Abstract
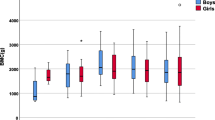
Adults with Type 1 diabetes mellitus show a high risk of bone fracture, probably as a consequence of a decreased bone mass and microarchitectural bone alterations. The aim of the study was to investigate the potential negative effects of type 1 diabetes on bone geometry, quality, and bone markers in a group of children and adolescents. 96 children, mean age 10.5 ± 3.1 years, agreed to participate to the study. Bone geometry was evaluated on digitalized X-rays at the level of the 2nd metacarpal bone. The following parameters were investigated and expressed as SDS: outer diameter (D), inner diameter (d), cortical area (CA), and medullary area (MA). Bone strength was evaluated as Bending Breaking Resistance Index (BBRI) from the geometric data. Bone turnover markers (PINP, CTX-I, and BAP), sclerostin, Dkk-1, PTH, and 25OH-Vitamin D were also assessed. A group of healthy 40 subjects of normal body weight and height served as controls for the bone markers. D (− 0.99 ± 0.98), d (− 0.41 ± 0.88), CA (− 0.85 ± 0.78), and MA (− 0.46 ± 0.78) were all significantly smaller than in controls (p < 0.01). BBRI was significantly lower (− 2.61 ± 2.18; p < 0.0001). PTH, PINP, and BAP were higher in the diabetic children. Multiple regression analysis showed that CA and D were influenced by insulin/Kg/day and by BMI, while d was influenced by PINP only. Type 1 diabetic children show smaller and weaker bones. The increased bone turnover could play a key role since it might amplify the deficit in bone strength associated with the inadequate osteoblastic activity caused by the disease itself.


Similar content being viewed by others
References
Pan H, Wu N, Yang T, He W (2014) Association between bone mineral density and type 1 diabetes mellitus: a meta-analysis of cross-sectional studies. Diabetes Metab Res Rev 30:531–542
Coe LM, Irwin R, Lippner D, McCabe LR (2011) The bone marrow microenvironment contributes to type I diabetes induced osteoblast death. J Cell Physiol 226:477–483
Hie M, Iitsuka N, Otsuka T, Tsukamoto I (2011) Insulin-dependent diabetes mellitus decreases osteoblastogenesis associated with the inhibition of Wnt signaling through increased expression of Sost and Dkk1 and inhibition of Akt activation. Int J Mol Med 28:455–462
Patti A, Gennari L, Merlotti D, Dotta F, Nuti R (2013) Endocrine actions of osteocalcin. Int J Endocrinol. https://doi.org/10.1155/2013/846480
Armas LA, Akhter MP, Drincic A, Recker RR (2012) Trabecular bone histomorphometry in humans with type 1 diabetes mellitus. Bone 50:91–96
Moreira CA, Dempster DW (2015) Bone histomorphometry in diabetes mellitus. Osteoporos Int 26:2559–2560
Verroken C, Pieters W, Beddeleem L, Goemaere S, Zmierczak HG, Shadid S, Kaufman JM, Lapauw B (2017) Cortical bone size deficit in adult patients with type 1 diabetes mellitus. J Clin Endocrinol Metab 102:2887–2895
Shanbhogue VV, Hansen S, Frost M, Jørgensen NR, Hermann AP, Henriksen JE, Brixen K (2015) Bone geometry, volumetric density, microarchitecture, and estimated bone strength assessed by HR-pQCT in adult patients with type 1 diabetes mellitus. J Bone Miner Res 30:2188–2199
Turner CH, Burr DB (1993) Basic biomechanical measurements of bone: a tutorial. Bone 14:595–608
Bechtold S, Dirlenbach I, Raile K, Noelle V, Bonfig W, Schwarz HP (2006) Early manifestation of type 1 diabetes in children is a risk factor for changed bone geometry: data using peripheral quantitative computed tomography. Pediatrics 118:e627–e634
Saha MT, Sievänen H, Salo MK, Tulokas S, Saha HH (2008) Bone mass and structure in adolescents with type 1 diabetes compared to healthy peers. Osteoporos Int 20:1401–1406
Bechtold S, Putzker S, Bonfig W, Fuchs O, Dirlenbach I, Schwarz HP (2007) Bone size normalizes with age in children and adolescents with type 1 diabetes. Diabetes Care 30:2046–2050
