [age: n = 51, TBL: n = 58], and mean diameter of seminiferous tubules [mean DT; age: n = 37, TBL: n = 45]) for male common dolphins (Delphinus delphis) examined in New Zealand waters (1999–2020)
Combined testes weight and length were both larger in sexually mature animals (8.8 years of age, 198.3 cm TBL). However, significant variation in combined testes weight (492–5796 g) and combined testes length (275–965 mm) remained evident in mature males (Fig. 4). A steep increase in the mean seminiferous tubule diameter was further observed at approximately 190–200 cm TBL and 8–10 years of age (Fig. 4). An estimated 50% of males had reached sexual maturity at 351.38 mm combined testes length, 301.62 g combined testes weight, 0.92 index of testicular development, and 49.97 µm seminiferous tubule diameter (Table 2, Fig. 5). Comparison of the modelled variables with ELPD_LOO indicated that combined testes weight (g) was the best indicator of sexual maturity (Table 2). All testicular variables were better predictors of sexual maturity than age or TBL.
Bayesian cumulative logit regression of the sexual maturation of male common dolphins (Delphinus delphis) examined in New Zealand waters (1999–2020) through three stages (immature, pubescent, and mature) modelled as a function of one of six individual measures: age, total body length, combined testes weight, combined testes length, index of testicular development (combined testes weight/combined testes length), and mean seminiferous tubule diameter (mean DT)
Note that all variables, demographic and testicular, were highly positively correlated (Spearman’s rank correlation coefficients). The type and colour of the data points represent the individuals’ sexual maturity stage: immature = light blue circle, pubescent = medium blue triangle, and mature = dark blue square. The index of testicular development was log-transformed as it was a better predictor in that form.
Each measure has two plots shown (Fig. 5). The upper plot shows each maturity stage and the data values for the measure within each stage. The lines on the plot represent the posterior predictions of the transitions through stages. The lower plot shows the probability of being in each of the three stages where an increase in maturity stage is shown by lighter to darker lines and intervals, which track left to right. The thick lines show the estimated mean and the 95% credible intervals. The black points show the mean, and the thin horizontal lines show the 95% credible intervals of the estimated value of x. This is the point at which 50% of males were classified as pubescent (left point and line) and mature (right point and line). The measures for age and TBL on the x-axis are shown on the raw scale, but the models were fitted to log-transformed testicular variables, as indicated.
Reproductive seasonality
Mean combined testes weight (g) differed among seasons, indicating reproductive seasonality (Kruskal–Wallis test, p ≤ 0.01). The greatest and smallest combined testes weights were recorded in austral summer (December–February, x̄ ± SE: 8923 ± 378 g), and winter (June–August, x̄ ± SE: 710 ± 505 g; Fig. 6b), respectively. This aligns with the higher combined testes weights recorded at the start (Julian date 0–50) and end (Julian date 300–365) of the year (Fig. 6a). Significant differences in combined testes weight between seasons (p = 0.025) were observed. However, no difference in mean tubule diameter was noted between seasons (Kruskal–Wallis test, p = 0.106, Fig. 6c and Fig. 6d).
Annual variation in a combined testes weight (g) vs. Julian date (n = 20) and c mean seminiferous tubule diameter (µm) vs. Julian date (n = 15) and seasonal variation in b combined testes weight (g, n = 20) and d mean seminiferous tubule diameter (µm, n = 15) of mature male common dolphins (Delphinus delphis) examined in New Zealand waters between 1990 and 2020
Discussion
Stages of sexual maturation
This study examined the reproductive biology of 64 male common dolphins, including both stranded and bycaught individuals. This allowed comparison with prior international studies of male reproduction in common dolphin populations in both the eastern (n = 212, Murphy et al. 2005) and western (n = 161, Westgate and Read 2007) North Atlantic, and the western South Atlantic (n = 54, Grandi et al. 2022). Only three male common dolphins examined in our study were classified as pubescent, with just one having all demographic and testicular variables required for inclusion in Bayesian modelling.
Comparison of predictors of sexual maturity
For common dolphins, the sexual maturity status, assessed via histological examination, was strongly related to both the demographic variables (age and TBL length) and testicular measurements. In particular, sexual maturity is characterised by a rapid increase in the size of the testis and seminiferous tubules (Murphy et al. 2005; Westgate and Read 2007; this study). This has been observed in other delphinid species such as bottlenose dolphins (Tursiops aduncus and Tursiops truncatus; Kasuya et al. 1997; Kemper et al. 2014) and pilot whales (Globicephala melas edwardii; Betty et al. 2019). The best predictor of sexual maturity in New Zealand common dolphins was combined testes weight, which aligns with international studies (Murphy et al. 2005; Westgate and Read 2007). However, there was substantial overlap in the testicular and demographic variables among the maturity stages, likely due to variation in the maturation process among individuals (Murphy et al. 2005; Kemper et al. 2014; Betty et al. 2019).
The demographic variables, age and TBL, were useful predictors of sexual maturity, although they were not as accurate as the testicular variables (Fig. 5). This has important management implications, as TBL is the easiest and most accessible parameter to collect in the field. Therefore, when post-mortem examinations cannot be undertaken, precise linear measurements of TBL are useful. Additionally, the usefulness of both demographic and testicular variables may change with a larger dataset.
