Abstract
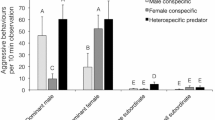
Intraspecific aggression and competition can govern access to food and shelter in many marine organisms, but these interactions can be impacted by life stage and presence of predators. Juvenile lobsters (Homarus americanus) have social hierarchies that govern access to valuable resources like food and shelter, but these hierarchies could be impacted by the presence of invasive green crabs (Carcinus maenas). The goal of this study was to determine whether the presence of green crab effluent affected aggressive behavior in different sizes of juvenile lobsters, with the hypothesis that all juvenile lobsters would be more likely to share shelter under threat of predation. Small juveniles (15 mm carapace length) and larger juveniles (45 mm) had substantially different interactions and responses to predator threat cues, with small individuals more likely to both fight over shelter and share shelter when exposed to green crab effluent. There were also several size-based behavioral differences in control treatments, as small juvenile lobsters challenged each other more frequently for shelter and spent vastly more time in the shelter, less time stationary outside the shelter, and less time fighting. Despite being vulnerable to green crab predation, larger juvenile lobsters were generally unaffected by crab chemical cues. This suggests that life history plays a strong role in predator avoidance behavior and that both lethal and nonlethal effects of invasive species on native prey can vary markedly with life stage. Future research should examine these crab-lobster interactions in lobster nursery habitats in the field, as invasive green crabs in these habitats could change behavior, activity patterns and survival of juvenile lobsters.


Similar content being viewed by others
Availability of data
The datasets generated during and/or analyzed during the current study are available in the Figshare repository, and a link will be provided here after publication.
References
Baillie CJ, Grabowski JH (2018) Competitive and agonistic interactions between the invasive Asian shore crab and juvenile American lobster. Ecology 99:2067–2079. https://doi.org/10.1002/ecy.2432
Barshaw D, Lavalli K (1988) Predation upon postlarval lobsters Homarus americanus by cunners Tautogolabrus adspersus and mud crabs Neopanope sayi on three different substrates: eelgrass, mud and rocks. Mar Ecol Prog Ser 48:119–123. https://doi.org/10.3354/meps048119
Blakeslee AMH, McKenzie CH, Darling JA, Byers JE, Pringle JM, Roman J (2010) A hitchhiker’s guide to the Maritimes: anthropogenic transport facilitates long-distance dispersal of an invasive marine crab to Newfoundland. Divers Distrib 16:879–891. https://doi.org/10.1111/j.1472-4642.2010.00703.x
Borroni PF, Handrich LS, Atema J (1986) The role of narrowly tuned taste cell populations in lobster (Homarus americanus) feeding behavior. Behav Neurosci 100:206–212
Breen PA, Mann KH (1976) Changing lobster abundance and the destruction of kelp beds by sea urchins. Mar Biol 34:137–142. https://doi.org/10.1007/BF00390755
Bruce M, Doherty T, Kaplan J, Sutherland C, Atema J (2018) American lobsters, Homarus americanus, use vision for initial opponent evaluation and subsequent memory. Bull Mar Sci 94:517–532. https://doi.org/10.5343/bms.2017.1147
Chase ID, Seitz K (2011) Self-structuring properties of dominance hierarchies. In: Huber R, Bannasch DL, Brennan P (eds) Advances in genetics. Elsevier, pp 51–81
Cobb JS, Tamm GR (1975) Dominance status and molt order in lobsters (Homarus americanus). Mar Behav Physiol 3:119–124. https://doi.org/10.1080/10236247509378501
Cowan DF, Solow AR, Beet A (2001) Patterns in abundance and growth of juvenile lobster, Homarus americanus. Mar Freshw Res 52:1095. https://doi.org/10.1071/MF01191
Demeo A, Riley JG (2006) Hemigrapsus sanguineus (Asian shore crab) as predator of juvenile Homarus americanus (American lobster). Maine Agric Forest Exp Station Tech Bull 194.
