Abstract
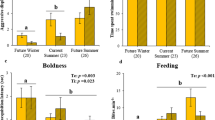
The effect of temperature on predator–prey dynamics has the potential to be an important factor regulating ecological interactions and is becoming increasingly common due to climate-change-driven range shifts and species introductions. Here, we use mesocosm experiments to evaluate the thermal sensitivity of burst swimming (a proxy for prey escape ability) and mortality rates of cohorts of introduced tropical and resident temperate marine fishes in the presence of a local temperate predator. Increasing temperature (17–21–25 °C) resulted in a reduction in predation rates (by Hypoplectrodes maccullochi, family Serranidae) on the tropical prey species (Abudefduf vaigiensis, family Pomacentridae) which were more warm adapted, whereas predation rates on the temperate prey species (Atypichthys strigatus, family Kyphosidae) remained unchanged over the 17–21 °C range (25 °C not tested). These changes were linearly related to predator–prey burst swimming ratios, which decreased with increasing temperature for the tropical prey but remained largely unchanged for the temperate prey. By demonstrating the temperature sensitivity of predator–prey interactions, our work highlights the importance of linking physiology with ecology to understand the consequences of climate-driven range shifts and species introductions.



Similar content being viewed by others
References
Abrahams MV, Mangel M, Hedges K (2007) Predator–prey interactions and changing environments: who benefits? Philos Trans R Soc B Biol Sci 362:2095–2104. https://doi.org/10.1098/rstb.2007.2102
Allan BJM, Domenici P, Munday PL, McCormick MI (2015) Feeling the heat: the effect of acute temperature changes on predator–prey interactions in coral reef fish. Conserv Physiol. https://doi.org/10.1093/conphys/cov011
Almany GR (2004) Does increased habitat complexity reduce predation and competition in coral reef fish assemblages? Biol Abstr 106:275–284 (Vol 99, Iss 6, Ref 76189)
Angilletta MJ (2009) Thermal adaptation: a theoretical and empirical synthesis. Oxford University Press, Oxford
Booth D, Figueira W, Gregson M, Brown L, Berreta G (2007) Occurrence of tropical fishes in temperate southeastern Australia: role of the East Australian current. Estuar Coast Shelf Sci 72:102–114. https://doi.org/10.1016/j.ecss.2006.10.003
Dell AI, Pawar S, Savage VM (2014) Temperature dependence of trophic interactions are driven by asymmetry of species responses and foraging strategy. Biol Abstr 83:70–84. https://doi.org/10.1111/1365-2656.12081 (Vol 100, Iss 12, Ref 182076)
Diaz JA (1994) Effects of body-temperature on the predatory behaviour of the lizard Psammodromus algirus hunting winged and wingless prey. Herpetol J 4:145–150
Domenici P (2010) Context-dependent variability in the components of fish escape response: integrating locomotor performance and behavior. J Exp Zool Part A Ecol Genet Physiol 313A:59–79. https://doi.org/10.1002/jez.580
Domenici P, Claireaux G, McKenzie DJ (2007) Environmental constraints upon locomotion and predator–prey interactions in aquatic organisms: an introduction. Philos Trans R Soc B Biol Sci 362:1929–1936. https://doi.org/10.1098/rstb.2007.2078
Donelson JM, Munday PL, McCormick MI, Pitcher CR (2012) Rapid transgenerational acclimation of a tropical reef fish to climate change. Nat Clim Change 2:30–32
Englund G, Öhlund G, Hein CL, Diehl S (2011) Temperature dependence of the functional response. Ecol Lett 14:914–921. https://doi.org/10.1111/j.1461-0248.2011.01661.x
Estes JA, Terborgh J, Brashares JS, Power ME, Berger J, Bond WJ, Carpenter SR, Essington TE, Holt RD, Jackson JBC, Marquis RJ, Oksanen L, Oksanen T, Paine RT, Pikitch EK, Ripple WJ, Sandin SA, Scheffer M, Schoener TW, Shurin JB, Sinclair ARE, Soulé ME, Virtanen R, Wardle DA (2011) Trophic downgrading of planet earth. Science 333:301–306. https://doi.org/10.1126/science.1205106
Figueira W, Booth D (2010) Increasing ocean temperatures allow tropical fishes to survive overwinter in temperate waters. Glob Change Biol 16:506–516. https://doi.org/10.1111/j.1365-2486.2009.01934.x