Several challenges come with assessing sexual maturity via histological examination of testicular tissue. For example, testicular tissue of odontocetes is susceptible to rapid post-mortem autolysis (Laws 1961; Kemper et al. 2014; Betty et al. 2019) and can be further impacted by freeze–thaw artefacts which are known to affect histological quality (Schäfer and Kaufmann 1999). In the present study, the testes of some dolphins (n = 22) were frozen prior to fixation, while others were fixed fresh (n = 23). Such challenges can be mitigated by the exploration of alternative methods to assess sexual maturity in males. For example, blubber testosterone methods, from biopsy samples, can be assessed in live, free-ranging animals and takes away the need to rely on testicular tissue (Kellar et al. 2009). Like any method of data collection, there are still advantages and disadvantages to these alternative methods. Other distinguishing features, such as the prominent post-anal hump, have been used real time (in field) or retrospectively (via photo-ID) to identify mature male common dolphins (Heyning and Perrin 1994; Neumann et al. 2002; Murphy 2004; Ngqulana et al. 2017).
Attainment of sexual maturity
Male common dolphins in New Zealand waters attain sexual maturity at an average body length (TBL) of 198.3 cm (n = 61) as estimated by the logistic regression method (or 200 cm, n = 11, as estimated using the SOFI method). These estimates are comparable to previous studies of male common dolphins (Table S2). In the eastern North Atlantic, sexual maturity is attained between approximately 200 (Collet and Saint-Girons 1984; Murphy et al. 2005) and 204 cm (Read et al. 2019). This also aligns with the eastern tropical Pacific where males are estimated to attain sexual maturity at approximately 202 cm (Oliver 1973; Gurevich and Stewart 1978). In the western North Atlantic, males are reported to attain sexual maturity at c.215 cm (Westgate 2005). In contrast, males from the North Pacific attain sexual maturity at c.182 cm (Ferrero and Walker 1995). This likely reflects males in the two populations having different asymptotic lengths (i.e., length at physical maturity). Males in the North Pacific attain physical maturity at 188 cm, whereas males in the western North Atlantic attain physical maturity at 221.5 cm. Asymptotic length is positively correlated with length at sexual maturity, meaning the larger the asymptotic length, the greater the total body length at sexual maturity (Stamps et al. 1998). In the western North Atlantic, immature and mature males overlap between 184 and 209 cm (Grandi et al. 2022).
The average age at attainment of sexual maturity in New Zealand common dolphins is 8.77 years (n = 51) as estimated by the logistic regression method (or 8.75 years, n = 13, as estimated using the SOFI method). This is the youngest reported ASM estimate for male common dolphins in any international population (Table S2). In the eastern North Atlantic (Irish and French dataset), males attain sexual maturity at 11.86 years (Murphy et al. 2005), and at approximately 10.5 years in North-West Spain (Read et al. 2019) and the North Pacific (Ferrero and Walker 1995). In the western North Atlantic, sexual maturity is attained at 9.45 years (Westgate and Read 2007). In Argentine waters, immature and mature males overlap in age between 6 and 9 years (Grandi et al. 2022). Due to a small sample size (n = 52), LSM and ASM estimates were not obtained for the western South Atlantic.
Geographic variation in attainment of sexual maturity can arise from differences in biological and/or environmental factors, including habitat, diet composition, anthropogenic impacts, and population size and density (Murphy 2004; Clutton-Brock and Sheldon 2010; Kemper et al. 2014; Alberts 2019; Barceló et al. 2022). Interestingly, male common dolphins in New Zealand attain sexual maturity at a similar TBL to other populations, despite being at the younger end of reported ASM in international populations. Asymptotic length of males from New Zealand waters (212.1 cm, Palmer 2023), align with asymptotic lengths for males in the eastern North Atlantic (211.6–214 cm; Murphy et al. 2005) and western South Atlantic (211 cm; Grandi et al. 2022). Additionally, New Zealand waters are relatively productive (Murphy et al. 2001), potentially providing abundant prey to support faster growth rates, assuming no disruption to the food web (Stockin et al. 2022, 2023). There is no evidence, however, that male common dolphins in New Zealand have an expedited growth rate (Palmer 2023) compared to other populations (Westgate 2005; Murphy et al. 2005).
Intra-specific competition for access to females may reflect the slightly older age at sexual maturity for males in comparison to females in New Zealand (Palmer et al. 2022). This longer stage of growth before sexual maturity could indicate sexual dimorphism within the population, as a larger size is more favourable for competition (Read et al. 1993; Murphy et al. 2005). Additionally, male common dolphins in New Zealand waters obtain sexual maturity at a marginally younger age compared to their international counterparts (Ferrero and Walker 1995; Murphy et al. 2005; Westgate and Read 2007; Read et al. 2019). This may indicate earlier allocation of resources to testicular mass and the post-anal hump (Ngqulana et al. 2017). Such a strategy may reflect the mating system of common dolphins in New Zealand, where females mate with many males, inducing sperm competition (Murphy et al. 2005).
Alternative explanations for the younger ASM are methodological error and a small sample size. While aging cetaceans is not without error (Hohn 1989; Rosas et al. 2003), an underestimated ASM is unlikely in the current study due to strict quality controls and best practice applied (Murphy et al. 2014). Specifically, blind readings were made across multiple experienced readers, with no apriori biological knowledge of the individual. The sample size for this study is smaller compared to most other studies on male reproduction in common dolphins (e.g., Murphy et al. 2005), and so, this may impact the outputs. The authors acknowledge the small sample size of this study, which spans more than a quarter of a century, offering a baseline for which mortality effects on the populations reproductive biology can be monitored. With increasing sample size, the aim is to establish whether the reproductive parameters reported here represent a natural baseline for the population and identify any temporal variation should it exist.
