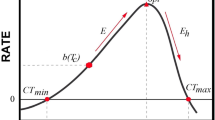
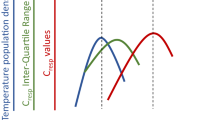
Abstract
As climate change continues, anticipating species’ responses to rising temperatures requires an understanding of the drivers of thermal sensitivity, which itself may vary over space and time. We measured metabolic rates of three representative marine invertebrate species (hermit crabs Pagurus hirsutiusculus, periwinkle snails Littorina sitkana, and mussels Mytilus trossulus) and evaluated the relationship between thermal sensitivity (Q10) and thermal history. We tested the hypothesis that thermal history drives thermal sensitivity and quantified how this relationship differs over time (short-term to seasonal time scales) and between species. Organisms were collected from tide pools in Sitka, Alaska where we also recorded temperatures to characterize thermal history prior to metabolic rate assays. Using respirometry, we estimated mass-specific oxygen consumption (MO2) at ambient and increased temperatures for one individual per species per tide pool across three seasons. We evaluated relationships between thermal sensitivity and pool temperatures for time periods ranging from 1 day to 1 month prior to collection. For all species, thermal sensitivity was related to thermal history for the shorter time periods (1 day to 1 week). However, the direction of the relationships and most important thermal parameters (i.e., maximum, mean, or range) differed between species and seasons. We found that on average, P. hirsutiusculus and L. sitkana were more thermally sensitive than M. trossulus. These findings show that variability in thermal history over small spatial scales influences individuals’ metabolic response to warming and may be indicative of these species’ ability to acclimate to future climate change.





Similar content being viewed by others
Data availability
The datasets and R scripts that support this study are available during peer review on the Dryad Local-scale thermal history influences metabolic response of marine invertebrates Repository, https://datadryad.org/stash/share/jwPvQUfa9zqlikQv7nXU4nuFehaRsg9fk9HSIvN7pNg and on zenodo/github: https://doi.org/10.5281/zenodo.6464075, https://github.com/racine-rangel/RangelandSorte_ThermalHistory.
References
Abraham JP, Baringer M, Bindoff N, Boyer T, Cheng L, Church J, Conroy J, Domingues C, Fasullo J, Gilson J et al (2013) A review of global ocean temperature observations: Implications for ocean heat content estimates and climate change. Rev Geophys 51:450–483
Allen M, Antwi-Agyei P, Aragon-Durand F, Babiker M, Bertoldi P, Bind M, Brown S, Buckeridge M, Camilloni I, Cartwright A, others (2019) Technical Summary: Global warming of 1.5° C. An IPCC Special Report on the impacts of global warming of 1.5° C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change, sustainable development, and efforts to eradicate poverty.
Anestis A, Lazou A, Pörtner HO, Michaelidis B (2007) Behavioral, metabolic, and molecular stress responses of marine bivalve Mytilus galloprovincialis during long-term acclimation at increasing ambient temperature. Am J Physiol-Regulat Integr Comp Physiol. https://doi.org/10.1152/ajpregu.00124.2007
Angilletta MJ Jr (2009) Thermal adaptation: a theoretical and empirical synthesis. Oxford University Press, Oxford
Angilletta M, Sears M (2000) The metabolic cost of reproduction in an oviparous lizard. Funct Ecol 14:39–45
Angilletta MJ Jr (2001) Variation in metabolic rate between populations of a geographically widespread lizard. Physiol Biochem Zool 74:11–21
Auer SK, Dick CA, Metcalfe NB, Reznick DN (2018) Metabolic rate evolves rapidly and in parallel with the pace of life history. Nat Commun 9:1–6
Ayala-Díaz M, Richardson JML, Anholt BR (2017) Local site differences in survival and parasitism of periwinkles ( Littorina sitkana Philippi, 1846). Ecol Evol 7:1021–1029. https://doi.org/10.1002/ece3.2708
Bartheld JL, Gaitán-Espitia JD, Artacho P, Salgado-Luarte C, Gianoli E, Nespolo RF (2015) Energy expenditure and body size are targets of natural selection across a wide geographic range, in a terrestrial invertebrate. Funct Ecol 29:1463–1474
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. J Stat Soft. https://doi.org/10.18637/jss.v067.i01
Bates TW, Hicks DW (2005) Locomotory behavior and habitat selection in littoral gastropods on Caribbean limestone shores. J Shellfish Res 24:75–84
Benito D, Ahvo A, Nuutinen J, Bilbao D, Saenz J, Etxebarria N, Lekube X, Izagirre U, Lehtonen KK, Marigómez I et al (2019) Influence of season-depending ecological variables on biomarker baseline levels in mussels (Mytilus trossulus) from two Baltic Sea subregions. Sci Total Environ 689:1087–1103
Bennett S, Duarte CM, Marbà N, Wernberg T (2019) Integrating within-species variation in thermal physiology into climate change ecology. Philos Trans R Soc B 374:20180550
Bertness MD (1981) Conflicting advantages in resource utilization: the hermit crab housing dilemma. Am Nat 118:432–437
Birk M (2020) respirometry: Tools for conducting and analyzing respirometry experiments, R package, version 0.7.0.
