Abstract
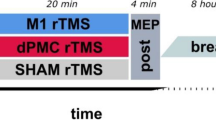
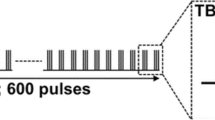
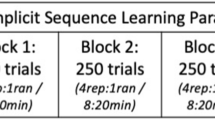
Tecchio et al. (J Neurophysiology 104: 1134–1140, 2010) reported that the application of anodal tDCS at primary motor cortex (M1) immediately after practice of a procedural motor skill enhanced consolidation, which in turn improved offline gain. Tecchio et al. noted, however, that this study did not account for known after-effects associated with this form of non-invasive stimulation. The present study was designed to explicitly reevaluate Tecchio et al.’s claim. As in the original study, individuals experienced either anodal or sham stimulation at M1 after practice of a serial reaction time task (SRTT) followed by test trials 15-min later. Two additional novel conditions experienced the test trials after 120-min rather than 15-min thus allowing potential stimulation after-effects to dissipate. The expectation was that if anodal stimulation influences post-practice consolidation leading to offline gain, this effect would be present not only at 15-min but also after 120-min. In agreement with the working hypothesis, findings revealed offline gain at both 15-min and the longer 2-h time period. Unexpectedly, we found no interaction between real and sham conditions. The lack of difference between Real and Sham effects weakens confidence in the potential of post-practice tDCS for consolidation enhancement, while it is more consistent with other claims that decoupling practice and anodal tDCS stimulation in time can reduce the effectiveness of exogenous stimulation for procedural skill gain.




Similar content being viewed by others
Notes
The distributions for mean RT from the training and test data were not normally distributed. For this reason the median RT for each training and test block was considered a more suitable performance measure for all the reported analyses.
Minus values for the normalized data used for these analyses reflects superior performance for the trained sequence compared to the random sequence.
We appreciate an anonymous reviewer noting the feedback protocol difference between the represent study and that of Tecchio et al. (2010) and the potential implications for the current findings.
References
Amadi U, Allman C, Johansen-Berg H, Stagg CJ (2015) The homeostatic interaction between anodal transcranial direct current stimulation and motor learning in humans is related to GABAA activity. Brain Stimul 8:898–905
Ambrus GG, Chaieb L, Stilling R, Rothkegel H, Antal A, Paulus W (2016) Monitoring transcranial direct current stimulation induced changes in cortical excitability during the serial reaction time task. Neurosci Lett 616:98–104
Batsikadze G, Moliadze V, Paulus W, Kuo MF, Nitsche MA (2013) Partially non-linear stimulation intensity-dependent effects of direct current stimulation on motor cortex excitability in humans. J Physiol 591:1987–2000
Boggio PS, Nunes A, Rigonatti SP, Nitsche MA, Pascual-Leone A, Fregni F (2007) Repeated sessions of noninvasive brain DC stimulation is associated with motor function improvement in stroke patients. Restor Neurol Neurosci 25:123–129
Brashers-Krug T, Shadmehr R, Bizzi E (1996) Consolidation in human motor memory. Nature 382:252–255
Buch ER, Santarnecchi E, Antal A, Born J, Celnik PA, Classen JA et al (2017) Effects of tDCS on motor learning and memory formation: a consensus and critical position paper. Clin Neurophysiol 128:589–603
Ciechanski P, Kirton A (2017) Transcranial direct-current stimulation can enhance motor learning in children. Cereb Cortex 27:2758–2767
Cuypers K, Leenus D, van den Berg J, Nitsche FE, JNitsche MA, Thijs H, Wenderoth N et al (2013) Is motor learning mediated by tDCS intensity? PLoS One 8:e67344
Diekelmann S, Born J (2007) One memory, two ways to consolidate? Nat Neurosci 10:1085–1086
Doyon J, Bellec P, Amsel R, Penhune V, Monchi O, Carrier J, Lehéricy S, Benali H (2009) Contributions of the basal ganglia and functionally related brain structures to motor learning. Behav Brain Res 199:61–75
Fregni F, Boggio PS, Mansur CG, Wagner T, Ferreira MJ, Lima MC, Rigonatti SP, Marcolin MA, Freedman SD, Nitsche MA, Pascual-Leone A (2005) Transcranial direct current stimulation of the unaffected hemisphere in stroke patients. NeuroReport 16:1551–1555
Fritsch B, Reis J, Martinowich K, Schambra HM, Ji Y, Cohen LG, Lu B (2010) Direct current stimulation promotes BDNF-dependent synaptic plasticity: potential implications for motor learning. Neuron 66:198–204
Hikosaka O, Nakahara H, Rand MK, Sakai K, Lu X, Nakamura K, Miyachi S, Doya K (1999) Parallel neural networks for learning sequential procedures. Trends Neurosci 22:464–471
