Abstract
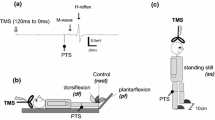
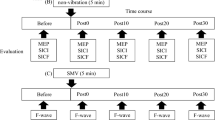
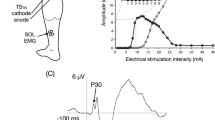
The purpose of this study was to examine corticospinal modulation of spinal reflex excitability, by determining the effect of transcranial magnetic stimulation (TMS) on soleus H-reflexes while they were almost completely suppressed by lower extremity vibration. In 15 healthy adults, a novel method of single-limb vibration (0.6 g, 30 Hz, 0.33 mm displacement) was applied to the non-dominant leg. Soleus muscle responses were examined in six stimulation conditions: (1) H-reflex elicited by tibial nerve stimulation, (2) tibial nerve stimulation during vibration, (3) subthreshold TMS, (4) subthreshold TMS during vibration, (5) tibial nerve stimulation 10 ms after a subthreshold TMS pulse, and (6) tibial nerve stimulation 10 ms after a subthreshold TMS pulse, during vibration. With or without vibration, subthreshold TMS produced no motor evoked potentials and had no effect on soleus electromyography (p > 0.05). In the absence of vibration, H-reflex amplitudes were not affected by subthreshold TMS conditioning (median (md) 35, interquartile range (IQ) 18–56 vs. md 46, IQ 22–59% of the maximal M wave (Mmax), p > 0.05). During vibration, however, unconditioned H-reflexes were nearly abolished, and a TMS conditioning pulse increased the H-reflex more than fourfold (md 0.3, IQ 0.1–0.7 vs. md 2, IQ 0.9–5.0% of Mmax, p < 0.008). Limb vibration alone had no significant effect on corticospinal excitability. In the absence of vibration, a subthreshold TMS pulse did not influence the soleus H-reflex. During limb vibration, however, while the H-reflex was almost completely suppressed, a subthreshold TMS pulse partially restored the H-reflex. This disinhibition of the H-reflex by a corticospinal signal may represent a mechanism involved in the control of voluntary movement. Corticospinal signals that carry the descending motor command may also reduce presynaptic inhibition, temporarily increasing the impact of sensory inputs on motoneuron activation.





Similar content being viewed by others
Availability of data and material
Data are available upon reasonable request.
Code availability
Not applicable.
References
Andrews JC, Sankar T, Stein RB, Roy FD (2020) Characterizing the effect of low intensity transcranial magnetic stimulation on the soleus H-reflex at rest. Exp Brain Res 238:2725–2731. https://doi.org/10.1007/s00221-020-05879-8
Baudry S, Duchateau J (2020) Aftereffects of prolonged Achilles tendon vibration on postural control are reduced in older adults. Exp Gerontol 131:110822. https://doi.org/10.1016/j.exger.2019.110822
Bongiovanni LG, Hagbarth KE, Stjernberg L (1990) Prolonged muscle vibration reducing motor output in maximal voluntary contractions in man. J Physiol 423:15–26. https://doi.org/10.1113/jphysiol.1990.sp018008
Budini F, Laudani L, Bernardini S, Macaluso A (2017) Local vibration inhibits H-reflex but does not compromise manual dexterity and does not increase tremor. Hum Mov Sci 55:221–228. https://doi.org/10.1016/j.humov.2017.08.018
Burke D, Hagbarth KE, Lofstedt L, Wallin BG (1976) The responses of human muscle spindle endings to vibration of non-contracting muscles. J Physiol 261:673–693. https://doi.org/10.1113/jphysiol.1976.sp011580
Burke D, Hicks R, Gandevia SC, Stephen J, Woodforth I, Crawford M (1993) Direct comparison of corticospinal volleys in human subjects to transcranial magnetic and electrical stimulation. J Physiol 470:383–393. https://doi.org/10.1113/jphysiol.1993.sp019864
Calancie B, Broton JG, Klose KJ, Traad M, Difini J, Ayyar DR (1993) Evidence that alterations in presynaptic inhibition contribute to segmental hypo- and hyperexcitability after spinal cord injury in man. Electroencephalogr Clin Neurophysiol 89:177–186. https://doi.org/10.1016/0168-5597(93)90131-8
Chang SH, Tseng SC, McHenry CL, Littmann AE, Suneja M, Shields RK (2012) Limb segment vibration modulates spinal reflex excitability and muscle mRNA expression after spinal cord injury. Clin Neurophysiol 123:558–568. https://doi.org/10.1016/j.clinph.2011.08.001