Figueira W, Booth D, Gregson M (2008) Selective mortality of a coral reef damselfish: role of predator–competitor synergisms. Oecologia 156:215–226. https://doi.org/10.1007/s00442-008-0985-2
Figueira W, Biro P, Booth D, Valenzuela V (2009) Performance of tropical fish recruiting to temperate habitats: role of ambient temperature and implications of climate change. Mar Ecol Prog Ser 384:231–239. https://doi.org/10.3354/meps08057
Galaiduk R, Figueira WF, Kingsford MJ, Curley BG (2013) Factors driving the biogeographic distribution of two temperate Australian damselfishes and ramifications for range shifts. Mar Ecol Prog Ser 484:189–202. https://doi.org/10.3354/meps10300
Giles N (1984) Development of the overhead fright response in wild and predator-naive three-spined sticklebacks, Gasterosteus aculeatus L. Anim Behav 32:276–279. https://doi.org/10.1016/S0003-3472(84)80347-4
Grigaltchik VS, Ward AJW, Seebacher F (2012) Thermal acclimation of interactions: differential responses to temperature change alter predator–prey relationship. Proc R Soc B 279:4058–4064. https://doi.org/10.1098/rspb.2012.1277
Gurevitch J, Morrison JA, Hedges LV (2000) The interaction between competition and predation: a meta-analysis of field experiments. Biol Abstr 155:435–453 (Vol 104, Iss 9, Ref 125297)
Hein CL, Öhlund G, Englund G (2014) Fish introductions reveal the temperature dependence of species interactions. Proc R Soc B. https://doi.org/10.1098/rspb.2013.2641
Holt RD (1977) Predation, apparent competition, and the structure of prey communities. Biol Abstr 12:197–229. https://doi.org/10.1016/0040-5809(77)90042-9 (Vol 102, Iss 12, Ref 175678)
Kordas RL, Harley CDG, O’Connor MI (2011) Community ecology in a warming world: the influence of temperature on interspecific interactions in marine systems. J Exp Mar Biol Ecol 400:218–226. https://doi.org/10.1016/j.jembe.2011.02.029
López MS, Coutinho R, Ferreira CEL, Rilov G (2010) Predator–prey interactions in a bioinvasion scenario: differential predation by native predators on two exotic rocky intertidal bivalves. Mar Ecol Prog Ser 403:101–112. https://doi.org/10.3354/meps08409
Lyon JP, Ryan TJ, Scroggie MP (2008) Effects of temperature on the fast-start swimming performance of an Australian freshwater fish. Ecol Freshw Fish 17:184–188. https://doi.org/10.1111/j.1600-0633.2007.00244.x
Meekan MG, Vigliola L, Hansen A, Doherty PJ, Halford A, Carleton JH (2006) Bigger is better: size-selective mortality throughout the life history of a fast-growing clupeid, Spratelloides gracilis. Mar Ecol Prog Ser 317:237–244
Miller LP, Matassa CM, Trussell GC (2014) Climate change enhances the negative effects of predation risk on an intermediate consumer. Glob Change Biol 20:3834–3844. https://doi.org/10.1111/Gcb.12639
Monaco CJ, Helmuth B (2011) Tipping points, thresholds and the keystone role of physiology in marine climate change research. Adv Mar Biol 60:123–160. https://doi.org/10.1016/B978-0-12-385529-9.00003-2
Monaco CJ, Wethey DS, Helmuth B (2016) Thermal sensitivity and the role of behavior in driving an intertidal predator–prey interaction. Ecol Monogr 86:429–447. https://doi.org/10.1002/ecm.1230
Monclús R, Rödel HG, Von Holst D, De Miguel J (2005) Behavioural and physiological responses of naïve European rabbits to predator odour. Anim Behav 70:753–761. https://doi.org/10.1016/j.anbehav.2004.12.019
Morley JW, Buckel JA (2014) Effects of temperature and prey size on predator–prey interactions between bluefish and bay anchovy. J Exp Mar Biol Ecol 461:449–457. https://doi.org/10.1016/j.jembe.2014.08.023
Munday PL, McCormick MI, Nilsson GE (2012) Impact of global warming and rising CO2 levels on coral reef fishes: what hope for the future? J Exp Biol 215:3865–3873
Ockendon N, Baker DJ, Carr JA, White EC, Almond REA, Amano T, Bertram E, Bradbury RB, Bradley C, Butchart SHM, Doswald N, Foden W, Gill DJC, Green RE, Sutherland WJ, Tanner EVJ, Pearce-Higgins JW (2014) Mechanisms underpinning climatic impacts on natural populations: altered species interactions are more important than direct effects. Glob Change Biol 20:2221–2229. https://doi.org/10.1111/gcb.12559