Bond NA, Cronin MF, Freeland H, Mantua N (2015) Causes and impacts of the 2014 warm anomaly in the NE Pacific. Geophys Res Lett 42:3414–3420
Braswell B, Schimel DS, Linder E, Moore B (1997) The response of global terrestrial ecosystems to interannual temperature variability. Science 278:870–873
Buckland-Nicks J, Chia F-S, Behrens S (1973) Oviposition and development of two intertidal snails, Littorina sitkana and Littorina scutulata. Can J Zool 51:359–365
Calosi P, Bilton DT, Spicer JI (2008) Thermal tolerance, acclimatory capacity and vulnerability to global climate change. Biol Let 4:99–102
Careau V, Gifford ME, Biro PA (2014) Individual (co) variation in thermal reaction norms of standard and maximal metabolic rates in wild-caught slimy salamanders. Funct Ecol 28:1175–1186
Cheng BS, Komoroske LM, Grosholz ED (2017) Trophic sensitivity of invasive predator and native prey interactions: integrating environmental context and climate change. Funct Ecol 31:642–652
Clarke A (1993) Seasonal acclimatization and latitudinal compensation in metabolism: do they exist? Funct Ecol 7:139–149
Connor KM, Gracey AY (2012) High-resolution analysis of metabolic cycles in the intertidal mussel Mytilus californianus. Am J Physiol-Regulat, Integr Comp Physiol 302:R103–R111
Crickenberger S, Hui T, Yuan FL, Bonebrake T, Williams G (2020) Preferred temperature of intertidal ectotherms: broad patterns and methodological approaches. J Therm Biol 87:102468
da Silva CRB, Riginos C, Wilson RS (2019) An intertidal fish shows thermal acclimation despite living in a rapidly fluctuating environment. J Comp Physiol B 189:385–398
Deutsch C, Ferrel A, Seibel B, Pörtner H-O, Huey RB (2015) Climate change tightens a metabolic constraint on marine habitats. Science 348:1132–1135
Dillon ME, Woods HA, Wang G, Fey SB, Vasseur DA, Telemeco RS, Marshall K, Pincebourde S (2016) Life in the frequency domain: the biological impacts of changes in climate variability at multiple time scales. Integr Comp Biol 56:14–30
Drake MJ, Miller NA, Todgham AE (2017) The role of stochastic thermal environments in modulating the thermal physiology of an intertidal limpet, Lottia digitalis. J Exp Biol 220:3072–3083
Gaitán-Espitia JD, Bacigalupe LD, Opitz T, Lagos NA, Osores S, Lardies MA (2017) Exploring physiological plasticity and local thermal adaptation in an intertidal crab along a latitudinal cline. J Therm Biol 68:14–20
Gazeau F, Alliouane S, Bock C, Bramanti L, López Correa M, Gentile M, Hirse T, Pörtner H-O, Ziveri P (2014) Impact of ocean acidification and warming on the Mediterranean mussel (Mytilus galloprovincialis). Front Mar Sci 1:62
Giomi F, Mandaglio C, Ganmanee M, Han G-D, Dong Y-W, Williams GA, Sarà G (2016) The importance of thermal history: costs and benefits of heat exposure in a tropical, rocky shore oyster. J Exp Biol 219:686–694
Glazier DS (2015) Is metabolic rate a universal ‘pacemaker’for biological processes? Biol Rev 90:377–407
Guppy M, Withers P (1999) Metabolic depression in animals: physiological perspectives and biochemical generalizations. Biol Rev 74:1–40
Halsey LG, Matthews P, Rezende E, Chauvaud L, Robson AA (2015) The interactions between temperature and activity levels in driving metabolic rate: theory, with empirical validation from contrasting ectotherms. Oecologia 177:1117–1129
Hayford HA, O’Donnell MJ, Carrington E (2018) Radio tracking detects behavioral thermoregulation at a snail’s pace. J Exp Mar Biol Ecol 499:17–25
Healy TM, Schulte PM (2012) Thermal acclimation is not necessary to maintain a wide thermal breadth of aerobic scope in the common killifish (Fundulus heteroclitus). Physiol Biochem Zool 85:107–119
Helmuth B, Broitman BR, Blanchette CA, Gilman S, Halpin P, Harley CD, O’Donnell MJ, Hofmann GE, Menge B, Strickland D (2006) Mosaic patterns of thermal stress in the rocky intertidal zone: implications for climate change. Ecol Monogr 76:461–479
Hiebert TC, Butler BA, Shanks AL (2015) Oregon estuarine invertebrates: Rudys’ illustrated guide to common species.