Roggen I, Gies I, Vanbesien J, Louis O, De Schepper J (2013) Trabecular bone mineral density and bone geometry of the distal radius at completion of pubertal growth in childhood type 1 diabetes. Horm Res Paediatr 79:68–74
Fowlkes JL, Bunn RC, Thrailkill KM (2011) Contributions of the insulin/insulin-like growth factor-1 axis to diabetic osteopathy. J Diabetes Metab 25:S1-S3
Fowlkes JL, Nyman JS, Bunn RC, Jo C, Wahl EC, Liu L, Cockrell GE, Morris LM, Lumpkin CK Jr, Thrailkill KM (2013) Osteo-promoting effects of insulin-like growth factor I (IGF-I) in a mouse model of type 1 diabetes. Bone 57:36–40
Botolin S, McCabe LR (2007) Bone loss and increased bone adiposity in spontaneous and pharmacologically induced diabetic mice. Endocrinology 148:198–205
Neumann T, Lodes S, Kästner B, Franke S, Kiehntopf M, Lehmann T, Müller UA, Wolf G, Sämann A (2014) High serum pentosidine but not esRAGE is associated with prevalent fractures in type 1 diabetes independent of bone mineral density and glycaemic control. Osteoporos Int 25:1527–1533
Baroncelli GI, Federico G, Bertelloni S, Sodini F, De Terlizzi F, Cadossi R, Saggese G (2003) Assessment of bone quality by quantitative ultrasound of proximal phalanges of the hand and fracture rate in children and adolescents with bone and mineral disorders. Pediatr Res 54:125–136
Catalano A, Pintaudi B, Morabito N, Di Vieste G, Giunta L, Bruno ML, Cucinotta D, Lasco A, Di Benedetto A (2014) Gender differences in sclerostin and clinical characteristics in type 1 diabetes mellitus. Eur J Endocrinol 171:293–300
Chobot AP, Haffke A, Polanska J, Halaba ZP, Deja G, Jarosz-Chobot P, Pluskiewicz W (2012) Quantitative ultrasound bone measurements in pre-pubertal children with type 1 diabetes. Ultrasound Med Biol 38:1109–1115
Faienza MF, Ventura A, Delvecchio M, Fusillo A, Piacente L, Aceto G, Colaianni G, Colucci S, Cavallo L, Grano M, Brunetti G (2017) High sclerostin and dickkopf-1 (DKK-1) Serum levels in children and adolescents with type 1 diabetes mellitus. J Clin Endocrinol Metab 102:1174–1181
Szulc P, Delmas PD (2008) Biochemical markers of bone turnover: potential use in the investigation and management of postmenopausal osteoporosis. Osteoporos Int 19:1683–1704
Hygum K, Starup-Linde J, Harsløf T, Vestergaard P, Langdahl BL (2017) Mechanisms in endocrinology: diabetes mellitus, a state of low bone turnover—a systematic review and meta-analysis. Eur J Endocrinol 176:R137-R157
Baron R, Kneissel M (2013) WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat Med 19:179–192
Neumann T, Hofbauer LC, Rauner M, Lodes S, Kästner B, Franke S, Kiehntopf M, Lehmann T, Müller UA, Wolf G, Hamann C, Sämann A (2014) Clinical and endocrine correlates of circulating sclerostin levels in patients with type 1 diabetes mellitus. Clin Endocrinol 80:649–655
Gennari L, Merlotti D, Valenti R, Ceccarelli E, Ruvio M, Pietrini MG, Capodarca C, Franci MB, Campagna MS, Calabrò A, Cataldo D, Stolakis K, Dotta F, Nuti R (2012) Circulating sclerostin levels and bone turnover in type 1 and type 2 diabetes. J Clin Endocrinol Metab 97:1737–1744
Tsentidis C, Gourgiotis D, Kossiva L, Marmarinos A, Doulgeraki A, Karavanaki K (2017) Increased levels of Dickkopf-1 are indicative of Wnt/β-catenin downregulation and lower osteoblast signaling in children and adolescents with type 1 diabetes mellitus, contributing to lower bone mineral density. Osteoporos Int 28:945–953
Cacciari E, Milani S, Balsamo A, Spada E, Bona G, Cavallo L, Cerutti F, Gargantini L, Greggio N, Tonini G, Cicognani A (2006) Italian cross-sectional growth charts for height, weight and BMI (2 to 20 year). J Endocrinol Invest 29:581–593
McCarthy HD, Jarrett KV, Crawley HF (2001) The development of waist circumference percentiles in British children aged 5.0–16.9 y. 55:902–907