Hummel F, Cohen LG (2005) Improvement of motor function with noninvasive cortical stimulation in a patient with chronic stroke. Neurorehabil Neural Repair 19:14–19
Kantak SS, Mummidisetty CK, Stinear JW (2012) Primary motor and premotor cortex in implicit sequence learning–evidence for competition between implicit and explicit human motor memory systems. Eur J Neurosci 36:2710–2715
Karni A, Meyer G, Rey-Hipolito C, Jezzard P, Adams MM, Turner R, Ungerleider LG (1995) The acquisition of skilled motor performance: fast and slow experience-driven changes in primary motor cortex. Proc Natl Acad Sci 95:861–868
Karok S, Witney AG (2013) Enhanced motor learning following task-concurrent dual transcranial direct current stimulation. PLoS One 8:e85693
Lin CH, Wu AD, Udompholkul P, Knowlton BJ (2010) Contextual interference effects in sequence learning for young and older adults. Psychol Aging 25:929–939
Mayhew SD, Porcaro C, Tecchio F, Bagshaw AP (2017) fMRI characterisation of widespread brain networks relevant for behavioural variability in fine hand motor control with and without visual feedback. Neuroimage 148:330–342
Muellbacher W, Ziemann U, Wissel J, Dang N, Kofler M, Facchini S, Boroojerdi B, Poewe W, Hallett M (2002) Early consolidation in human primary motor cortex. Nature 415:640–644
Naros G, Geyer M, Koch S, Mayr L, Ellinger T, Grimm F et al (2016) Enhanced motor learning with bilateral transcranial direct current stimulation: impact of polarity or current flow direction? Clin Neurophysiol 127:2119–2126
Nitsche MA, Paulus W (2000) Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol 15:633–639
Nitsche MA, Schauenburg A, Lang N, Liebetanz D, Exner C, Paulus W et al (2003) Facilitation of implicit motor learning by weak transcranial direct current stimulation of the primary motor cortex in the human. J Cognit Neurosci 15:619–626
Penhune VB, Steele CJ (2012) Parallel contributions of cerebellar, striatal and M1 mechanisms to motor sequence learning. Behav Brain Res 15:579–591
Prichard G, Weiller C, Fritsch B, Reis J (2014) Effects of different electrical brain stimulation protocols on subcomponents of motor skill learning. Brain Stimulation 7:532–540
Reis J, Fritsch B (2011) Modulation of motor performance and motor learning by transcranial direct current stimulation. Curr Opin Neurol 24:590–596
Reis J, Schambra HM, Cohen LG, Buch ER, Fritsch B, Zarahn E et al (2009) Noninvasive cortical stimulation enhances motor skill acquisition over multiple days through an effect on consolidation. Proc Natl Acad Sci 106:1590–1595
Reis J, Fischer T, Prichard G, Weiller C, Cohen LG, Fritsch B (2015) Time- but Not sleep-dependent consolidation of tDCS-enhanced visuomotor skills. Cereb Cortex 25:109–117
Saucedo Marquez CM, Zhang X, Swinnen SP, Meesen R, Wenderoth N (2013) Task-specific effect of transcranial direct current stimulation on motor learning. Front Hum Neurosci 7:333. https://doi.org/10.3389/fnhum.2013.00333
Sidaway B, Ala B, Baughman K, Glidden J, Cowie S, Peabody A, Roundy D, Spaulding J, Stephens R, Wright DL (2016) Contextual interference can facilitate motor learning in older adults and in individuals with Parkinson’s Disease. J Mot Behav 48:509–518
Stagg CJ, Jayaram G, Pastor D, Kincses ZT, Matthews PM, Johansen-Berg H (2011) Polarity and timing-dependent effects of transcranial direct current stimulation in explicit motor learning. Neuropsychologia 49:800–804
Tecchio F, Zappasodi F, Assenza G, Tombini M, Vollaro S, Barbati G, Rossini PM (2010) Anodal transcranial direct current stimulation enhances procedural consolidation. J Neurophysiol 104:1134–1140
Walker M, Brakefield T, Hobson JA, Strickgold R (2003) Dissociable stages of human memory consolidation and reconsolidation. Nature 425:616–620
Waters-Metenier S, Husain M, Wiestler T, Diedrichsen J (2014) Bihemispheric transcranial direct current stimulation enhances effector-independent representations of motor synergy and sequence learning. J Neurosci 34:1037–1050
Zimerman M, Nitsch M, Giraux P, Gerloff C, Cohen LG, Hummel FC (2013) Neuroenhancement of the aging brain: restoring skill acquisition in old subjects. Ann Neurol 73:10–15
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by John C. Rothwell.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, J., McCulloch, A., Kim, H. et al. Application of anodal tDCS at primary motor cortex immediately after practice of a motor sequence does not improve offline gain. Exp Brain Res 238, 29–37 (2020). https://doi.org/10.1007/s00221-019-05697-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05697-7