Hochachka PW, Somero GN (2002) Biochemical adaptation: mechanism and process in physiological evolution. Oxford University Press
Hoegh-Guldberg O, Bruno JF (2010) The Impact of climate change on the world’s marine ecosystems. Science 328:1523–1528. https://doi.org/10.1126/science.1189930
Hofmann GE, Todgham AE (2010) Living in the now: physiological mechanisms to tolerate a rapidly changing environment. Annu Rev Physiol 72:127–145
Huey RB, Kearney MR, Krockenberger A, Holtum JA, Jess M, Williams SE (2012) Predicting organismal vulnerability to climate warming: roles of behaviour, physiology and adaptation. Philosophical Transact Roy Soc B: Biol Sci 367:1665–1679
Irlich UM, Terblanche JS, Blackburn TM, Chown SL (2009) Insect rate-temperature relationships: environmental variation and the metabolic theory of ecology. Am Nat 174:819–835
Iwabuchi BL, Gosselin LA (2020) Implications of acute temperature and salinity tolerance thresholds for the persistence of intertidal invertebrate populations experiencing climate change. Ecol Evol 10:7739–7754
Juárez OE, Lafarga-De la Cruz F, Leyva-Valencia I, López-Landavery E, García-Esquivel Z, Díaz F, Re-Araujo D, Vadopalas B, Galindo-Sánchez CE (2018) Transcriptomic and metabolic response to chronic and acute thermal exposure of juvenile geoduck clams Panopea globosa. Mar Genomics 42:1–13
Kearney M, Shine R, Porter WP (2009) The potential for behavioral thermoregulation to buffer “cold-blooded” animals against climate warming. Proc Natl Acad Sci 106:3835–3840
Kern P, Cramp RL, Franklin CE (2015) Physiological responses of ectotherms to daily temperature variation. J Exp Biol 218:3068–3076
Killen SS, Christensen EA, Cortese D, Zavorka L, Norin T, Cotgrove L, Crespel A, Munson A, Nati JJ, Papatheodoulou M et al (2021) Guidelines for reporting methods to estimate metabolic rates by aquatic intermittent-flow respirometry. J Exp Biol. https://doi.org/10.1242/jeb.242522
Lê S, Josse J, Husson F et al (2008) FactoMineR: an R package for multivariate analysis. J Stat Softw 25:1–18
Lesser MP (2016) Climate change stressors cause metabolic depression in the blue mussel, Mytilus edulis, from the Gulf of Maine. Limnol Oceanogr 61:1705–1717
Marshall DJ, Dong Y, -w., McQuaid CD, Williams GA, (2011) Thermal adaptation in the intertidal snail Echinolittorina malaccana contradicts current theory by revealing the crucial roles of resting metabolism. J Exp Biol 214:3649–3657. https://doi.org/10.1242/jeb.059899
Marshall DJ, Rezende EL, Baharuddin N, Choi F, Helmuth B (2015) Thermal tolerance and climate warming sensitivity in tropical snails. Ecol Evol 5:5905–5919
May C, Hillerbrand N, Thompson LM, Faske TM, Martinez E, Parry D, Agosta SJ, Grayson KL (2018) Geographic variation in larval metabolic rate between northern and southern populations of the invasive gypsy moth. J Insect Sci 18:5
McGaw I (2003) Behavioral thermoregulation in Hemigrapsus nudus, the amphibious purple shore crab. Biol Bull 204:38–49
Menge BA, Sutherland JP (1987) Community regulation: variation in disturbance, competition, and predation in relation to environmental stress and recruitment. Am Nat 130:730–757
Menge BA, Olson AM, Dahlhoff EP (2002) Environmental stress, bottom-up effects, and community dynamics: integrating molecular-physiological and ecological approaches. Integr Comp Biol 42:892–908
Miller LP, Denny MW (2011) Importance of behavior and morphological traits for controlling body temperature in littorinid snails. Biol Bull 220:209–223
Miller LP, Dowd WW (2017) Multimodal in situ datalogging quantifies inter-individual variation in thermal experience and persistent origin effects on gaping behavior among intertidal mussels (Mytilus californianus). J Exp Biol 220:4305–4319
Miller LP, Allen BJ, King FA, Chilin DR, Reynoso VM, Denny MW (2015) Warm microhabitats drive both increased respiration and growth rates of intertidal consumers. Mar Ecol Prog Ser 522:127–143
