Abstract
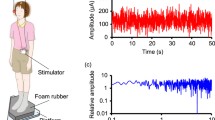
Vestibular nuclei and cerebellar function comprise vestibular neural networks that control vestibular-related responses. However, the vestibular-related responses to simultaneous stimulation of these regions are unclear. This study aimed to examine whether the combination of noisy galvanic vestibular stimulation (nGVS) and cerebellar transcranial direct current stimulation (ctDCS) using a complex transcranial electrical stimulation device alters vestibular-dominant standing stability and vestibulo-ocular reflex (VOR) function. The center of foot pressure (COP) sway and VOR of participants (28 healthy, young adults) were assessed under four conditions of transcranial electrical stimulation using nGVS and ctDCS. The COP was calculated with the participant standing on a soft-foam surface with eyes closed using a force plate to evaluate body sway. VOR measurements were collected via passive head movements and fixation on a target projected onto the front wall using a video head impulse test (vHIT). VOR gain was calculated in six directions using a semicircular canal structure based on the ratio of eye movement to head movement. The nGVS + ctDCS and nGVS + sham ctDCS conditions decreased COP sway compared to the sham nGVS + ctDCS and sham nGVS + sham ctDCS conditions. No significant differences were observed in the main effect of stimulation or the interaction of stimulation and direction on the vHIT parameters. The results of this study suggest that postural stability may be independently affected by nGVS. Our findings contribute to the basic neurological foundation for the clinical application of neurorehabilitation using transcranial electrical stimulation of the vestibular system.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Agrawal Y, Schubert MC, Migliaccio AA, Zee DS, Schneider E, Lehnen N, Carey JP (2014) Evaluation of quantitative head impulse testing using search coils versus video-oculography in older individuals. Otol Neurotol 35:283–288. https://doi.org/10.1097/MAO.0b013e3182995227
Chen F, Chen Z, Zhang Y, Wei X, Zhao H, Hu J, Cheng Y, Ren X, Zhang Q (2021) Association analysis of HIMP and SHIMP quantitative parameters in patients with vestibular neuritis and healthy participants. Front Neurol 12:748990. https://doi.org/10.3389/fneur.2021.748990
Dakin CJ, Son GM, Inglis JT, Blouin JS (2007) Frequency response of human vestibular reflexes characterized by stochastic stimuli. J Physiol 583:1117–1127. https://doi.org/10.1113/jphysiol.2007.133264
Dilda V, Morris TR, Yungher DA, MacDougall HG, Moore ST (2014) Central adaptation to repeated galvanic vestibular stimulation: implications for pre-flight astronaut training. PLoS ONE 9:e112131. https://doi.org/10.1371/journal.pone.0112131
Dlugaiczyk J, Gensberger KD, Straka H (2019) Galvanic vestibular stimulation: from basic concepts to clinical applications. J Neurophysiol 121:2237–2255. https://doi.org/10.1152/jn.00035.2019
Fujimoto C, Yamamoto Y, Kamogashira T, Kinoshita M, Egami N, Uemura Y, Togo F, Yamasoba T, Iwasaki S (2016) Noisy galvanic vestibular stimulation induces a sustained improvement in body balance in elderly adults. Sci Rep 6:37575. https://doi.org/10.1038/srep37575
Fujimoto C, Kinoshita M, Kamogashira T, Egami N, Kawahara T, Uemura Y, Yamamoto Y, Yamasoba T, Iwasaki S (2019) Noisy galvanic vestibular stimulation has a greater ameliorating effect on posture in unstable subjects: a feasibility study. Sci Rep 9:17189. https://doi.org/10.1038/s41598-019-53834-7
Galea JM, Jayaram G, Ajagbe L, Celnik P (2009) Modulation of cerebellar excitability by polarity-specific noninvasive direct current stimulation. J Neurosci 29:9115–9122. https://doi.org/10.1523/JNEUROSCI.2184-09.2009
Gomez-Tames J, Asai A, Mikkonen M, Laakso I, Tanaka S, Uehara S, Otaka Y, Hirata A (2019) Group-level and functional-region analysis of electric-field shape during cerebellar transcranial direct current stimulation with different electrode montages. J Neural Eng 16:036001. https://doi.org/10.1088/1741-2552/ab0ac5