Craven C, Hitzig SL, Mittmann N (2012) Impact of impairment and secondary health conditions on health preference among Canadians with chronic spinal cord injury. J Spinal Cord Med 35:361–370. https://doi.org/10.1179/2045772312Y.0000000046
Crone C, Nielsen J (1989) Methodological implications of the post activation depression of the soleus H-reflex in man. Exp Brain Res 78:28–32. https://doi.org/10.1007/BF00230683
Curtis DR, Eccles JC (1960) Synaptic action during and after repetitive stimulation. J Physiol 150:374–398. https://doi.org/10.1113/jphysiol.1960.sp006393
De Gail P, Lance JW, Neilson PD (1966) Differential effects on tonic and phasic reflex mechanisms produced by vibration of muscles in man. J Neurol Neurosurg Psychiatry 29:1–11. https://doi.org/10.1136/jnnp.29.1.1
de Noordhout AM, Rapisarda G, Bogacz D, Gerard P, De Pasqua V, Pennisi G, Delwaide PJ (1999) Corticomotoneuronal synaptic connections in normal man: an electrophysiological study. Brain 122(Pt 7):1327–1340. https://doi.org/10.1093/brain/122.7.1327
Goulart F, Valls-Sole J, Alvarez R (2000) Posture-related changes of soleus H-reflex excitability. Muscle Nerve 23:925–932. https://doi.org/10.1002/(sici)1097-4598(200006)23:6%3c925::aid-mus13%3e3.0.co;2-k
Grey MJ, Klinge K, Crone C, Lorentzen J, Biering-Sorensen F, Ravnborg M, Nielsen JB (2008) Post-activation depression of soleus stretch reflexes in healthy and spastic humans. Exp Brain Res 185:189–197. https://doi.org/10.1007/s00221-007-1142-6
Guzman-Lopez J, Costa J, Selvi A, Barraza G, Casanova-Molla J, Valls-Sole J (2012) The effects of transcranial magnetic stimulation on vibratory-induced presynaptic inhibition of the soleus H reflex. Exp Brain Res 220:223–230. https://doi.org/10.1007/s00221-012-3131-7
Harwood B, Scherer J, Brown RE, Cornett KMD, Kenno KA, Jakobi JM (2017) Neuromuscular responses of the plantar flexors to whole-body vibration. Scand J Med Sci Sports 27:1569–1575. https://doi.org/10.1111/sms.12803
Hultborn H, Meunier S, Morin C, Pierrot-Deseilligny E (1987a) Assessing changes in presynaptic inhibition of I a fibres: a study in man and the cat. J Physiol 389:729–756. https://doi.org/10.1113/jphysiol.1987.sp016680
Hultborn H, Meunier S, Pierrot-Deseilligny E, Shindo M (1987b) Changes in presynaptic inhibition of Ia fibres at the onset of voluntary contraction in man. J Physiol 389:757–772. https://doi.org/10.1113/jphysiol.1987.sp016681
Hultborn H, Illert M, Nielsen J, Paul A, Ballegaard M, Wiese H (1996) On the mechanism of the post-activation depression of the H-reflex in human subjects. Exp Brain Res 108:450–462. https://doi.org/10.1007/BF00227268
Jamali M, Carriot J, Chacron MJ, Cullen KE (2013) Strong correlations between sensitivity and variability give rise to constant discrimination thresholds across the otolith afferent population. J Neurosci 33:11302–11313. https://doi.org/10.1523/JNEUROSCI.0459-13.2013
Kawahira K, Higashihara K, Matsumoto S, Shimodozono M, Etoh S, Tanaka N, Sueyoshi Y (2004) New functional vibratory stimulation device for extremities in patients with stroke. Int J Rehabil Res 27:335–337. https://doi.org/10.1097/00004356-200412000-00015
Kohn AF, Floeter MK, Hallett M (1997) Presynaptic inhibition compared with homosynaptic depression as an explanation for soleus H-reflex depression in humans. Exp Brain Res 116:375–380. https://doi.org/10.1007/pl00005765
Krause A, Gollhofer A, Freyler K, Jablonka L, Ritzmann R (2016) Acute corticospinal and spinal modulation after whole body vibration. J Musculoskelet Neuronal Interact 16:327–338
Lance JW, Degail P, Neilson PD (1966) Tonic and phasic spinal cord mechanisms in man. J Neurol Neurosurg Psychiatry 29:535. https://doi.org/10.1136/jnnp.29.6.535
Lapole T, Temesi J, Gimenez P, Arnal PJ, Millet GY, Petitjean M (2015) Achilles tendon vibration-induced changes in plantar flexor corticospinal excitability. Exp Brain Res 233:441–448. https://doi.org/10.1007/s00221-014-4125-4