Reproductive seasonality
Reproductive seasonality was evident in New Zealand male common dolphins, with the largest combined testes weight observed in austral summer (2898.5 ± 464.2 g). Combined testes weight was, on average, three times heavier than in austral winter (710 ± 505 g). Fluctuations in combined testes weight across seasons aligned with testicular activity, which was also greatest in austral summer and lowest in austral winter. Similar patterns have been observed in Northern Hemisphere populations. For example, in the western North Atlantic, the mean mass of regressed testes was 802.9 ± 455.5 g, compared to 4049.6 ± 1317.4 g for testes in full production (Westgate and Read 2007). The full production peak in the western North Atlantic was observed in July (boreal summer) and aligned with the female reproductive cycle for that region. Reproductive seasonality is also observed in the eastern North Atlantic common dolphin population. Spermatogenesis occurs throughout the year, with a marked increase in testicular activity and mass from late May to September. A more active period is reported in July and August, which aligns with the female reproductive cycle (Murphy et al. 2005, 2009).
For males to maximise reproductive output, they align their reproductive cycle with females (Pomeroy 2011). Reproduction tends to also be associated with the availability of resources (Clapham 1996; Bronson 2009). Interestingly, seasonality is not so distinctly observed in females in New Zealand waters, which is more similar to common dolphins examined from the eastern tropical Pacific (Danil and Chivers 2007). For female common dolphins in New Zealand waters, breeding occurs year-round with slight peaks between August and November, which corresponds to late austral winter to late austral spring (Palmer et al. 2022). This does not align with testicular mass (which is associated with sperm production) of male common dolphins in New Zealand waters which is greatest in austral summer. Common dolphins in New Zealand waters have been previously reported to copulate outside the ‘mating period’ (Neumann 2001), which has been presumed to be recreational (Murphy et al. 2005). These previous observations could have been successful copulations as female common dolphins do not appear to have a defined mating period. Additionally, testicular activity occurs year-round, so there is the possibility that mating is still occurring successfully, even though the male and female ‘peaks’ do not align in the New Zealand population.
Male mating strategy
Although male investment in reproductive tissues varies among delphinid species, there is a strong relationship between body mass and testes mass in cetaceans (MacLeod 2010). Delphinids exhibit one of the greatest investments in male reproductive tissues as well as having the largest testes relative to body size of all amniotes (MacLeod and MacLeod 2009). For example, the most investment is noted in dusky dolphins (Lagenorhynchus obscurus) where testicular tissue accounts for up to 8% of total body mass (Van Waerebeek and Read 1994). Common dolphins were ranked comparatively high among cetacean species for the relative investment in testicular tissue as they account for 3.2% of the total body mass in the species (Macleod 2010). Such high reproductive investment is also observed in male common dolphins in both the Northern Hemisphere (Murphy et al. 2005; Westgate and Read 2007) and Southern Hemisphere (this study; Plon et al. 2012). For common dolphins in the western North Atlantic, the testes mass of sexually mature individuals is reported to vary between 2.3 and 4.4% of body weight (Westgate and Read 2007) and for males off of South Africa, on average, testes account for 2% of total body weight (Plon et al. 2012). Sperm competition has been suggested as the primary driver of the large reproductive investment in male common dolphins (Dixson and Anderson 2004), but not the only factor (Kenagy and Trombulak 1986; MacLeod 2010). Common dolphins are considered to have a promiscuous mating system which would support this large investment (Kelley et al. 2014; Ngqulana et al. 2017; Vella et al. 2021). Additionally, as post-anal humps are positively correlated with testis size, it is thought they may act as a visual signal via mate choice by females and establishing dominance hierarchies among males (Lewis 1991; Neumann et al. 2002; Murphy 2004; Murphy et al. 2005; Murphy and Rogan 2006). Future research into how post-anal humps are related to mating systems, maturity, and growth is recommended for the New Zealand population.
Conclusions
This study provides first insights into the reproductive biology of male common dolphins in New Zealand waters. Knowledge of parameters such as ASM, LSM, reproductive seasonality, and predictors of sexual maturity will aid better understanding of the long-term viability of this population and inform future management decisions. Specific information on the Australasian population is timely, since density-dependent changes may already be in play and, without prior baseline data, remain undetected. Reproductive parameters allow for early detection of population-level changes which is integral for effective species management and conservation. While support exists for a single Australasian population management plan due to the genetic structure of New Zealand and Australian common dolphins, ongoing trans-Tasman collaboration is crucial to allow management of Australasian common dolphins to be effective.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Alberts SC (2019) Social influences on survival and reproduction: Insights from a long-term study of wild baboons. J Anim Ecol 88:47–66. https://doi.org/10.1111/1365-2656.12887
Allen SJ, Tyne JA, Kobryn HT, Bejder L, Pollock KH, Loneragan NR (2014) Patterns of dolphin bycatch in a north-western Australian trawl fishery. PLoS ONE 9:e93178. https://doi.org/10.1371/journal.pone.0093178