Hag L, Abrahamsen ER, Hougaard DD (2020) Normative vestibular-ocular reflex gain values for the vertical semicircular canals. Res Vestib Sci 19:62–70. https://doi.org/10.21790/rvs.2020.19.2.62
Halmagyi GM, Chen L, MacDougall HG, Weber KP, McGarvie LA, Curthoys IS (2017) The video head impulse test. Front Neurol 8:258. https://doi.org/10.3389/fneur.2017.00258
Herzog R, Berger TM, Pauly MG, Xue H, Rueckert E, Münchau A, Bäumer T, Weissbach A (2022) Cerebellar transcranial current stimulation—an intraindividual comparison of different techniques. Front Neurosci 16:987472. https://doi.org/10.3389/fnins.2022.987472
Inukai Y, Saito K, Sasaki R, Kotan S, Nakagawa M, Onishi H (2016) Influence of transcranial direct current stimulation to the cerebellum on standing posture control. Front Hum Neurosci 10:325. https://doi.org/10.3389/fnhum.2016.00325
Inukai Y, Otsuru N, Masaki M, Saito K, Miyaguchi S, Kojima S, Onishi H (2018) Effect of noisy galvanic vestibular stimulation on center of pressure sway of static standing posture. Brain Stimul 11:85–93. https://doi.org/10.1016/j.brs.2017.10.007
Inukai Y, Miyaguchi S, Saito M, Otsuru N, Onishi H (2020) Effects of different stimulation conditions on the stimulation effect of noisy galvanic vestibular stimulation. Front Hum Neurosci 14:581405. https://doi.org/10.3389/fnhum.2020.581405
Iwasaki S, Yamamoto Y, Togo F, Kinoshita M, Yoshifuji Y, Fujimoto C, Yamasoba T (2014) Noisy vestibular stimulation improves body balance in bilateral vestibulopathy. Neurology 82:969–975. https://doi.org/10.1212/WNL.0000000000000215
Kandel E, Schwartz J, Jessell T, Siegelbaum S, Hudspeth AJ (2012) The vestibular system. In: Kandel E, Schwartz J, Jessell T, Siegelbaum S, Hudspeth AJ (eds) Principles of neural science, 5th edn. McGraw-Hill, New York, pp 917–934
Koganemaru S, Mikami Y, Matsuhashi M, Truong DQ, Bikson M, Kansaku K, Mima T (2020) Cerebellar transcranial alternating current stimulation modulates human gait rhythm. Neurosci Res 156:265–270. https://doi.org/10.1016/j.neures.2019.12.003
Kumari N, Taylor D, Signal N (2019) The effect of cerebellar transcranial direct current stimulation on motor learning: a systematic review of randomized controlled trials. Front Hum Neurosci 13:328. https://doi.org/10.3389/fnhum.2019.00328
Laakso I, Tanaka S, Koyama S, De Santis V, Hirata A (2015) Inter-subject variability in electric fields of motor cortical tDCS. Brain Stimul 8:906–913. https://doi.org/10.1016/j.brs.2015.05.002
Lam CK, Tokuno CD, Staines WR, Bent LR (2016) The direction of the postural response to a vestibular perturbation is mediated by the cerebellar vermis. Exp Brain Res 234:3689–3697. https://doi.org/10.1007/s00221-016-4766-6
Lam CK, Staines WR, Tokuno CD, Bent LR (2017) The medium latency muscle response to a vestibular perturbation is increased after depression of the cerebellar vermis. Brain Behav 7:e00782. https://doi.org/10.1002/brb3.782
Lee S, Smith PF, Lee WH, McKeown MJ (2021) Frequency-specific effects of galvanic vestibular stimulation on response-time performance in Parkinson's disease. Front Neurol 12:758122. https://doi.org/10.3389/fneur.2021.758122
Lee JY, Kim MB (2020) Importance of video head impulse test parameters for recovery of symptoms in acute vestibular neuritis. Otol Neurotol 41:964–971. https://doi.org/10.1097/MAO.0000000000002669
MacDougall HG, McGarvie LA, Halmagyi GM, Rogers SJ, Manzari L, Burgess AM, Curthoys IS, Weber KP (2016) A new saccadic indicator of peripheral vestibular function based on the video head impulse test. Neurology 87:410–418. https://doi.org/10.1212/WNL.0000000000002827
Mannarelli D, Pauletti C, Petritis A, Delle Chiaie R, Currà A, Trompetto C, Fattapposta F (2020) Effects of cerebellar tDCS on inhibitory control: evidence from a Go/NoGo task. Cerebellum 19:788–798. https://doi.org/10.1007/s12311-020-01165-z
Matsugi A, Douchi S, Hasada R, Mori N, Okada Y, Yoshida N, Nishishita S, Hosomi K, Saitoh Y (2020a) Cerebellar repetitive transcranial magnetic stimulation and noisy galvanic vestibular stimulation change vestibulospinal function. Front Neurosci 14:388. https://doi.org/10.3389/fnins.2020.00388