Magladery JW, McDougal DB Jr (1950) Electrophysiological studies of nerve and reflex activity in normal man. I. Identification of certain reflexes in the electromyogram and the conduction velocity of peripheral nerve fibers. Bull Johns Hopkins Hosp 86:265–290
Martin BJ, Roll JP, Gauthier GM (1986) Inhibitory effects of combined agonist and antagonist muscle vibration on H-reflex in man. Aviat Space Environ Med 57:681–687
McHenry CL, Wu J, Shields RK (2014) Potential regenerative rehabilitation technology: implications of mechanical stimuli to tissue health. BMC Res Notes 7:334. https://doi.org/10.1186/1756-0500-7-334
Mileva KN, Bowtell JL, Kossev AR (2009) Effects of low-frequency whole-body vibration on motor-evoked potentials in healthy men. Exp Physiol 94:103–116. https://doi.org/10.1113/expphysiol.2008.042689
Murillo N, Kumru H, Vidal-Samso J, Benito J, Medina J, Navarro X, Valls-Sole J (2011) Decrease of spasticity with muscle vibration in patients with spinal cord injury. Clin Neurophysiol 122:1183–1189. https://doi.org/10.1016/j.clinph.2010.11.012
Murillo N, Valls-Sole J, Vidal J, Opisso E, Medina J, Kumru H (2014) Focal vibration in neurorehabilitation. Eur J Phys Rehabil Med 50:231–242
Nielsen J, Petersen N, Crone C (1995) Changes in transmission across synapses of Ia afferents in spastic patients. Brain 118(Pt 4):995–1004. https://doi.org/10.1093/brain/118.4.995
Noma T, Matsumoto S, Etoh S, Shimodozono M, Kawahira K (2009) Anti-spastic effects of the direct application of vibratory stimuli to the spastic muscles of hemiplegic limbs in post-stroke patients. Brain Inj 23:623–631. https://doi.org/10.1080/02699050902997896
Noma T, Matsumoto S, Shimodozono M, Etoh S, Kawahira K (2012) Anti-spastic effects of the direct application of vibratory stimuli to the spastic muscles of hemiplegic limbs in post-stroke patients: a proof-of-principle study. J Rehabil Med 44:325–330. https://doi.org/10.2340/16501977-0946
Paoloni M, Mangone M, Scettri P, Procaccianti R, Cometa A, Santilli V (2010) Segmental muscle vibration improves walking in chronic stroke patients with foot drop: a randomized controlled trial. Neurorehabil Neural Repair 24:254–262. https://doi.org/10.1177/1545968309349940
Paoloni M, Giovannelli M, Mangone M et al (2013) Does giving segmental muscle vibration alter the response to botulinum toxin injections in the treatment of spasticity in people with multiple sclerosis? A single-blind randomized controlled trial. Clin Rehabil 27:803–812. https://doi.org/10.1177/0269215513480956
Poon DE, Roy FD, Gorassini MA, Stein RB (2008) Interaction of paired cortical and peripheral nerve stimulation on human motor neurons. Exp Brain Res 188:13–21. https://doi.org/10.1007/s00221-008-1334-8
Ritzmann R, Kramer A, Gollhofer A, Taube W (2013) The effect of whole body vibration on the H-reflex, the stretch reflex, and the short-latency response during hopping. Scand J Med Sci Sports 23:331–339. https://doi.org/10.1111/j.1600-0838.2011.01388.x
Roll JP, Vedel JP (1982) Kinaesthetic role of muscle afferents in man, studied by tendon vibration and microneurography. Exp Brain Res 47:177–190. https://doi.org/10.1007/BF00239377
Roll JP, Vedel JP, Ribot E (1989) Alteration of proprioceptive messages induced by tendon vibration in man: a microneurographic study. Exp Brain Res 76:213–222. https://doi.org/10.1007/BF00253639
Rosenkranz K, Pesenti A, Paulus W, Tergau F (2003) Focal reduction of intracortical inhibition in the motor cortex by selective proprioceptive stimulation. Exp Brain Res 149:9–16. https://doi.org/10.1007/s00221-002-1330-3
Rossi S, Hallett M, Rossini PM, Pascual-Leone A, Safety of TMSCG (2009) Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol 120:2008–2039. https://doi.org/10.1016/j.clinph.2009.08.016
Rossi S, Hallett M, Rossini PM, Pascual-Leone A (2011) Screening questionnaire before TMS: an update. Clin Neurophysiol 122:1686. https://doi.org/10.1016/j.clinph.2010.12.037
Rothwell JC, Day BL, Berardelli A, Marsden CD (1984) Effects of motor cortex stimulation on spinal interneurones in intact man. Exp Brain Res 54:382–384. https://doi.org/10.1007/BF00236241