Tanner JM (1962) Growth at adolescence, 2nd edn. Blackwell, Oxford
Greulich WW, Pyle SI (1959) Radiographic atlas of skeletal development of the hand and wrist, 2nd edn. Stanford University Press, Stanford
Zamberlan N, Radetti G, Paganini C, Gatti D, Rossini M, Braga V, Adami S (1996) Evaluation of cortical thickness and bone density by roentgen microdensitometry in growing males and females. Eur J Pediatr 155:377–382
Genant HK, Engelke K, Fuerst T, Glüer CC, Grampp S, Harris ST, Jergas M, Lang T, Lu Y, Majumdar S, Mathur A, Takada M (1996) Noninvasive assessment of bone mineral and structure: state of the art. J Bone Miner Res 11:707–730
Radetti G, Franceschi R, Adami S, Longhi S, Rossini M, Gatti D (2014) Higher circulating parathormone is associated with smaller and weaker bones in obese children. Calcif Tissue Int 95:1–7
Gatti D, Colapietro F, Fracassi E, Sartori E, Antoniazzi F, Braga V, Rossini M, Adami S (2003) The volumetric bone density and cortical thickness in adult patients affected by osteogenesis imperfecta. J Clin Densitom 6:173–177
Wilczek ML, Kälvesten J, Algulin J, Beiki O, Brismar TB (2013) Digital X-ray radiogrammetry of hand or wrist radiographs can predict hip fracture risk—a study in 5420 women and 2837 men. Eur Radiol 23:1383–1391
Hamed EA, Faddan NHA, Elhafeez HAA, Sayed D (2011) Parathormone-25(OH)-vitamin D axis and bone status in children and adolescents with type 1 diabetes mellitus. Pediatr Diabetes 12(6):536–546
Neumann T, Hofbauer LC, Rauner M, Lodes S, Kästner B, Franke S et al (2014) Clinical and endocrine correlates of circulating sclerostin levels in patients with type 1 diabetes mellitus. Clin Endocrinol 80(5):649–655
Aljabri KS, Bokhari SA, Khan MJ (2010) Glycemic changes after vitamin D supplementation in patients with type 1 diabetes mellitus and vitamin D deficiency. Ann Saudi Med 30(6):454–458
Buhary BM, Almohareb O, Aljohani N, Alrajhi S, Elkaissi S, Sherbeeni S et al (2017) Association of glycosylated hemoglobin levels with vitamin D status. J Clin Med Res 9(12):1013–1018
Braga V, Gatti D, Rossini M, Colapietro F, Battaglia E, Viapiana O, Adami S (2004) Bone turnover markers in patients with osteogenesis imperfecta. Bone 34:1013–1016
Rossini M, Gatti D, Adami S (2013) Involvement of WNT/β-catenin signaling in the treatment of osteoporosis. Calcif Tissue Int 93:121–132
Fassio A, Rossini M, Viapiana O, Idolazzi L, Benini C, Vantaggiato E, Gatti D (2017) New strategies for the prevention and treatment of systemic and local bone loss; from pathophysiology to clinical application. Curr Pharm Des. https://doi.org/10.2174/1381612823666170713104431
Fricke O, Schoenau E (2007) The ‘Functional Muscle-Bone Unit’: probing the relevance of mechanical signals for bone development in children and adolescents. Growth Horm IGF Res 17:1–9
Acknowledgements
The authors would like to thank Caterina Fraccarollo for ELISA assays and the LURM (Laboratorio Universitario di Ricerca Medica) Research Centre of University of Verona, where this study was partially performed.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
(Roberto Franceschi, Silvia Longhi, Vittoria Cauvin, Angelo Fassio, Giuseppe Gallo, Fiorenzo Lupi, Petra Reinstadler, Antonio Fanolla, Davide Gatti, and Giorgio Radetti) have no financial conflicts of interest. The authors have full control of all primary data and they agree to allow the journal to review their data if requested.
Human and Animal Rights and Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
Rights and permissions
About this article
Cite this article
Franceschi, R., Longhi, S., Cauvin, V. et al. Bone Geometry, Quality, and Bone Markers in Children with Type 1 Diabetes Mellitus. Calcif Tissue Int 102, 657–665 (2018). https://doi.org/10.1007/s00223-017-0381-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-017-0381-1