Abstract
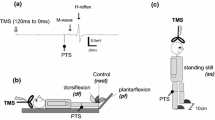
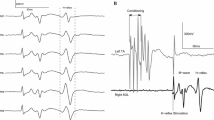
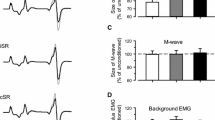
The soleus H-reflex modulation pattern was investigated during stepping following transspinal stimulation over the thoracolumbar region at 15, 30, and 50 Hz with 10 kHz carry-over frequency above and below the paresthesia threshold. The soleus H-reflex was elicited by posterior tibial nerve stimulation with a single 1 ms pulse at an intensity that the M-wave amplitudes ranged from 0 to 15% of the maximal M-wave evoked 80 ms after the test stimulus, and the soleus H-reflex was half the size of the maximal H-reflex evoked on the ascending portion of the recruitment curve. During treadmill walking, the soleus H-reflex was elicited every 2 or 3 steps, and stimuli were randomly dispersed across the step cycle which was divided in 16 equal bins. For each subject and condition, the soleus M-wave and H-reflex were normalized to the maximal M-wave. The soleus background electromyographic (EMG) activity was estimated as the linear envelope for 50 ms duration starting at 100 ms before posterior tibial nerve stimulation for each bin. The gain was determined as the slope of the relationship between the soleus H-reflex and the soleus background EMG activity. The soleus H-reflex phase-dependent amplitude modulation remained unaltered during transspinal stimulation, regardless frequency, or intensity. Similarly, the H-reflex slope and intercept remained the same for all transspinal stimulation conditions tested. Locomotor EMG activity was increased in knee extensor muscles during transspinal stimulation at 30 and 50 Hz throughout the step cycle while no effects were observed in flexor muscles. These findings suggest that transspinal stimulation above and below the paresthesia threshold at 15, 30, and 50 Hz does not block or impair spinal integration of proprioceptive inputs and increases activity of thigh muscles that affect both hip and knee joint movement. Transspinal stimulation may serve as a neurorecovery strategy to augment standing or walking ability in upper motoneuron lesions.





Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Change history
15 March 2024
In this article, the family name of the author Abdullah M. Sayed Ahmad was incorrect. This has been corrected.
References
Angeli CA, Edgerton VR, Gerasimenko YP, Harkema SJ (2014) Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 137(Pt 5):1394–1409. https://doi.org/10.1093/brain/awu038
Capaday C, Stein RB (1986) Amplitude modulation of the soleus H-reflex in the human during walking and standing. J Neurosci 6(5):1308–1313. https://doi.org/10.1523/JNEUROSCI.06-05-01308.1986
Capaday C, Stein RB (1987) Differences in the amplitude of the human soleus H reflex during walking and running. J Physiol 392(1):513–522. https://doi.org/10.1113/jphysiol.1987.sp016794
Courtine G, Harkema SJ, Dy CJ, Gerasimenko YP, Dyhre-Poulsen P (2007) Modulation of multisegmental monosynaptic responses in a variety of leg muscles during walking and running in humans. J Physiol 582(3):1125–1139. https://doi.org/10.1113/jphysiol.2007.128447
Dimitrijevic MR, Gerasimenko Y, Pinter MM (1998) Evidence of spinal central pattern generator in humans. Ann N Y Acad Sci 860:360–376. https://doi.org/10.1111/j.1749-6632.1998.tb09062.x
Dy CJ, Gerasimenko YP, Edgerton VR, Dyhre-Poulsen P, Courtine G, Harkema SJ (2010) Phase-dependent modulation of percutaneously elicited multisegmental muscle responses after spinal cord injury. J Neurophysiol 130(5):2808–2820. https://doi.org/10.1152/jn.00316.2009
Formento E, Minassian K, Wagner F, Mignardot JB, Le Goff-Mignardot CG, Rowald A, Bloch J, Micera S, Capogrosso M, Courtine G (2018) Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat Neurosci 21(12):1729–1741. https://doi.org/10.1038/s41593-018-0262-6
Gorodnichev RM, Pivovarova EA, Puhov A, Moiseev SA, Savochin AA, Moshonkina TR, Chsherbakova NA, Kilimnik VA, Selionov VA, Kozlovskaya IB, Edgerton VR, Gerasimenko YP (2012) Transcutaneous electrical stimulation of the spinal cord: a noninvasive tool for the activation of stepping pattern generators in humans. Hum Physiol 38(2):158–167. https://doi.org/10.1134/S0362119712020065
Harkema S, Gerasimenko Y, Hodes J, Burdick J, Angeli C, Chen Y, Ferreira C, Willhite A, Rejc E, Grossman RG, Edgerton VR (2011) Effect of epidural stimulation of the lumbrosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. The Lancet 377(9781):1938–1947. https://doi.org/10.1016/S0140-6736(11)60547-3
Hofstoetter US, Minassian K, Hofer C, Mayr W, Rattay F, Dimitrijevic MR (2008) Modification of reflex responses to lumbar posterior root stimulation by motor tasks in healthy subjects. Artif Organs 32(8):644–648. https://doi.org/10.1111/j.1525-1594.2008.00616.x