Barceló A, Sandoval-Castillo J, Stockin KA, Bilgmann K, Attard CRM, Zanardo N, Parra GJ, Hupman K, Reeves IM, Betty EL, Tezanos-Pinto G, Beheregaray LB, Möller LM (2021) A matter of scale: population genomic structure and connectivity of fsheries at-risk common dolphins (Delphinus delphis) from Australasia. Front Mar Sci. https://doi.org/10.3389/fmars.2021.616673
Barceló A, Sandoval-Castillo J, Brauer CJ, Bilgmann K, Parra GJ, Beheregaray LB, Möller LM (2022) Seascape genomics of common dolphins (Delphinus delphis) reveals adaptive diversity linked to regional and local oceanography. BMC Ecol Evol 22:1–18. https://doi.org/10.1186/s12862-022-02038-1
Bearzi G, Reeves RR, Notarbartolo-di-Sciara G, Politi E, Canadas A, Frantzis A, Mussi B (2003) Ecology, status and conservation of short-beaked common dolphins Delphinus delphis in the Mediterranean Sea. Mamm Rev 33:224–252. https://doi.org/10.1046/j.1365-2907.2003.00032.x
Betty EL, Stockin KA, Smith AN, Bollard B, Orams MB, Murphy S (2019) Sexual maturation in male long-finned pilot whales (Globicephala melas edwardii): defining indicators of sexual maturity. J Mammal 100:1387–1402. https://doi.org/10.1093/jmammal/gyz086
Bronson F (2009) Climate change and seasonal reproduction in mammals. Philos Trans R Soc 364:3331–3340. https://doi.org/10.1098/rstb.2009.0140
Bürkner P-C (2017) brms: an R package for bayesian multilevel models using stan. J Statistical Softw. 80:1–28. https://doi.org/10.18637/jss.v080.i01
Chen I, Watson A, Chou LS (2011) Insights from life history traits of Risso’s dolphins (Grampus griseus) in Taiwanese waters: shorter body length characterizes northwest Pacific population. Mar Mamm Sci 27:E43–E64. https://doi.org/10.1111/j.1748-7692.2010.00429.x
Chivers SJ (2009) Cetacean life history. In: Perrin WF, Würsig B, Thewissen JGM (eds) Encyclopedia of marine mammals. Academic Press, Cambridge, pp 215–220. https://doi.org/10.1016/B978-0-12-373553-9.00055-9
Clapham PJ (1996) The social and reproductive biology of humpback whales: an ecological perspective. Mamm Rev 26:27–49. https://doi.org/10.1111/j.1365-2907.1996.tb00145.x
Clutton-Brock T, Sheldon BC (2010) Individuals and populations: the role of long-term, individual-based studies of animals in ecology and evolutionary biology. Trends Ecol Evol 25:562–573. https://doi.org/10.1016/j.tree.2010.08.002
Collet A, Saint Girons H (1984) Preliminary study of the male reproductive cycle in common dolphins, Delphinus delphis, in the eastern North Atlantic. Rep Int Whal Comm Spec Issue 6:355–360
Danil K, Chivers SJ (2007) Growth and reproduction of female shortbeaked common dolphins, Delphinus delphis, in the eastern tropical Pacifc. Can J Zool 85:108–121. https://doi.org/10.1139/z06-188
Danilewicz D, Claver JA, Pérez Carrera AL, Secchi ER, Fontoura NF (2004) Reproductive biology of male franciscanas (Pontoporia blainvillei)(Mammalia: Cetacea) from Rio Grande do Sul, southern Brazil. Fish Bull 102:581–592
De Bruyn P, Tosh CA, Bester MN, Cameron EZ, McIntyre T, Wilkinson IS (2011) Sex at sea: alternative mating system in an extremely polygynous mammal. Anim Behav 82:445–451. https://doi.org/10.1016/j.anbehav.2011.06.006
delBarco-Trillo J, Ferkin MH (2004) Male mammals respond to a risk of sperm competition conveyed by odours of conspecific males. Nature 431:446–449. https://doi.org/10.1038/nature02845
Dines JP, Otárola-Castillo E, Ralph P, Alas J, Daley T, Smith AD, Dean MD (2014) Sexual selection targets cetacean pelvic bones. Evol 68:3296–3306. https://doi.org/10.1111/evo.12516
Dines JP, Mesnick SL, Ralls K, May-Collado L, Agnarsson I, Dean MD (2015) A trade-off between precopulatory and postcopulatory trait investment in male cetaceans. Evolution 69:1560–1572. https://doi.org/10.1111/evo.12676
Dixson AF, Anderson MJ (2004) Sexual behavior, reproductive physiology and sperm competition in male mammals. Physiol Behav 83:361–371. https://doi.org/10.1016/j.physbeh.2004.08.022
Ferrero R, Walker W (1995) Growth and reproduction of the common dolphin, Delphinus delphis Linnaeus, in the ofshore waters of the North Pacifc Ocean. Fish Bull 93:483–494
Du Fresne SP, Grant AR, Norden WS, Pierre JP (2007) Factors afecting cetacean bycatch in a New Zealand trawl fshery. Department of Conservation Research & Development Series 282:18. https://www.doc.govt.nz/globalassets/documents/science-and-technical/drds282.pdf. Accessed 9 Jan 2022
Galligan TM, Schwacke LH, Houser DS, Wells RS, Rowles T, Boggs AS (2018) Characterization of circulating steroid hormone profiles in the bottlenose dolphin (Tursiops truncatus) by liquid chromatography–tandem mass spectrometry (LC–MS/MS). Gen Comp Endocrinol 263:80–91. https://doi.org/10.1016/j.ygcen.2018.04.003
Geraci JR, Lounsbury VJ (2005) Marine mammals ashore: a feld guide for strandings. National Aquarium in Baltimore, Baltimore
Grandi MF, Heredia FM, Sosa Drouville A, Dellabianca NA, Crespo EA (2022) Body growth and reproductive parameters of common dolphins from the southwestern Atlantic. Mar Mamm Sci 38:1470–1488. https://doi.org/10.1111/mms.12934
Gurevich VS, Stewart BS (1978) Structure of kill of the common dolphin Delphinus delphis from the eastern tropical Pacific in 1977. Southwest Fisheries Center, La Joll
Hamer DJ, Ward TM, McGarvey R (2008) Measurement, management and mitigation of operational interactions between the South Australian Sardine Fishery and short-beaked common dolphins (Delphinus delphis). Biol Conserv 141:2865–2878. https://doi.org/10.1016/j.biocon.2008.08.024
Heyning JE, Perrin WF (1994) Evidence for two species of common dolphins (genus Delphinus) from the eastern North Pacific Contributions in Science. Nat History Mus Los Angel Cty 442:1–35. https://doi.org/10.5962/p.226804
Hogg C, Rogers T, Shorter A, Barton K, Miller P, Nowacek D (2009) Determination of steroid hormones in whale blow: it is possible. Mar Mamm Sci 25:605–618. https://doi.org/10.1111/j.1748-7692.2008.00277.x
Hohn AA, Chivers SJ, Barlow J (1985) Reproductive maturity and seasonality of male spotted dolphins, Stenella attenuata, in the eastern tropical Pacific. Mar Mamm Sci 1:273–293. https://doi.org/10.1111/j.1748-7692.1985.tb00016.x
Hohn AA (1989) Variation in life-history traits: the influence of introduced variation. Dissertation, University of California.
