Abstract
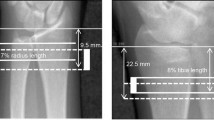
Obesity has been considered to have a protective effect against the risk of fractures in adults. However, a high frequency of fracture is described in obese adults with Prader–Willi syndrome. To evaluate bone geometry, density and strength in a group of adult obese patients with Prader–Willi syndrome (PWS) and to examine the modulating effect on bone of treatment with growth hormone (GH) and sex steroids. This was a cross-sectional study performed in 41 (17 males, 24 females) obese subjects with genetically confirmed PWS, aged 29.4 ± 8.6 years. Forty-six healthy subjects (22 males and 24 females) served as controls. Digitalized X-rays were evaluated at the level of the 2nd metacarpal bone to assess bone geometry, i.e. cross-sectional area (CSA), cortical area (CA), medullary area (MA), metacarpal index (MI) and bone strength evaluated as bending breaking resistance index (BBRI). DEXA was also used to evaluate body composition and bone mineral density (total body, lumbar spine and femoral neck). PWS subjects, after adjusting for height and bone size, had a reduced CSA, CA and BBRI, while bone density was not different. GH treatment had a positive effect and sex steroids a negative effect on bone size and strength. PWS subjects showed a reduced bone size at the metacarpus leading to a reduced strength, while bone density was appropriate for size. GH treatment improves bone geometry but not bone density. Bone strength was significantly reduced in PWS patients who did not receive GH and had been treated with sex steroids.
Similar content being viewed by others
References
Bakker I, Twisk JW, Van Mechelen W, Kemper HC (2003) Fat-free body mass is the most important body composition determinant of 10-yr longitudinal development of lumbar bone in adult men and women. J Clin Endocrinol Metab 88:2607–2613
Taes YE, Lapauw B, Vanbillemont G et al (2009) Fat mass is negatively associated with cortical bone size in young healthy male siblings. J Clin Endocrinol Metab 94:2325–2331
Hsu YH, Venners SA, Terwedow HA et al (2006) Relation of body composition, fat mass, and serum lipids to osteoporotic fractures and bone mineral density in Chinese men and women. Am J Clin Nutr 83:146–154
Rocher E, Chappard C, Jaffre C, Benhamou CL, Courteix D (2008) Bone mineral density in prepubertal obese and control children: relation to body weight, lean mass, and fat mass. J Bone Miner Metab 26:73–78
Ng AC, Melton LJ 3rd, Atkinson EJ et al (2013) Relationship of adiposity to bone volumetric density and microstructure in men and women across the adult lifespan. Bone 55:119–125
Longhi S, Pasquino B, Calcagno A et al (2013) Small metacarpal bones of low quality in obese children. Clin Endocrinol (Oxf) 78:79–85
Butler MG (2011) rader-Willi syndrome: obesity due to genomic imprinting. Curr Genomics 12:204–215
Goldstone AP (2004) Prader-Willi syndrome: advances in genetics, pathophysiology and treatment. Trends Endocrinol Metab 15:12–20
Cassidy SB, Schwartz S, Miller JL, Driscoll DJ (2012) Prader-Willi syndrome. Genet Med 14:10–26
Theodoro MF, Talebizadeh Z, Butler MG (2006) Body composition and fatness patterns in Prader- Willi syndrome: comparison with simple obesity. Obesity 14:1685–1690
Kennedy L, Bittel DC, Kibiryeva N, Kalra SP, Torto R, Butler MG (2006) Circulating adiponectin levels, body composition, and obesity-related variables in Prader-Willi syndrome: comparison with obese subjects. Int J Obes 30:382–387
Goldstone AP, Thomas EL, Brynes AE et al (2001) isceral adipose tissue and metabolic complications of obesity are reduced in Prader-Willi syndrome female adults: evidence for novel influences on body fat distribution. J Clin Endocrinol Metab 86:4330–4338
Talebizadeh Z, Butler M (2005) Insulin resistance and obesity-related factors in Prader-Willi syndrome: comparison with obese subjects. Clin Genet 67:230–239
Van Mil EG, Westerterp KR, Gerver WJ, Van Marken Lichtenbelt WD, Kester AD, Saris WH (2001) Body composition in Prader-Willi syndrome compared with nonsyndromal obesity: relationship to physical activity and growth hormone function. J Pediatr 139:708–714
Reus L, van Vlimmeren LA, Staal JB, Otten BJ, Nijhuis-van der Sanden MW (2012) The effect of growth hormone treatment or physical training on motor performance in Prader-Willi syndrome: A systematic review. Neurosci Biobehav Rev 36:1817–1838