Hofstoetter US, McKay WB, Tansey KE, Mayr W, Kern H, Minassian K (2014) Modification of spasticity by transcutaneous spinal cord stimulation in individuals with incomplete spinal cord injury. J Spinal Cord Med 37(2):202–211. https://doi.org/10.1179/2045772313Y.0000000149
Hofstoetter US, Freundl B, Binder H, Minassian K (2018) Common neural structures activated by epidural and transcutaneous lumbar spinal cord stimulation: elicitation of posterior root-muscle reflexes. PLoS ONE 13(1):e0192013. https://doi.org/10.1371/journal.pone.0192013
Islam MA, Pulverenti TS, Knikou M (2021) Neuronal actions of transspinal stimulation on locomotor networks and reflex excitability during walking in humans with and without spinal cord injury. Front Hum Neurosci 15:620414. https://doi.org/10.3389/fnhum.2021.620414
Knikou M (2008) The H-reflex as a probe: pathways and pitfalls. J Neurosci Methods 171(1):1–12. https://doi.org/10.1016/j.jneumeth.2008.02.012
Knikou M (2012) Function of group IB inhibition during assisted stepping in human spinal cord injury. J Clin Neurophysiol 29(3):271–277. https://doi.org/10.1097/WNP.0b013e318257c2b7
Knikou M (2013) Neurophysiological characterization of transpinal evoked potentials in human leg muscles. Bioelectromagnetics 34(8):630–640. https://doi.org/10.1002/bem.21808
Knikou M (2014) Transpinal and transcortical stimulation alter corticospinal excitability and increase spinal output. PLoS ONE 9(7):e102313. https://doi.org/10.1371/journal.pone.0102313
Knikou M, Mummidisetty CK (2011) Reduced reciprocal inhibition during assisted stepping in human spinal cord injury. Exp Neurol 231(1):104–112. https://doi.org/10.1016/j.expneurol.2011.05.021
Knikou M, Mummidisetty CK (2014) Locomotor training improves premotoneuronal control after chronic spinal cord injury. J Neurophysiol 111(11):2264–2275. https://doi.org/10.1152/jn.00871.2013
Knikou M, Murray LM (2018) Neural interactions between transspinal evoked potentials and muscle spindle afferents in humans. J Electromyogr Kinesiol 43:174–183. https://doi.org/10.1016/j.jelekin.2018.10.005
Knikou M, Murray LM (2019) Repeated transspinal stimulation decreases soleus H-reflex excitability and restores spinal inhibition in human spinal cord injury. PLoS ONE 14(9):e0223135. https://doi.org/10.1371/journal.pone.0223135
Knikou M, Angeli CA, Ferreira CK, Harkema SJ (2009a) Soleus H-reflex modulation during body weight support treadmill walking in spinal cord intact and injured subjects. Exp Brain Res 193(3):397–407. https://doi.org/10.1007/s00221-008-1636-x
Knikou M, Angeli CA, Ferreira CK, Harkema SJ (2009b) Soleus H-reflex gain, threshold, and amplitude as function of body posture and load in spinal cord intact and injured subjects. Int J Neurosci 119(11):2056–2073. https://doi.org/10.1080/00207450903139747
Knikou M, Hajela N, Mummidisetty CK, Xiao M, Smith AC (2011) Soleus H-reflex phase-dependent modulation is preserved during stepping within a robotic exoskeleton. Clin Neurophysiol 122(7):1396–1404. https://doi.org/10.1016/j.clinph.2010.12.044
Knikou M, Dixon L, Santora D, Ibrahim MM (2015) Transspinal constant-current long-lasting stimulation: a new method to induce cortical and corticospinal plasticity. J Neurophysiol 114(3):1486–1499. https://doi.org/10.1152/jn.00449.2015
Llewellyn M, Yang JF, Prochazka A (1990) Human H-reflexes are smaller in difficult beam walking than in normal treadmill walking. Exp Brain Res 83:22–28. https://doi.org/10.1007/BF00232189
Marchand-Pauvert V, Nicolas G, Burke D, Pierrot-Deseilligny E (2002) Suppression of the H reflex in humans by disynaptic autogenetic inhibitory pathways activated by the test volley. J Physiol 542(Pt 3):963–976. https://doi.org/10.1113/jphysiol.2002.021683
Menard A, Leblond H, Gossard JP (2002) Sensory integration in presynaptic inhibitory pathways during fictive locomotion in the cat. J Neurophysiol 88(1):163–171. https://doi.org/10.1152/jn.2002.88.1.163
Minassian K, Jilge B, Rattay F, Pinter MM, Binder H, Gerstenbrand F, Dimitrijevic MR (2004) Stepping-like movements in humans with complete spinal cord injury induced by epidural stimulation of the lumbar cord: electromyographic study of compound muscle action potentials. Spinal Cord 42(7):401–416. https://doi.org/10.1038/sj.sc.3101615
Minassian K, Hofstoetter US, Danner SM, Mayr W, Bruce JA, McKay WB, Tansey KE (2016) Spinal rhythm generation by step-induced feedback and transcutaneous posterior root stimulation in complete spinal cord-injured individuals. Neurorehabil Neural Repair 30:233–243. https://doi.org/10.1177/1545968315591706