Kasuya T, Izumisawa Y, Komyo Y, Ishino Y, Maejima Y (1997) Life history parameters of bottlenose dolphins off Japan. Rep Int Mar Biol Res Inst 7:71–107
Katsumata E (2010) Study on reproduction of captive marine mammals. J Reprod Dev 56:1–8. https://doi.org/10.1262/jrd.09-212E
Kellar NM, Trego ML, Marks CI, Chivers SJ, Danil K, Archer FI (2009) Blubber testosterone: A potential marker of male reproductive status in short-beaked common dolphins. Mar Mamm Sci 25:507–522. https://doi.org/10.1111/j.1748-7692.2009.00291.x
Kelley TC, Higdon JW, Ferguson SH (2014) Large testes and brain sizes in odontocetes (order Cetacea, suborder Odontoceti): the influence of mating system on encephalization. Can J Zool 92:721–726. https://doi.org/10.1139/cjz-2014-0044
Kemper CM, Trentin E, Tomo I (2014) Sexual maturity in male Indo-Pacific bottlenose dolphins (Tursiops aduncus): evidence for regressed/pathological adults. J Mammal 95:357–368. https://doi.org/10.1644/13-MAMM-A-007.1
Kenagy G, Trombulak SC (1986) Size and function of mammalian testes in relation to body size. J Mammal 67:1–22. https://doi.org/10.2307/1380997
Kesselring T, Viquerat S, Isseldijk L, Langeheine M, Wohlsein P, Gröne A, Bergmann M, Siebert U, Brehm R (2019) Testicular morphology and spermatogenesis in harbour porpoises (Phocoena phocoena). Theriogenology 126:177–186. https://doi.org/10.1016/j.theriogenology.2018.11.031
Lavery TJ, Butterfield N, Kemper CM, Reid RJ, Sanderson K (2008) Metals and selenium in the liver and bone of three dolphin species from South Australia, 1988–2004. Sci Total Environ 390:77–85. https://doi.org/10.1016/j.scitotenv.2007.09.016
Laws RM (1961) Reproduction, growth and age of southern fin whales. Discovery Rep 31:327–486
Learmonth JA, Murphy S, Luque PL, Reid RJ, Patterson IA, Brownlow A, Ross HM, Barley JP, Begoña Santos M, Pierce GJ (2014) Life history of harbor porpoises (Phocoena phocoena) in Scottish (UK) waters. Mar Mamm Sci 4:1427–1455. https://doi.org/10.1111/mms.12130
Lewis T (1991) The development of a postanal hump in male common dolphins, Delphinus delphis. 9th Biennial Conference on the Biology of Marine Mammals, Chicago. 5–9.
