Abstract
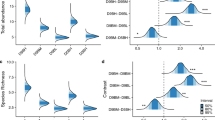
Harpacticoid copepods of the Chernaya Bay (White Sea) intertidal zone were collected in 45 surveys carried out from spring to autumn over a 25-year period (1996–2020) at three sites that differed in sediment properties. There were no significant long-term trends or seasonal cycles in total abundance. Regarding the species composition, the differences between sites were the most important source of variability over the whole period while the fine-scale (within-habitat) variability was low. Epibenthic species prevailed in fine silty sand, both burrowing and epibenthic species prevailed in medium sand, and interstitial and burrowing species prevailed in coarse sand. A comparison of the data on harpacticoid assemblages from a number of geographically remote loci corroborated the generality of this pattern. In a temporal dimension, the structure of each community was stable until the early 2000s, when the proportion of epibenthic, burrowing and interstitial species changed following changes in sediment properties (increasing siltation at sandy sites and decreasing siltation at the silty site). At each site, there was an increasing long-term trend in diversity (both in total richness and in expected species number). This increase was particularly apparent at sandy sites because of the appearance of epibenthic species. We suggest that sediment composition is the key factor determining the composition of harpacticoid assemblages in space and time. The “ecomorphological profile”, i.e., the proportion of species with different lifestyle and morphological traits, is a useful and informative indicator for describing and typifying these assemblages.








Similar content being viewed by others
Data availability
The datasets generated and analyzed during this study, if not included its supplementary information files, are available from the corresponding author on reasonable request.
References
Anderson M, Gorley R, Clarke K (2008) PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth
Ansari Z, Parulekar A (1993) Environmental stability and seasonality of a harpacticoid copepod community. Mar Biol 115:279–286. https://doi.org/10.1007/BF00346345
Azovsky A (2000) Concept of scale in marine ecology: linking the words or the worlds? Web Ecol 1:28–34. https://doi.org/10.5194/we-1-28-2000
Azovsky A, Chertoprood E, Saburova M, Polikarpov I (2004) Spatio-temporal variability of micro- and meiobenthic communities in the White Sea intertidal sandflat. Estuar Coast Shelf Sci 60:663–671. https://doi.org/10.1016/j.ecss.2004.03.005
Azovsky A, Garlitska L, Chertoprud E (2016) Multi-scale taxonomic diversity of marine harpacticoids: does it differ at high and low latitudes? Mar Biol 163(5):94–105. https://doi.org/10.1007/s00227-016-2876-0
Azovsky A, Chertoprud E, Saburova M (2022) Small-scale spatiotemporal variability and distance–decay relationships in intertidal micro- and meiobenthic assemblages. Mar Ecol (in press).
Bell S, Walters K, Hall M (1987) Habitat utilization by harpacticoid copepods: a morphometric approach. Mar Ecol Progr Ser 35:59–64. https://www.jstor.org/stable/24825009
Bodin P, Leguellec C (1992) Meiobenthos of the bay of Saint-Brieuc (North Brittany, France). 2. Harpacticoid copepod diversity and species assemblages. Oceanol Acta 15:673–686
Burkovsky I, Mazei Y (2010) Long-term dynamics of marine interstitial ciliate community. Protistology 6(3):147–172
Burkovsky I, Mazei Y (2017) 21-year dynamics of marine benthic ciliate community in the White Sea intertidal flat: gradual or discrete? Russ J Ecosyst Ecol 2(2):1–15. https://doi.org/10.21685/2500-0578-2017-2-1
Castel J, Lasserre P (1979) Opportunistic copepods in temperate lagoons of Arcachon Bay: differential distribution and temporal heterogeneity. Estuar Coast Mar Sci 9:357–368. https://doi.org/10.1016/0302-3524(79)90011-2