Rudomin P, Schmidt RF (1999) Presynaptic inhibition in the vertebrate spinal cord revisited. Exp Brain Res 129:1–37. https://doi.org/10.1007/s002210050933
Rymer WZ, Hasan Z (1981) Prolonged time course for vibratory suppression of stretch reflex in the decerebrate cat. Exp Brain Res 44:101–112. https://doi.org/10.1007/BF00238754
Sayenko DG, Masani K, Alizadeh-Meghrazi M, Popovic MR, Craven BC (2010) Acute effects of whole body vibration during passive standing on soleus H-reflex in subjects with and without spinal cord injury. Neurosci Lett 482:66–70. https://doi.org/10.1016/j.neulet.2010.07.009
Serranova T, Valls-Sole J, Munoz E, Genis D, Jech R, Seeman P (2008) Abnormal corticospinal tract modulation of the soleus H reflex in patients with pure spastic paraparesis. Neurosci Lett 437:15–19. https://doi.org/10.1016/j.neulet.2008.03.068
Souron R, Besson T, McNeil CJ, Lapole T, Millet GY (2017a) An acute exposure to muscle vibration decreases knee extensors force production and modulates associated central nervous system excitability. Front Hum Neurosci 11:519. https://doi.org/10.3389/fnhum.2017.00519
Souron R, Besson T, Millet GY, Lapole T (2017b) Acute and chronic neuromuscular adaptations to local vibration training. Eur J Appl Physiol 117:1939–1964. https://doi.org/10.1007/s00421-017-3688-8
Souron R, Baudry S, Millet GY, Lapole T (2019) Vibration-induced depression in spinal loop excitability revisited. J Physiol 597:5179–5193. https://doi.org/10.1113/jp278469
Stachowski NJ, Dougherty KJ (2021) Spinal Inhibitory Interneurons: gatekeepers of Sensorimotor Pathways. Int J Mol Sci. https://doi.org/10.3390/ijms22052667
Steyvers M, Levin O, Verschueren SM, Swinnen SP (2003) Frequency-dependent effects of muscle tendon vibration on corticospinal excitability: a TMS study. Exp Brain Res 151:9–14. https://doi.org/10.1007/s00221-003-1427-3
Valls-Sole J, Alvarez R, Tolosa ES (1994) Vibration-induced presynaptic inhibition of the soleus H reflex is temporarily reduced by cortical magnetic stimulation in human subjects. Neurosci Lett 170:149–152. https://doi.org/10.1016/0304-3940(94)90261-5
Yen CL, McHenry CL, Petrie MA, Dudley-Javoroski S, Shields RK (2017) Vibration training after chronic spinal cord injury: evidence for persistent segmental plasticity. Neurosci Lett 647:129–132. https://doi.org/10.1016/j.neulet.2017.03.019
Acknowledgements
This study was supported in part by awards to RKS from the National Institutes of Health (R01-HD084645, R01-HD082109). The authors acknowledge engineer Jason Wu, MS for technical assistance with the controlled vibration system.
Funding
This study was supported in part by awards to RKS from the National Institutes of Health (R01-HD084645, R01-HD082109). The funding source had no other role in the research.
Author information
Authors and Affiliations
Contributions
CLB developed experimental paradigm, collected, analyzed, and interpreted data, drafted portions of the manuscript, and approved the final article. RKS developed the rationale for the project, provided oversight of all experimentation and analysis, procured funding, edited, and approved the final article. SLD analyzed and interpreted data, drafted portions of the manuscript, edited and approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest or competing interests.
Ethics approval
The study was performed in accordance with the principles of the Declaration of Helsinki. Approval was granted by the Institutional Review Board at the University of Iowa.
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable. No identifying information about participants is included in this article.
Additional information
Communicated by Winston D Byblow.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bringman, C.L., Shields, R.K. & DeJong, S.L. Corticospinal modulation of vibration-induced H-reflex depression. Exp Brain Res 240, 803–812 (2022). https://doi.org/10.1007/s00221-022-06306-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-022-06306-w