Lockyer C (1995) A review of factors involved in zonation in odontocete teeth, and an investigation of the likely impact of environmental factors and major life events on harbour porpoise tooth structure. Oceanogr Lit Rev 10:1042
Mackay AI, Goldsworthy SD (2017) Mitigating operational interactions with short-beaked common dolphin (Delphinus delphis): application of the South Australian Sardine Fishery industry Code of Practice 2016-17. Report to PIRSA Fisheries and Aquaculture. South Australian Research and Development Institute (Aquatic Sciences), Adelaide. SARDI Publication No. F2010/000726-8. SARDI Res Rep Ser 970:44
MacLeod CD (2010) The relationship between body mass and relative investment in testes mass in cetaceans: implications for inferring interspecific variations in the extent of sperm competition. Mar Mamm Sci 26:370–380. https://doi.org/10.1111/j.1748-7692.2009.00348.x
MacLeod CD, MacLeod R (2009) The relationship between body mass and relative investment in testes mass in amniotes and other vertebrates. Oikos 118:903–916. https://doi.org/10.1111/j.1600-0706.2008.17426.x
Matkin CO, Ward Testa J, Ellis GM, Saulitis EL (2014) Life history and population dynamics of southern Alaska resident killer whales (Orcinus orca). Mar Mamm Sci 30:460–479. https://doi.org/10.1111/mms.12049
Meissner AM, Christiansen F, Martinez E, Pawley MD, Orams MB, Stockin KA (2015) Behavioural effects of tourism on oceanic common dolphins, Delphinus sp., in New Zealand: the effects of Markov analysis variations and current tour operator compliance with regulations. PLoS ONE 10:0116962. https://doi.org/10.1371/journal.pone.0116962
Murphy S, Rogan E (2006) External morphology of the short-beaked common dolphin, Delphinus delphis: Growth, allometric relationships and sexual dimorphism. Acta Zool 87:315–329. https://doi.org/10.1111/j.1463-6395.2006.00245.x
Murphy R, Pinkerton M, Richardson K, Bradford-Grieve J, Boyd P (2001) Phytoplankton distributions around New Zealand derived from SeaWiFS remotely-sensed ocean colour data. N Z J Mar Freshwater Res 35:343–362. https://doi.org/10.1080/00288330.2001.9517005
Murphy S, Collet A, Rogan E (2005) Mating strategy in the male common dolphin (Delphinus delphis): what gonadal analysis tells us. J Mammal 86:1247–1258. https://doi.org/10.1644/1545-1542(2005)86[1247:MSITMC]2.0.CO;2
Murphy S, Winship A, Dabin W, Jepson PD, Deaville R, Reid RJ, Spurrier C, Rogan E, López A, González AF (2009) Importance of biological parameters in assessing the status of Delphinus delphis. Mar Ecol Prog Ser 388:273–291. https://doi.org/10.3354/meps08129
Murphy S, Perrott M, McVee J, Read FL, Stockin KA (2014) Deposition of growth layer groups in dentine tissue of captive common dolphins Delphinus delphis. NAMMCO Sci Publ. DOI 10(7557/3):3017
Murphy S, Petitguyot MA, Jepson PD, Deaville R, Lockyer C, Barnett J, Perkins M, Penrose R, Davison NJ, Minto C (2020) Spatio-temporal variability of harbor porpoise life history parameters in the North-East Atlantic. Front Mar Sci. https://doi.org/10.3389/fmars.2020.502352
Murphy S (2004) The biology and ecology of the short-beaked common dolphin Delphinus delphis in the North-east Atlantic. PhD thesis, National University Ireland, University College Cork
Myrick AC Jr, Hohn AA, Sloan PA, Kimura M, Stanley DD (1983) Estimating age of spotted and spinner dolphins (Stenella attenuata and Stenella longirostris) from teeth. NOAA Techinical memorandum NMFS. https://swfsc-publications.fsheries.noaa.gov/publications/TM/SWFSC/NOAA-TM-NMFSSWFC-30.PDF Accessed 10 Oct 2022.
Neimanis AS, Read AJ, Foster RA, Gaskin DE (2000) Seasonal regression in testicular size and histology in harbour porpoises (Phocoena phocoena) from the Bay of Fundy and Gulf of Maine. J Zool 250:221–229. https://doi.org/10.1111/j.1469-7998.2000.tb01072.x
Neimanis A (1996) Ontogeny and seasonal regression of testes of the harbor porpoise. MS thesis, University of Guelph, Canada.
Neumann DR (2001) Activity budget of free-ranging common dolphins (Delphinus delphis) in the northwestern Bay of Plenty, New Zealand. Aquat Mamm 27:121–136
Neumann DR, Orams MB (2006) Impacts of ecotourism on short-beaked common dolphins (Delphinus delphis) in Mercury Bay, New Zealand. Aquat Mamm 32:1–9. https://doi.org/10.1578/AM.32.1.2006.1
Neumann DR, Russell K, Orams MB, Baker CS, Duignan P (2002) Identifying sexually mature, male short-beaked common dolphins (Delphinus delphis) at sea, based on the presence of a postanal hump. Aquat Mamm 28:181–187
Neumann DR, Orams M (2005) Behaviour and ecology of common dolphins (Delphinus delphis) and the impact of tourism in Mercury Bay, North Island, New Zealand. Science & Technical Publishing, Department of Conservation. https://www.doc.govt.nz/globalassets/documents/science-and-technical/sfc254.pdf. Accessed 25 Sep 2022
Ngqulana SG, Hofmeyr GG, Plön S (2017) Sexual dimorphism in long-beaked common dolphins (Delphinus capensis) from KwaZulu-Natal, South Africa. J Mammal 98:1389–1399. https://doi.org/10.1093/jmammal/gyx086
Nicolosi P, Loy A (2019) Geometric morphometric methods as complementary tools to investigate variability in common dolphins (Delphinus sp.) using museum specimens. Aquat Conserv 31:22–35. https://doi.org/10.1002/aqc.3042
O’Hara T, George J, Tarpley R, Burek K, Suydam R (2002) Sexual maturation in male bowhead whales (Balaena mysticetus) of the Bering-Chukchi-Beaufort Seas stock. J Cetacean Res Manag 4:143–148
Olesiuk PF, Bigg MA, Ellis GM (1990) Life history and population dynamics of resident killer whales (Orcinus orca) in the coastal waters of British Columbia and Washington State. Rep Int Whal Comm Spec 12:209–243
Olesiuk PF, Ellis GM, Ford JK (2005) Life history and population dynamics of northern resident killer whales (Orcinus orca) in British Columbia. Canadian Science Advisory Secretariat Ottawa, ON, Canada. https://www.dfo-mpo.gc.ca/csas-sccs/publications/resdocs-docrech/2005/2005_045-eng.htm Accessed 15 Oct 2022
Oliver CW (1973) Gonadal appearence and sexual matuirty in eastern tropical Pacific common dolphins. Ms thesis, University of California, Los Angeles.