Ohlund G, Hedstrom P, Norman S, Hein CL, Englund G (2015) Temperature dependence of predation depends on the relative performance of predators and prey. Proc R Soc B Biol Sci. https://doi.org/10.1098/rspb.2014.2254
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42
Pincebourde S, Sanford E, Casas J, Helmuth B (2012) Temporal coincidence of environmental stress events modulates predation rates. Ecol Lett 15:680–688. https://doi.org/10.1111/j.1461-0248.2012.01785.x
Pintor LM, Byers JE (2015) Individual variation in predator behavior and demographics affects consumption of non-native prey. Behav Ecol. https://doi.org/10.1093/beheco/arv1013
Poloczanska ES, Brown CJ, Sydeman WJ, Kiessling W, Schoeman DS, Moore PJ, Brander K, Bruno JF, Buckley LB, Burrows MT, Duarte CM, Halpern BS, Holding J, Kappel CV, O’Connor MI, Pandolfi JM, Parmesan C, Schwing F, Thompson SA, Richardson AJ (2013) Global imprint of climate change on marine life. Nat Clim Change 3:919–925. https://doi.org/10.1038/nclimate1958
Scharf FS, Buckel JA, McGinn PA, Juanes F (2003) Vulnerability of marine forage fishes to piscivory: effects of prey behavior on susceptibility to attack and capture. J Exp Mar Biol Ecol 294:41–59. https://doi.org/10.1016/s0022-0981(03)00260-0
Selden RL, Batt RD, Saba VS, Pinsky ML (2018) Diversity in thermal affinity among key piscivores buffers impacts of ocean warming on predator–prey interactions. Glob Chang Biol 24:117–131. https://doi.org/10.1111/gcb.13838
Smith SM, Fox RJ, Booth DJ, Donelson JM (2018) ‘Stick with your own kind, or hang with the locals?’ Implications of shoaling strategy for tropical reef fish on a range-expansion frontline. Glob Change Biol 24:1663–1672. https://doi.org/10.1111/gcb.14016
Smolinsky R, Gvozdik L (2014) Effect of temperature extremes on the spatial dynamics of predator–prey interactions: a case study with dragonfly nymphs and newt larvae. J Therm Biol 39:12–16. https://doi.org/10.1016/j.jtherbio.2013.11.004
Sunday JM, Bates AE, Dulvy NK (2012) Thermal tolerance and the global redistribution of animals. Nat Clim Change 2:686–690
Verges A, Steinberg PD, Hay ME, Poore AGB, Campbell AH, Ballesteros E, Heck KL Jr, Booth DJ, Coleman MA, Feary DA, Figueira W, Langlois T, Marzinelli EM, Mizerek T, Mumby PJ, Nakamura Y, Roughan M, van Sebille E, Sen Gupta A, Smale DA, Tomas F, Wernberg T, Wilson SK (2014) The tropicalization of temperate marine ecosystems: climate-mediated changes in herbivory and community phase shifts. Proc R Soc B Biol Sci 281:20140846. https://doi.org/10.1098/rspb.2014.0846
Webb PW, Zhang HB (1994) The relationship between responsiveness and elusiveness of heat-shocked goldfish (Carrassius auratus) to attacks by rainbow trout (Oncorhynchus mykiss). Can J Zool 72:423–426. https://doi.org/10.1139/z94-059
Acknowledgements
Thanks to Ronen Galaiduk and Belle McCarthy for assistance with collections and husbandry, and to Jenni Donnelson for valuable comments on the manuscript. And thanks to the staff at the Sydney Institute of Marine Science and BioSys Consulting for continued support of the controlled temperature seawater system. All work was conducted under NSW Fisheries Permit number F96/146-6.0. This is contribution number 243 to the Sydney Institute of Marine Science.
Funding
This work was supported by a NSW Environmental Trust grant to D Booth and W Figueira.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All research was conducted in accordance with the University of Sydney’s guidelines for animal ethics and under permits 5200 and 5214 to W. Figueira.
Data availability
The datasets during and/or analysed during the current study are available from the corresponding author on reasonable request.
Additional information
Responsible Editor: E. Hunter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by A. Verges and an undisclosed expert.
Rights and permissions
About this article
Cite this article
Figueira, W.F., Curley, B. & Booth, D.J. Can temperature-dependent predation rates regulate range expansion potential of tropical vagrant fishes?. Mar Biol 166, 73 (2019). https://doi.org/10.1007/s00227-019-3521-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-019-3521-5