Matsugi A, Oku K, Mori N (2020b) The effects of stochastic galvanic vestibular stimulation on body sway and muscle activity. Front Hum Neurosci 14:591671. https://doi.org/10.3389/fnhum.2020.591671
Matsugi A, Shiozaki T, Tanaka H (2022) Vestibulo-ocular reflex is modulated by noisy galvanic vestibular stimulation. Front Neurol 13:826739. https://doi.org/10.3389/fneur.2022.826739
McCall AA, Miller DM, Yates BJ (2017) Descending influences on vestibulospinal and vestibulosympathetic reflexes. Front Neurol 8:112. https://doi.org/10.3389/fneur.2017.00112
McDonnell MD, Ward LM (2011) The benefits of noise in neural systems: bridging theory and experiment. Nat Rev Neurosci 12:415–426. https://doi.org/10.1038/nrn3061
McGarvie LA, MacDougall HG, Halmagyi GM, Burgess AM, Weber KP, Curthoys IS (2015) The video head impulse test (vHIT) of semicircular canal function—age-dependent normative values of VOR gain in healthy subjects. Front Neurol 6:154. https://doi.org/10.3389/fneur.2015.00154
McLaren R, Smith PF, Taylor RL, Niazi IK, Taylor D (2023) Scoping out noisy galvanic vestibular stimulation: a review of the parameters used to improve postural control. Front Neurosci 17:1156796. https://doi.org/10.3389/fnins.2023.1156796
Mitsutake T, Sakamoto M, Kawaguchi A, Tamari M, Horikawa E (2020) Greater functional activation during galvanic vestibular stimulation is associated with improved postural stability: a GVS-fMRI study. Somatosens Mot Res 37:257–261. https://doi.org/10.1080/08990220.2020.1803256
Mitsutake T, Imura T, Hori T, Sakamoto M, Tanaka R (2021a) Effects of combining online anodal transcranial direct current stimulation and gait training in stroke patients: a systematic review and meta-analysis. Front Hum Neurosci 15:782305. https://doi.org/10.3389/fnhum.2021.782305
Mitsutake T, Sakamoto M, Horikawa E (2021b) Comparing activated brain regions between noisy and conventional galvanic vestibular stimulation using functional magnetic resonance imaging. NeuroReport 32:583–587. https://doi.org/10.1097/WNR.0000000000001629
Mitsutake T, Taniguchi T, Nakazono H, Yoshizuka H, Sakamoto M (2022) Effects of noisy galvanic vestibular stimulation on the muscle activity and joint movements in different standing postures conditions. Front Hum Neurosci 16:891669. https://doi.org/10.3389/fnhum.2022.891669
Moss F, Ward LM, Sannita WG (2004) Stochastic resonance and sensory information processing: a tutorial and review of application. Clin Neurophysiol 115:267–281. https://doi.org/10.1016/j.clinph.2003.09.014
Mulavara AP, Fiedler MJ, Kofman IS, Wood SJ, Serrador JM, Peters B, Cohen HS, Reschke MF, Bloomberg JJ (2011) Improving balance function using vestibular stochastic resonance: optimizing stimulus characteristics. Exp Brain Res 210:303–312. https://doi.org/10.1007/s00221-011-2633-z
Mulavara AP, Kofman IS, De Dios YE, Miller C, Peters BT, Goel R, Galvan-Garza R, Bloomberg JJ (2015) Using low levels of stochastic vestibular stimulation to improve locomotor stability. Front Syst Neurosci 9:117. https://doi.org/10.3389/fnsys.2015.00117
Nakamura J, Okada Y, Shiozaki T, Tanaka H, Ueta K, Ikuno K, Morioka S, Shomoto K (2021) Reliability and laterality of the soleus H-reflex following galvanic vestibular stimulation in healthy individuals. Neurosci Lett 755:135910. https://doi.org/10.1016/j.neulet.2021.135910
Nakazono H, Taniguchi T, Mitsutake T, Takeda A, Yamada E, Ogata K (2022) Phase-dependent modulation of the vestibular–cerebellar network via combined alternating current stimulation influences human locomotion and posture. Front Neurosci 16:1057021. https://doi.org/10.3389/fnins.2022.1057021
Nitsche MA, Paulus W (2000) Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol 527:633–639. https://doi.org/10.1111/j.1469-7793.2000.t01-1-00633.x
Patterson J, Rodriguez A, Barin K, Janky KL (2020) Effect of gaze angle during the vertical video head impulse test across two devices in healthy adults and subjects with vestibular loss. Otol Neurotol 41:e751–e758. https://doi.org/10.1097/MAO.0000000000002652
Potok W, Bächinger M, van der Groen O, Cretu AL, Wenderoth N (2021) Transcranial random noise stimulation acutely lowers the response threshold of human motor circuits. J Neurosci 41:3842–3853. https://doi.org/10.1523/JNEUROSCI.2961-20.2021