Butler MG, Haber L, Mernaugh R, Carlson MG, Price R, Feurer ID (2001) Decreased bone mineral density in Prader-Willi syndrome: comparison with obese subjects. Am J Med Genet 103:216–222
Vestergaard P, Kristensen K, Bruun JM et al (2004) Reduced bone mineral density and increased bone turnover in Prader-Willi syndrome compared with controls matched for sex and body mass index—a cross-sectional study. J Pediatr 144:614–619
Carrel AL, Myers SE, Whitman BY, Eickhoff J, Allen DB (2010) Long-term growth hormone therapy changes the natural history of body composition and motor function in children with Prader-Willi syndrome. J Clin Endocrinol Metab 95:1131–1136
Deal CL, Tony M, Höybye C, Allen DB, Tauber M, Christiansen JS (2013) Growth Hormone Research Society workshop summary: consensus guidelines for recombinant human growth hormone therapy in Prader-Willi syndrome. J Clin Endocrinol Metab 98:E1072–E1087
Kroonen LT, Herman M, Pizzutillo PD, Macewen GD (2006) Prader-Willi Syndrome: clinical concerns for the orthopaedic surgeon. J Pediatr Orthop 26:673–679
Sinnema M, Maaskant MA, van Schrojenstein Lantman-de Valk HM et al (2011) Physical health problems in adults with Prader-Willi syndrome. Am J Med Genet A 155:2112–2124
Carrel AL, Myers SE, Whitman BY, Allen DB (2002) Benefits of long-term GH therapy in Prader-Willi syndrome: a 4-year study. J Clin Endocrinol Metab 87:1581–1585
Carrel AL, Myers SE, Whitman BY, Eickhoff J, Allen DB (2010) Long-term growth hormone therapy changes the natural history of body composition and motor function in children with Prader-Willi syndrome. J Clin Endocrinol Metab 95:1131–1136
Zamberlan N, Radetti G, Paganini C et al (1996) Evaluation of cortical thickness and bone density by roentgen microdensitometry in growing males and females. Eur J Pediatr 155:377–382
Turner CH, Burr DB (1993) Basic biomechanical measurements of bone: a tutorial. Bone 14:595–608
Butler JV, Whittington JE, Holland AJ, Boer H, Clarke D, Webb T (2002) Prevalence of, and risk factors for, physical ill-health in people with Prader-Willi syndrome: a population-based study. Dev Med Child Neurol 44:248–255
Mackie EJ, Tatarczuch L, Mirams M (2011) The growth plate chondrocyte and endochondral ossification. J Endocrinol 211:109–121
Ohlsson C, Mohan S, Sjogren K et al (2009) The role of liver-derived insulin-like growth factor-I. Endocr Rev 30:494–535
Olson LE, Ohlsson C, Mohan S (2011) The role of GH/IGF-I-mediated mechanisms in sex differences in cortical bone size in mice. Calcif Tissue Int 88:1–8
Walenkamp MJ, Wit JM (2007) Genetic disorders in the GH IGF-I axis in mouse and man. Eur J Endocrinol 157:S15–S26
Yakar S, Courtland HW, Clemmons D (2010) IGF-1 and bone: new discoveries from mouse models. J Bone Miner Res 25:2543–2552
Mohan S, Baylink DJ (1999) Role of growth hormone/insulin-like growth factor axis. In: Glowacki J, Rosen CJ, Bilezikian JP (eds) The aging skeleton. Academic, San Diego, pp 209–219
Ohlsson C, Bengtsson BA, Isaksson OG, Andreassen TT, Slootweg MC (1998) Growth hormone and bone. Endocr Rev 19:55–79
Boonchaya-Anant P, Holick MF, Apovian CM (2014) Serum 25-hydroxyvitamin D levels and metabolic health status in extremely obese individuals. Obesity (Silver Spring) 22:2539–2543
Högler W, Shaw N (2010) Childhood growth hormone deficiency, bone density, structures and fractures: scrutinizing the evidence. Clin Endocrinol 72:281–289
Jørgensen AP, Ueland T, Sode-Carlsen R et al (2013) Two years of growth hormone treatment in adults with Prader-Willi syndrome do not improve the low BMD. J Clin Endocrinol Metab 98:E753–E760
Acknowledgments
We are grateful to the patients with PWS and their families for their willingness to participate in this research. We are indebted with Dr Lan Nguyen for editing the english version of the manuscript.
Conflict of interest
Dr. Longhi, Dr. Grugni, Dr. Gatti, Dr. Spinozzi, Dr. Sartorio, Dr. Adami, Dr. Fanolla, and Dr. Radetti have no conflict of interest.
Human and Animal Rights and Informed Consent
Informed consent was obtained from the subjects, under parental guidance for PWS patients and the investigations had been approved by an institutional human research committee (Ethical Committee of Istituto Auxologico Italiano).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Longhi, S., Grugni, G., Gatti, D. et al. Adults with Prader–Willi Syndrome have Weaker Bones: Effect of Treatment with GH and Sex Steroids. Calcif Tissue Int 96, 160–166 (2015). https://doi.org/10.1007/s00223-014-9949-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-014-9949-1