DFO (2018) Seafisheries landed value by region, 2018. http://www.dfo-mpo.gc.ca/stats/commercial/land-debarq/sea-maritimes/s2018av-eng.htm. Accessed 15 Jan 2020
Ellis S, Cowan D (2001) Volunteer-based monitoring of juvenile American lobster, Homarus americanus. Mar Freshw Res 52:1103–1112. https://doi.org/10.1071/MF01194
Freeman AS, Byers JE (2006) Divergent induced responses to an invasive predator in marine mussel populations. Science 313:831–833. https://doi.org/10.1126/science.1125485
Goldstein JS, Morrissey EM, Moretti ED, Watson WH (2017) A comparison of the distribution and abundance of European green crabs and American lobsters in the Great Bay Estuary, New Hampshire, USA. Fish Res 189:10–17. https://doi.org/10.1016/j.fishres.2017.01.002
Grosholz ED, Ruiz GM (1996) Predicting the impact of introduced marine species: Lessons from the multiple invasions of the European green crab Carcinus maenas. Biol Conserv 78:59–66. https://doi.org/10.1016/0006-3207(94)00018-2
Haarr ML, Rochette R (2012) The effect of geographic origin on interactions between adult invasive green crabs Carcinus maenas and juvenile American lobsters Homarus americanus in Atlantic Canada. J Exp Mar Biol Ecol 422–423:88–100. https://doi.org/10.1016/j.jembe.2012.04.016
Hanson JM (2009) Predator-prey interactions of American lobster (Homarus americanus) in the southern Gulf of St. Lawrence, Canada. N Z J Mar Freshw Res 43:69–88. https://doi.org/10.1080/00288330909509983
Hoeppner S (1997) The importance of chemoreception for the recognition of dominance hierarchies in the American lobster, Homarus americanus. Beta Beta Beta Biol Soc 68:91–101
Jones PL, Shulman MJ (2008) Subtidal-intertidal trophic links: American lobsters [Homarus americanus (Milne-Edwards)] forage in the intertidal zone on nocturnal high tides. J Exp Mar Biol Ecol 361:98–103. https://doi.org/10.1016/j.jembe.2008.05.004
Karnofsky EB, Atema J, Elgin RH (1989) Field observations of social behavior, shelter use, and foraging in the lobster, Homarus americanus. Biol Bull 176:239–246. https://doi.org/10.2307/1541982
Kimbro DL, Grabowski JH, Hughes AR, Piehler MF, White JW (2017) Nonconsumptive effects of a predator weaken then rebound over time. Ecology 98:656–667. https://doi.org/10.1002/ecy.1702
Lawton P (1987) Diel activity and foraging behavior of juvenile American lobsters, Homarus americanus. Can J Fish Aquat Sci 44:1195–1205. https://doi.org/10.1139/f87-142
League-Pike PE, Shulman MJ (2009) Intraguild predators: behavioral changes and mortality of the green crab (Carcinus maenas) during interactions with the American lobster (Homarus americanus) and Jonah crab (Cancer borealis). J Crustac Biol 29:350–355. https://doi.org/10.1651/08-3066.1
Leonard GH, Bertness MD, Yund PO (1999) Crab predation, waterborne cues, and inducible defenses in the blue mussel, Mytilus edulis. Ecology 80:1. https://doi.org/10.2307/176976
Lord JP, Dalvano BE (2015) Differential response of the American lobster Homarus americanus to the invasive Asian shore crab Hemigrapsus sanguineus and green crab Carcinus maenas. J Shellfish Res 34:1091–1096. https://doi.org/10.2983/035.034.0336
Lord JP, Williams LM (2017) Increase in density of genetically diverse invasive Asian shore crab (Hemigrapsus sanguineus) populations in the Gulf of Maine. Biol Invasions 19:1153–1168. https://doi.org/10.1007/s10530-016-1304-1
Lord JP, Moser RM, Buonocore EM, Sylvester EE, Morales MJ, Granitz AP, DiSipio Jr A, Blakely E, O’Sullivan-Evangelista SL, Mateo TF, Chlebove GJ, Carey CM, Lucas O (2021) Dominance hierarchies in marine invertebrates. Biol Bull 240:2–15. https://doi.org/10.1086/712973
Lubchenco J (1978) Plant species diversity in a marine intertidal community: importance of herbivore food preference and algal competitive abilities. Am Nat 112:23–39
Lynch BR, Rochette R (2009) Spatial overlap and interaction between sub-adult American lobsters, Homarus americanus, and the invasive European green crab Carcinus maenas. J Exp Mar Biol Ecol 369:127–135. https://doi.org/10.1016/j.jembe.2008.11.002
McMahan MD, Grabowski JH (2019) Nonconsumptive effects of a range-expanding predator on juvenile lobster (Homarus americanus) population dynamics. Ecosphere. https://doi.org/10.1002/ecs2.2867
O’Neill DJ, Cobb JS (1979) Some factors influencing the outcome of shelter competition in lobsters (Homarus americanus). Mar Behav Physiol 6:33–45. https://doi.org/10.1080/10236247909378551
Paille N, Sainte-Marie B, Br Êthes J-C (2002) Behavior, growth and survival of stage V lobsters (Homarus americanus) in relation to shelter availability and lobster density. Mar Freshw Behav Physiol 35:203–219. https://doi.org/10.1080/1023624021000019324
Quinn BK, Chassé J, Rochette R (2017) Potential connectivity among American lobster fisheries as a result of larval drift across the species’ range in eastern North America. Can J Fish Aquat Sci 74:1549–1563. https://doi.org/10.1139/cjfas-2016-0416