Moyen NE, Somero GN, Denny MW (2020) Mussel acclimatization to high, variable temperatures is lost slowly upon transfer to benign conditions. J Exp Biol. https://doi.org/10.1242/jeb.222893
Nelson D, Benstead JP, Huryn AD, Cross WF, Hood JM, Johnson PW, Junker JR, Gíslason GM, Ólafsson JS (2017) Experimental whole-stream warming alters community size structure. Glob Change Biol 23:2618–2628
Osovitz CJ, Hofmann GE (2005) Thermal history-dependent expression of the hsp70 gene in purple sea urchins: biogeographic patterns and the effect of temperature acclimation. J Exp Mar Biol Ecol 327:134–143
Pack KE, Rius M, Mieszkowska N (2021) Long-term environmental tolerance of the non-indigenous Pacific oyster to expected contemporary climate change conditions. Mar Environ Res 164:105226
Paganini AW, Miller NA, Stillman JH (2014) Temperature and acidification variability reduce physiological performance in the intertidal zone porcelain crab Petrolisthes cinctipes. J Exp Biol 217:3974–3980
Pandori LL, Sorte CJB (2019) The weakest link: sensitivity to climate extremes across life stages of marine invertebrates. Oikos 128:621–629
Pandori LL, Sorte CJB (2021) Spatial and temporal scales of exposure and sensitivity drive mortality risk patterns across life stages. Ecosphere 12:e03552
Pershing AJ, Record NR, Franklin BS, Kennedy BT, McClenachan L, Mills KE, Scott JD, Thomas AC, Wolff NH (2019) Challenges to natural and human communities from surprising ocean temperatures. Proc Natl Acad Sci 116:18378–18383
Petes LE, Mouchka ME, Milston-Clements RH, Momoda TS, Menge BA (2008) Effects of environmental stress on intertidal mussels and their sea star predators. Oecologia 156:671–680
Pettersen AK, Marshall DJ, White CR (2018) Understanding variation in metabolic rate. J Exp Biol. https://doi.org/10.1242/jeb.166876
Pörtner HO, Berdal B, Blust R, Brix O, Colosimo A, De Wachter B, Giuliani A, Johansen T, Fischer T, Knust R, Lannig G, Naevdal G, Nedenes A, Nyhammer G, Sartoris FJ, Serendero I, Sirabella P, Thorkildsen S, Zakhartsev M (2001) Climate induced temperature effects on growth performance, fecundity and recruitment in marine fish: developing a hypothesis for cause and effect relationships in Atlantic cod (Gadus morhua) and common eelpout (Zoarces viviparus). Cont Shelf Res 21:1975–1997. https://doi.org/10.1016/S0278-4343(01)00038-3
Rangel RE, Johnson DW (2019) Variation in metabolic rate and a test of differential sensitivity to temperature in populations of woolly sculpin (Clinocottus analis). J Exp Mar Biol Ecol 511:68–74. https://doi.org/10.1016/j.jembe.2018.11.007
Richter-Boix A, Katzenberger M, Duarte H, Quintela M, Tejedo M, Laurila A (2015) Local divergence of thermal reaction norms among amphibian populations is affected by pond temperature variation. Evolution 69:2210–2226
Royer TC (2005) Hydrographic responses at a coastal site in the northern Gulf of Alaska to seasonal and interannual forcing. Deep Sea Res Part II 52:267–288
Seebacher F, White CR, Franklin CE (2015) Physiological plasticity increases resilience of ectothermic animals to climate change. Nat Clim Chang 5:61–66
Seed R, Suchanek TH (1992) Population and community ecology of Mytilus. The Mussel Mytilus: Ecol Physiol, Gene Cult 25:87–170
Shah AA, Gill BA, Encalada AC, Flecker AS, Funk WC, Guayasamin JM, Kondratieff BC, Poff NL, Thomas SA, Zamudio KR et al (2017) Climate variability predicts thermal limits of aquatic insects across elevation and latitude. Funct Ecol 31:2118–2127
Somero G (2010) The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine ‘winners’ and ‘losers.’ J Exp Biol 213:912–920
Somero GN (2002) Thermal physiology and vertical zonation of intertidal animals: optima, limits, and costs of living. Integr Comp Biol 42:780–789
Sorte CJB, Bracken MES (2015) Warming and elevated CO2 interact to drive rapid shifts in marine community production. PLoS ONE 10:e0145191