Chertoprud E, Azovsky A (2006) Seasonal dynamics of the populations of intertidal harpacticoids (Harpacticoida: Copepoda) in the White Sea. Oceanology 46:71–80. https://doi.org/10.1134/S0001437006010097
Chertoprud E, Chertoprud M, Kondar’ D, Kornev P, Udalov A (2006) Harpacticoida taxocen diversity in the silty-sandy littoral zone of Kandalaksha Bay of the White Sea. Oceanology 46:492–500. https://doi.org/10.1134/S0001437006040060
Chertoprud E, Chertoprud M, Azovsky A, Garlitska L, Kondar’ D (2007) Spatial variability of the structure of the Harpacticoida (Copepoda) crustacean assemblages in intertidal and shallow-water zones of European seas. Oceanology 47:51–59. https://doi.org/10.1134/S0001437007010080
Clarke K, Somerfield P, Gorley R (2008) Testing of null hypotheses in exploratory community analyses: similarity profiles and biota-environment linkage. J Exp Mar Biol Ecol 366:56–69. https://doi.org/10.1016/j.jembe.2008.07.009
Colwell K, Chao A, Gotelli N, Lin S, Mao C, Chazdon R, Longino J (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. J Plant Ecol 5:3–21. https://doi.org/10.1093/jpe/rtr044
Coull B (1970) Shallow water meiobenthos of the Bermuda platform. Oecologia (berl.) 4:325–357. https://doi.org/10.1007/BF00393393
Coull B (1985a) Long-term variability of estuarine meiobenthos: an 11 year study. Mar Ecol Prog Ser 24:205–218
Coull B (1985b) The use of long-term biological data to generate testable hypotheses. Estuaries 8(2A):84–92. https://doi.org/10.2307/1351859
Coull B (1986) Long-term variability of meiobenthos: value, synopsis, hypothesis generation and predictive modelling. Hydrobiologia 142:271–279. https://doi.org/10.1007/BF00026764
Coull B, Dudley B (1985) Dynamics of meiobenthic copepod populations: a long-term study (1973–1983). Mar Ecol Progr Ser 24:219–229
Dornelas M, Gotelli N, McGill B, Shimadzu H, Moyes F, Sievers C, Magurran A (2014) Assemblage time series reveal biodiversity change but not systematic loss. Science 344(6181):296–299. https://doi.org/10.1126/science.1248484
George KH, Rose A (2004) First record of a monospecific harpacticoid fauna on a sandy beach. Meiofauna Marina 13:87–94
Giere O (2009) Meiobenthology. The microscopic motile fauna of aquatic sediments, 2nd edn. Springer, Berlin
Heip C (1979) Density and diversity of meiobenthic copepods: the oscillatory behaviour of population and community parameters. In: Naylor E, Hartnoll R (eds) Cyclic phenomena in marine plants and animals. Pergamon Press, Oxford, pp 43–47
Heip C (1980) Meiobenthos as a tool in the assessment of marine environmental quality. Rapp P-V Réun Cons Int Explor Mer 179:182–187
Herman P, Heip C (1983) Long-term dynamics of meiobenthic populations. Oceanol Acta Special Issue, pp 109–112
Herman P, Heip C (1987) The predictability of biological populations and communities: an example from the meiobenthos. Hydrobiologia 142:281–290. https://doi.org/10.1007/978-94-009-4049-9_24
Hewitt J, Thrush S, Dayton P, Bonsdorff E (2007) The effect of spatial and temporal heterogeneity on the design and analysis of empirical studies of scale-dependent systems. Am Nat 169(3):398–408. https://doi.org/10.1086/510925
Hicks G, Coull B (1983) The ecology of marine meiobenthic harpacticoid copepods. Oceanogr Mar Biol Annu Rev 21:67–175
Hockin D (1982) The effects of sediment particle diameter upon meiobenthic copepod community of an intertidal beach: a field and a laboratory experiment. J Anim Ecol 51:555–572. https://doi.org/10.2307/3984
Huys R, Herman R, Heip C (1986) Seasonal fluctuations in vertical distribution and breeding activity of a subtidal harpacticoid community in the Southern Bight, North Sea. Neth J Sea Res 20(4):375–383. https://doi.org/10.1016/0077-7579(86)90004-9
Kern J (1990) Active and passive aspects of meiobenthic copepod dispersal at two sites near Mustang Island, Texas. Mar Ecol Prog Ser 60:211–223. https://doi.org/10.3354/meps060211