Rayner G, Bird TJ, McGaw IJ (2019) Quantifying factors influencing American lobster (Homarus americanus) predation on the invasive green crab (Carcinus maenas) in Newfoundland. Canada Invertebr Biol. https://doi.org/10.1111/ivb.12245
Roman J (2006) Diluting the founder effect: cryptic invasions expand a marine invader’s range. Proc R Soc Lond B Biol Sci 273:2453–2459. https://doi.org/10.1098/rspb.2006.3597
Rossong MA, Williams PJ, Comeau M, Mitchell SC, Apaloo J (2006) Agonistic interactions between the invasive green crab, Carcinus maenas (Linnaeus) and juvenile American lobster, Homarus americanus (Milne Edwards). J Exp Mar Biol Ecol 329:281–288. https://doi.org/10.1016/j.jembe.2005.09.007
Rossong MA, Quijón PA, Williams PJ, Snelgrove PVR (2011) Foraging and shelter behavior of juvenile American lobster (Homarus americanus): the influence of a non-indigenous crab. J Exp Mar Biol Ecol 403:75–80. https://doi.org/10.1016/j.jembe.2011.04.008
Rossong MA, Quijón PA, Snelgrove PVR, Barrett TJ, McKenzie CH, Locke A (2012) Regional differences in foraging behaviour of invasive green crab (Carcinus maenas) populations in Atlantic Canada. Biol Invasions 14:659–669. https://doi.org/10.1007/s10530-011-0107-7
Rutishauser RL, Basu AC, Cromarty SI, Kravitz EA (2004) Long-term consequences of agonistic interactions between socially naïve juvenile American lobsters (Homarus americanus). Biol Bull 207:183–187. https://doi.org/10.2307/1543205
Sigurdsson GM, Rochette R (2013) Predation by green crab and sand shrimp on settling and recently settled American lobster postlarvae. J Crustac Biol 33:10–14. https://doi.org/10.1163/1937240X-00002107
Spanier E, McKenzie TP, Cobb JS, Clancy M (1998) Behavior of juvenile American lobsters, Homarus americanus, under predation risk. Mar Biol 130:397–406. https://doi.org/10.1007/s002270050260
Tyrrell MC, Guarino PA, Harris LG (2006) Predatory impacts of two introduced crab species: inferences from microcosms. Northeast Nat 13:375–390
Wahle RA (1992) Body-size dependent anti-predator mechanisms of the American lobster. Oikos 65:52–60. https://doi.org/10.2307/3544887
Wahle RA, Steneck RS (1991) Recruitment habitats and nursery grounds of the American lobster Homarus americanus: a demographic bottleneck? Mar Ecol Prog Ser 69:231–243. https://doi.org/10.3354/meps069231
Wahle RA, Steneck RS (1992) Habitat restrictions in early benthic life: experiments on habitat selection and in situ predation with the American lobster. J Exp Mar Biol Ecol 157:91–114. https://doi.org/10.1016/0022-0981(92)90077-N
Williams PJ, Floyd TA, Rossong MA (2006) Agonistic interactions between invasive green crabs, Carcinus maenas (Linnaeus), and sub-adult American lobsters, Homarus americanus (Milne Edwards). J Exp Mar Biol Ecol 329:66–74. https://doi.org/10.1016/j.jembe.2005.08.008
Williams PJ, MacSween C, Rossong M (2009) Competition between invasive green crab (Carcinus maenas) and American lobster (Homarus americanus). N Z J Mar Freshw Res 43:29–33. https://doi.org/10.1080/00288330909509979
Acknowledgements
This work was supported by the Moravian College Department of Biological Sciences. Thank you to undergraduate students Katie Mayer, Midelys Franceschini, and Sam Rappaport for their help keeping the lobsters alive, clean, and well-fed in the lab. Work performed under New England Aquarium IACUC Protocol 2019-02. Additional thanks to 3 anonymous reviewers and the associate editor for their substantive and helpful feedback, which improved this manuscript.
Funding
This work was supported by the Department of Biological Sciences and no outside agency.
Author information
Authors and Affiliations
Contributions
I am the sole author and conducted the research, analysis, and writing myself.
Corresponding author
Ethics declarations
Conflicts of interest
The author declares they have no conflict of interest.
Ethics approval
No human subjects were used in this study.
Animal welfare
No animals were killed or harmed during these experiments. Experiments with invertebrates are exempt from IACUC approval at most institutions including Moravian College, but because the lobsters were obtained from the New England Aquarium, their housing and use in these experiments was reviewed and approved by the New England Aquarium IACUC (Protocol 2019–02). IACUC approval is not required for invertebrate research at Moravian College.
Additional information
Responsible Editor: M. Thiel.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewers: undisclosed experts.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lord, J.P. Size affects intraspecific aggression and response to predation threat in juvenile American lobsters. Mar Biol 168, 51 (2021). https://doi.org/10.1007/s00227-021-03864-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-021-03864-5