Sorte CJB, Jones SJ, Miller LP (2011) Geographic variation in temperature tolerance as an indicator of potential population responses to climate change. J Exp Mar Biol Ecol 400:209–217
Steffensen J, Bushnell P, Schurmann H (1994) Oxygen consumption in four species of teleosts from Greenland: no evidence of metabolic cold adaptation. Polar Biol 14:49–54
Stillman JH (2003) Acclimation capacity underlies susceptibility to climate change. Science 301:65–65
Taylor PR (1982) Environmental resistance and the ecology of coexisting hermit crabs: thermal tolerance. J Exp Mar Biol Ecol 57:229–236
Team RC, others (2013) R: A language and environment for statistical computing.
Todgham AE, Iwama GK, Schulte PM (2006) Effects of the natural tidal cycle and artificial temperature cycling on Hsp levels in the tidepool sculpin Oligocottus maculosus. Physiol Biochem Zool 79:1033–1045
Toledo LF, Brito SP, Milsom WK, Abe AS, Andrade DV (2008) Effects of season, temperature, and body mass on the standard metabolic rate of tegu lizards (Tupinambis merianae). Physiol Biochem Zool 81:158–164
van Denderen D, Gislason H, van den Heuvel J, Andersen KH (2020) Global analysis of fish growth rates shows weaker responses to temperature than metabolic predictions. Glob Ecol Biogeogr 29:2203–2213
Vorhees AS, Gray EM, Bradley TJ (2013) Thermal resistance and performance correlate with climate in populations of a widespread mosquito. Physiol Biochem Zool 86:73–81
Xiong Q, Royer TC (1984) Coastal temperature and salinity in the northern Gulf of Alaska, 1970–1983. J Geophysical Resh 89:8061–8068
Yang Q, Cokelet ED, Stabeno PJ, Li L, Hollowed AB, Palsson WA, Bond NA, Barbeaux SJ (2019) How “The Blob” affected groundfish distributions in the Gulf of Alaska. Fish Oceanogr 28:434–453
Zippay ML, Helmuth B (2012) Effects of temperature change on mussel, Mytilus. Integr Zool 7:312–327
Acknowledgements
We would like to thank Sarah Mastroni and Julieta Gomez for their help with collection of specimens, Matt Bracken and Grant Gallaher for assistance with fieldwork, Lauren Pandori, Piper Wallingford, Connor White, Luke Miller, and Celia Symons for data analysis assistance, and Umi Hoshijima and Genevieve Bernatchez for equipment assistance. We would also like to thank the Sitka Sound Science Center and University of Alaska Southeast Sitka Campus for granting us access to use their facilities. We are also grateful to Sorte Lab members, Matt Bracken, and Donovan German for their incredibly helpful comments on earlier versions of the manuscript.
Funding
This work was supported by the National Science Foundation [OCE-1756173 to C.S. and M. Bracken] and support to R.R. from the International Women’s Fishing Association Scholarship and the National Academy of Sciences Ford Foundation.
Author information
Authors and Affiliations
Contributions
RR designed the study, performed the experiments, analyzed the data and created all figures, and drafted the manuscript. CS advised on experimental design, collected the temperature data, assisted in data interpretation, and revised the manuscript. All authors edited and revised the manuscript and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interests.
Ethical approval
All applicable national and institutional guidelines for sampling, care, and experimental use of organisms for the study have been followed. This work was approved and conducted under Alaska Department of Fish and Game Aquatic Resource Permits for Scientific/Collection purposes (ID numbers CF-18–071 and CF-20–050).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rangel, R.E., Sorte, C.J.B. Local-scale thermal history influences metabolic response of marine invertebrates to warming. Mar Biol 169, 126 (2022). https://doi.org/10.1007/s00227-022-04110-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-022-04110-2