Palmer E (2023) Life history of common dolphins in Aotearoa New Zealand waters. PhD thesis, Massey University, Auckland, New Zealand
Palmer EI, Betty EL, Murphy S, Perrott MR, Smith ANH, Stockin KA (2022) Reproductive biology of female common dolphins (Delphinus delphis) in New Zealand waters. Mar Biol 169:158. https://doi.org/10.1007/s00227-022-04139-3
Parra GJ, Bilgmann K, Peters KJ, Moller LM (2021) Abundance and potential biological removal of common dolphins subject to fishery impacts in South Australian waters. Front Mar Sci 8. https://doi.org/10.3389/fmars.2021.617075
Piroddi C, Bearzi G, Gonzalvo J, Christensen V (2011) From common to rare: the case of the Mediterranean common dolphin. Biol Conserv 144:2490–2498. https://doi.org/10.1016/j.biocon.2011.07.003
Plön S, Bernard R (2007) Testis, spermatogenesis, and testicular cycles. In: Jamieson BGM, Miller D (eds) Reproductive biology and phylogeny of cetacea: whales, porpoises and dolphins. CRC Press, Boca Raton
Plön S, Albrecht K, Cliff G, Froneman P (2012) Organ weights of three dolphin species (Sousa chinensis, Tursiops aduncus and Delphinus capensis) from South Africa: implications for ecological adaptation? J Cetacean Res Manage. 12:265–276. https://doi.org/10.47536/jcrm.v12i2.584
Plön S, Heyns-Veale ER, Smale MJ, Froneman PW (2020) Life history parameters and diet of Risso’s dolphins, Grampus griseus, from southeastern South Africa. Mar Mamm Sci 36:786–801. https://doi.org/10.1111/mms.12675
Pomeroy P (2011) Reproductive cycles of marine mammals. Anim Reprod Sci 124:184–193. https://doi.org/10.1016/j.anireprosci.2010.08.021
R Development Core Team (2021) R: a Language and Environment for Statistical Computing. Vienna, Austria. www.R-project.org/
Ramos RMA, Di Beneditto APM, Lima NRW (2000) Growth parameters of Pontoporia blainvillei and Sotalia fluviatilis (Cetacea) in northern Rio de Janeiro, Brazil. Aquat Mamm 26:65–75
Read AJ, Wells RS, Hohn AA, Scott MD (1993) Patterns of growth in wild bottlenose dolphins, Tursiops truncatus. J Zool 231:107–123. https://doi.org/10.1111/j.1469-7998.1993.tb05356.x
Read FL, Santos MB, González ÁF, Murphy S, López A, Rogan E, Peltier H, Ridoux V, Pierce GJ (2019) Strandings, life history and fsheries interactions of the short-beaked common dolphin (Delphinus delphis) in Galicia, NW Spain. https://www.ascobans.org/en/document/strandings-life-history-and-fsheries-interactions-short-beaked-common-dolphin-delphinus. Accessed 25 Jun 2022
Reddy K (1996) The reproductive biology of four species of South African dolphins. Rhodes University, South Africa
Robeck T, Monfort S (2006) Characterization of male killer whale (Orcinus orca) sexual maturation and reproductive seasonality. Theriogenology 66:242–250. https://doi.org/10.1016/j.theriogenology.2005.11.007
Robeck TR, O’Brien JK (2018) Reproductive physiology. In: Perrin WF, Würsig B, Thewissen JGM (eds) Encyclopedia of marine mammals. Academic Press, Cambridge, pp 807–811
Robeck T, Amaral R, da Silva V, Martin A, Montano G, Brown J (2019) Thyroid hormone concentrations associated with age, sex, reproductive status and apparent reproductive failure in the Amazon river dolphin (Inia geoffrensis). Conserv Physiol 7:coz041. https://doi.org/10.1093/conphys/coz041
Rosas FCW, Barreto AS, Monteiro-Filho ELdA (2003) Age and growth of the estuarine dolphin (Sotalia guianensis)(Cetacea, Delphinidae) on the Paraná coast, southern Brazil. Fish Bull 101(2):377–383
Schäfer AT, Kaufmann JD (1999) What happens in freezing bodies?: experimental study of histological tissue change caused by freezing injuries. Forensic Sci Int 102:149–158. https://doi.org/10.1016/S0379-0738(99)00043-2
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:5. https://doi.org/10.1038/nmeth.2089
Sørensen TB, Kinze CC (1994) Reproduction and reproductive seasonality in Danish harbour porpoises, Phocoena phocoena. Ophelia 39:159–176. https://doi.org/10.1080/00785326.1994.10429541
Stamps J, Mangel M, Phillips J (1998) A new look at relationships between size at maturity and asymptotic size. Am Nat 52:470–479. https://doi.org/10.1086/286183
Stockin KA, Law R, Duignan P, Jones G, Porter L, Mirimin L, Meynier L, Orams M (2007) Trace elements, PCBs and organochlorine pesticides in New Zealand common dolphins (Delphinus sp.). Sci Total Environ 387:333–345. https://doi.org/10.1016/j.scitotenv.2007.05.016
Stockin KA, Lusseau D, Binedell V, Wiseman N, Orams MB (2008) Tourism affects the behavioural budget of the common dolphin Delphinus sp. in the Hauraki Gulf. New Zealand Mar Ecol Prog 355:287–295. https://doi.org/10.3354/meps07386
Stockin KA, Duignan PJ, Roe WD, Meynier L, Alley M, Fettermann T (2009) Causes of mortality in stranded common dolphin (Delphinus sp.) from New Zealand waters between 1998 and 2008. Pac Conserv Biol 15:217–227. https://doi.org/10.1071/PC090217