Roche N, Geiger M, Bussel B (2015) Mechanisms underlying transcranial direct current stimulation in rehabilitation. Ann Phys Rehabil Med 58:214–219. https://doi.org/10.1016/j.rehab.2015.04.009
Schniepp R, Boerner JC, Decker J, Jahn K, Brandt T, Wuehr M (2018) Noisy vestibular stimulation improves vestibulospinal function in patients with bilateral vestibulopathy. J Neurol 265(Supplement 1):57–62. https://doi.org/10.1007/s00415-018-8814-y
Shugyo M, Ito T, Shiozaki T, Nishikawa D, Ohyama H, Fujita H, Yamanaka T, Kitahara T (2020) Comparison of the video head impulse test results with caloric test in patients with Meniere’s disease and other vestibular disorders. Acta Otolaryngol 140:728–735. https://doi.org/10.1080/00016489.2020.1766700
Sozzi S, Ghai S, Schieppati M (2023) The “postural rhythm” of the ground reaction force during upright stance and its conversion to body sway-the effect of vision, support surface and adaptation to repeated trials. Brain Sci 13:978. https://doi.org/10.3390/brainsci13070978
Starkov D, Vermorken B, Van Dooren TS, Van Stiphout L, Janssen M, Pleshkov M, Guinand N, Pérez Fornos A, Van Rompaey V, Kingma H, Van de Berg R (2021) The effect of different head movement paradigms on vestibulo-ocular reflex gain and saccadic eye responses in the suppression head impulse test in healthy adult volunteers. Front Neurol 12:729081. https://doi.org/10.3389/fneur.2021.729081
Straka H, Zwergal A, Cullen KE (2016) Vestibular animal models: contributions to understanding physiology and disease. J Neurol 263(Supplement 1):S10–S23. https://doi.org/10.1007/s00415-015-7909-y
Takakusaki K (2017) Functional neuroanatomy for posture and gait control. J Mov Disord 10:1–17. https://doi.org/10.14802/jmd.16062
Terney D, Chaieb L, Moliadze V, Antal A, Paulus W (2008) Increasing human brain excitability by transcranial high-frequency random noise stimulation. J Neurosci 28:14147–14155. https://doi.org/10.1523/JNEUROSCI.4248-08.2008
Valdés BA, Lajoie K, Marigold DS, Menon C (2021) Cortical effects of noisy galvanic vestibular stimulation using functional near-infrared spectroscopy. Sensors (basel) 21:1476. https://doi.org/10.3390/s21041476
Wuehr M, Nusser E, Decker J, Krafczyk S, Straube A, Brandt T, Jahn K, Schniepp R (2016) Noisy vestibular stimulation improves dynamic walking stability in bilateral vestibulopathy. Neurology 86:2196–2202. https://doi.org/10.1212/WNL.0000000000002748
Zobeiri OA, Cullen KE (2022) Distinct representations of body and head motion are dynamically encoded by Purkinje cell populations in the macaque cerebellum. eLife. https://doi.org/10.7554/eLife.75018
Acknowledgements
The authors express our gratitude to Genki Sato, Masato Yano, and Naoki Kondo for their continuous support, including assistance with data measurement, in the execution of this study.
Funding
This work was supported by the Takeda Science Foundation, and by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Nos. 20H04059 and 22H03460.
Author information
Authors and Affiliations
Contributions
Conceptualization: TM and HN. Formal analysis and investigation: TM, HN, TS, and DF. Writing: TM, HN, TS, and MS. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest associated with this manuscript.
Ethical approval
This study was approved by the Ethical Review Committee of Fukuoka International University of Health and Welfare (Approval No. 20-fiuhw-011). The study was conducted in accordance with the principles and guidelines of the Declaration of Helsinki.
Consent to participate
All participants provided written informed consent after the nature and purpose of the study were explained to them.
Additional information
Communicated by Melvyn A. Goodale.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mitsutake, T., Nakazono, H., Shiozaki, T. et al. Changes in vestibular-related responses to combined noisy galvanic vestibular stimulation and cerebellar transcranial direct current stimulation. Exp Brain Res 242, 99–108 (2024). https://doi.org/10.1007/s00221-023-06731-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-023-06731-5