McIntyre A, Murison D (1973) The meiofauna of a flatfish nursery ground. J Mar Biol Assoc UK 53:93–118. https://doi.org/10.1017/S0025315400056666
Mielke W (1984) Interstitielle Fauna von Galapagos. XXXI. Paramesochridae (Harpacticoida). Microfauna Marina 1:63–147
Moore C (1979a) Analysis of the associations of meiobenthic Copepoda of the Irish Sea. J Mar Biol Assoc UK 59:831–849. https://doi.org/10.1017/S0025315400036870
Moore C (1979b) The distribution and ecology of psammolittoral meiofauna around the Isle of Man. Cah Biol Mar 20:383–415
Rosli N, Leduc D, Rowden A, Probert P (2018) Review of recent trends in ecological studies of deep-sea meiofauna, with focus on patterns and processes at small to regional spatial scales. Mar Biodivers 48(1):13–34. https://doi.org/10.1007/s12526-017-0801-5
Rybnikov P, Kondar’ D, Azovsky A (2003) Properties of the White Sea littoral sediments and their influence on the fauna and distribution of Harpacticoida. Oceanology 43:91–102
Smirnova E, Azovsky A (2020) Modelling of spatially distributed predator–prey system with periodically migrating predator (case study of the White Sea intertidal harpacticoids and benthic microalgae). Oceanology 60:89–97. https://doi.org/10.1134/S000143702001021X
Snell Taylor S, Evans B, White E, Hurlbert A (2018) The prevalence and impact of transient species in ecological communities. Ecology 99:1825–1835. https://doi.org/10.1002/ecy.2398
Soltwedel T, Grzelak K, Hasemann C (2020) Spatial and temporal variation in deep-sea meiofauna at the LTER Observatory HAUSGARTEN in the Fram Strait (Arctic Ocean). Diversity 7:279. https://doi.org/10.3390/d12070279
Sun B, Fleeger J, Carney R (1993) Sediment microtopography and the small-scale spatial distribution of meiofauna. J Exp Mar Biol Ecol 167(1):73–90. https://doi.org/10.1016/0022-0981(93)90185-Q
Walters K, Bell S (1994) Significance of copepod emergence to benthic, pelagic and phytal linkages in a subtidal seagrass bed. Mar Ecol Prog Ser 108:237–249
Webb D, Parsons T (1992) Winter-spring recruitment patterns of epiphytic harpacticoid copepods in a temperate-zone seagrass bed. Mar Ecol Progr Ser 82:151–162
Willems K, Sharma Y, Heip C, Sandee A (1984) Long-term evolution of the meiofauna at a sandy station in lake Grevelingen, the Netherlands. Neth J Sea Res 18(3–4):418–433. https://doi.org/10.1016/0077-7579(84)90013-9
Williams R (1972) The abundance and biomass of the interstitial fauna of a graded series of shell gravels in relation to available space. J Anim Ecol 41:623–646. https://doi.org/10.2307/3199
Zeppilli D, Sarrazin J, Leduc D et al (2015) Is the meiofauna a good indicator for climate change and anthropogenic impacts? Mar Biodivers 45(3):505–535. https://doi.org/10.1007/s12526-015-0359-z
Acknowledgements
This research was performed according to the Development program of the Interdisciplinary Scientific and Educational School of M.V. Lomonosov Moscow State University “The future of the planet and global environmental change”.
Funding
This work was supported by the Russian Science Foundation (Grant No. 22-24-00007).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by EC and LG, the analysis was performed by AA. The first draft of the manuscript was written by AA and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethics approval
The manuscript had not been submitted to any other journal for simultaneous consideration. The submitted work is original and was not been published elsewhere in any form or language (partially or in full).
Additional information
Responsible Editor: P. Kraufvelin.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Azovsky, A.I., Chertoprud, E.S. & Garlitska, L.A. Environmental stability and long-term variability of harpacticoid copepod assemblages. Mar Biol 169, 67 (2022). https://doi.org/10.1007/s00227-022-04059-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-022-04059-2