Stockin KA, Yi S, Northcott G, Betty E, Machovsky-Capuska GE, Jones B, Perrott MR, Law R, Rumsby A, Thelen M, Graham L, Palmer E, Tremblay L (2021a) Per-and polyfluoroalkyl substances (PFAS), trace elements and life history parameters of mass-stranded common dolphins (Delphinus delphis) in New Zealand. Mar Pollut Bull 173:112896. https://doi.org/10.1016/j.marpolbul.2021.112896
Stockin KA, Pantos O, Betty EL, Pawley MDM, Doake F, Masterton H, Palmer EI, Perrott MR, Nelms SE, Machovsky-Capuska GE (2021b) Fourier transform infrared (FTIR) analysis identifies microplastics in stranded common dolphins (Delphinus delphis) from New Zealand waters. Mar Pollut Bull. https://doi.org/10.1016/j.marpolbul.2021.113084
Stockin KA, Amiot C, Meynier L, Purvin C, Machovsky-Capuska GE (2022) Understanding common dolphin and Australasian gannet feeding associations from nutritional and ethological perspectives. ICES J Mar Sci 79:7. https://doi.org/10.1093/icesjms/fsac133
Stockin KA, Machovsky-Capuska GE, Palmer EI, Amiot C (2023) Multidimensional nutritional and trace metal niche space differs between sexually immature and mature (Delphinus delphis). Environ Pollut. https://doi.org/10.1016/j.envpol.2023.121935
Thompson FN, Abraham ER, Berkenbusch K (2013) Common dolphin (Delphinus delphis) bycatch in New Zealand commercial trawl fsheries. PLoS ONE. https://doi.org/10.1371/journal.pone.0064438
Van Waerebeek K, Read AJ (1994) Reproduction of dusky dolphins, Lagenorhynchus obscurus, from coastal Peru. J Mammal 75:1054–1062. https://doi.org/10.2307/1382489
Vehtari A, Gelman A, Gabry J (2017) Practical Bayesian model evaluation using leave-one-out cross-validation and WAIC. Stat Comput 27:1413–1432. https://doi.org/10.1007/s11222-016-9696-4
Vella A, Murphy S, Giménez J, de Stephanis R, Mussi B, Vella JG, Larbi Doukara K, Pace DS (2021) The conservation of the endangered Mediterranean common dolphin (Delphinus delphis): Current knowledge and research priorities. Aquat Conserv 31:110–136. https://doi.org/10.1002/aqc.3538
Vu ET, Clark C, Catelani K, Kellar NM, Calambokidis J (2015) Seasonal blubber testosterone concentrations of male humpback whales (Megaptera novaeangliae). Mar Mamm Sci. https://doi.org/10.1111/mms.12191
Wells RS (2014) Social structure and life history of bottlenose dolphins near Sarasota Bay, Florida insights from four decades and five generations. In: Yamagiwa J, Karczmarski L (eds) Primates and cetaceans field research and conservation of complex mammalian societies. Springer, Berlin
Westgate AJ (2005) Population structure and life history of short-beak common dolphins (Delphinus delphis) in the North Atlantic. Duke University, North Carolina
Westgate AJ, Read AJ (2007) Reproduction in short-beaked common dolphins (Delphinus delphis) from the western North Atlantic. Mar Biol 150:1011. https://doi.org/10.1007/s00227-006-0394-1
Zeiss C (2013) Zeiss Software. https://www.zeiss.com/microscopy/en/home.html
Acknowledgements
The authors particularly thank Mana Moana and Department of Conservation Te Papa Atawhai, especially Hannah Hendriks, Laura Boren, and Anton van Helden for facilitating access to carcasses. Finally, the authors thank Katharina Peters for GIS assistance and Padraig Duignan, Wendi Roe, Odette Howarth, Laureline Meynier, and all Cetacean Ecology Research Group staff and students who over decades, assisted with necropsies.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. EP is supported by a Massey University Doctoral Scholarship, a Wildbase Research Grant and the Royal Society Te Apārangi Hutton Fund. KAS is supported by a Royal Society Te Apārangi Rutherford Discovery Fellowship (2019–2024). This research was partially funded by a Massey University Research Fund (MURF) grant awarded to KAS and ELB (RM22283), and a Marie Curie International Outgoing Fellowship awarded to SM (PIOF-GA-2010–276145).
Author information
Authors and Affiliations
Contributions
Conception and study design by EIP, ELB, and KAS. All authors contributed to the methodology. Investigation carried out by EIP, ELB, SM, MRP, and KAS. EIP and ANHS carried out statistical analyses. Original draft was written by EIP. All authors contributed to reviewing and editing of the final manuscript. KAS, ELB, and MRP provided supervision of EIP. Projection administration led by KAS. Funding acquisition led by KAS, SM, ELB, and EIP.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Compliance with ethical standards and ethics approval
All sampling was undertaken under the research permit 39239-MAR, RNW/22/2003/182, and RNW/HO/2008/03 research permits issued by the Department of Conservation Te Papa Atawhai. As samples were recovered only from carcasses and no live animals were sampled, we did not require animal ethics approval for this research.
Additional information
Responsible Editor: U. Siebert .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Palmer, E.I., Betty, E.L., Murphy, S. et al. Reproductive biology of male common dolphins (Delphinus delphis) in New Zealand waters. Mar Biol 170, 153 (2023). https://doi.org/10.1007/s00227-023-04266-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-023-04266-5