Moraud EM, Capogrosso M, Formento E, Wenger N, DiGiovanna J, Courtine G, Micera S (2016) Mechanisms underlying neuromodulation of spinal circuits for correcting gait and balance deficits after spinal cord injury. Neuron 89(4):814–828. https://doi.org/10.1016/j.neuron.2016.01.009
Mummidisetty CK, Smith AC, Knikou M (2013) Modulation of reciprocal and presynaptic inhibition during robotic-assisted stepping in humans. Clin Neurophysiol 124(3):557–564. https://doi.org/10.1016/j.clinph.2012.09.007
Pulverenti TS, Islam MA, Alsalman O, Murray LM, Harel NY, Knikou M (2019) Transspinal stimulation decreases corticospinal excitability and alters the function of spinal locomotor networks. J Neurophysiol 122(6):2331–2343. https://doi.org/10.1152/jn.00554.2019
Pulverenti TS, Zaaya M, Grabowski E, Grabowski M, Knikou M (2022) Brain and spinal cord paired stimulation coupled with locomotor training facilitates motor output in human spinal cord injury. Front Neurol 13:1000940. https://doi.org/10.3389/fneur.2022.1000940
Rath M, Vette AH, Ramasubramaniam S, Li K, Burdick J, Edgerton VR, Gerasimenko YP, Sayenko DG (2018) Trunk stability enabled by noninvasive spinal electrical stimulation after spinal cord injury. J Neurotrauma 35:2540–2553. https://doi.org/10.1089/neu.2017.5584
Rattay F, Minassian K, Dimitrijevic MR (2000) Epidural stimulation of posterior structures of the human lumbrosacral cord: 2. quantitative analysis by computer modeling. Spinal Cord 38(8):473–489. https://doi.org/10.1038/sj.sc.3101039
Rudomin P (2009) In search of lost presynaptic inhibition. Exp Brain Res 196(1):139–151. https://doi.org/10.1007/s00221-009-1758-9
Sayenko D, Rath M, Ferguson AR, Burdick J, Havton L, Edgerton VR, Gerasimenko Y (2019) Self-assisted standing enabled by non-invasive spinal stimulation after spinal cord injury. J Neurotrauma 36(9):1435–1450. https://doi.org/10.1089/neu.2018.5956
Shapkova EY, Schomburg ED (2001) Two types of motor modulation underlying human stepping evoked by spinal cord electrical stimulation (SCES). Acta Physiol Pharacol Bulg 26(3):155–157
Skiadopoulos A, Pulverenti TS, Knikou M (2022) Physiological effects of cathodal electrode configuration for transspinal stimulation in humans. J Neurophysiol 128(6):1663–1682. https://doi.org/10.1152/jn.00342.2022
Yang JF, Stein RB, James KB (1991) Contribution of peripheral afferents to the activation of the soleus muscle during walking in humans. Exp Brain Res 87(3):679–687. https://doi.org/10.1007/BF00227094
Zaaya M, Pulverenti TS, Islam MA, Knikou M (2020) Transspinal stimulation downregulates activity of flexor locomotor networks during walking in humans. J Electromyogr Kinesiol 52:102420. https://doi.org/10.1016/j.jelekin.2020.102420
Zaaya M, Pulverenti TS, Knikou M (2021) Transspinal stimulation and step training alter function of spinal networks in complete spinal cord injury. Spinal Cord Ser Cases 7(1):55. https://doi.org/10.1038/s41394-021-00421-6
Zehr E, Stein RB (1999) What functions do reflexes serve during human locomotion? Prog Neurobiol 58(2):185–205. https://doi.org/10.1016/s0301-0082(98)00081-1
Funding
This work was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) of the National Institutes of Health (NIH) under Grant Number R01HD100544 and by the New York State Department of Health (NYSDOH), Spinal Cord Injury Research Board (SCIRB) under Contracts C35594GG and C38333GG awarded to Maria Knikou. The funding sources were not involved in study design, data collection, data analysis, data interpretation, or decision to publish.
Author information
Authors and Affiliations
Contributions
AMS: performed experiments, analyzed data, and approved the final version of the manuscript.
MR, YA, MM, JF, SKS, AS: performed experiments, and approved the final version of the manuscript.
MK: conception and design of research, performed experiments, analyzed data, prepared figures, interpreted results of experiments, wrote the first draft of the manuscript, approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics statement
The studies involving human participants were reviewed and approved by the Institutional Review Board of the City University of New York. All participants provided their written informed consent to participate in this study.
Additional information
Communicated by Bill J Yates.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sayed Ahmad, A.M., Raphael, M., Han, J.F. et al. Soleus H-reflex amplitude modulation during walking remains physiological during transspinal stimulation in humans. Exp Brain Res (2024). https://doi.org/10.1007/s00221-024-06779-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00221-024-06779-x