Abstract
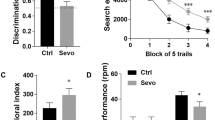
Postoperative cognitive impairment is especially common in older patients following major surgery. Although exposure to sevoflurane is known to cause memory deficits, few studies have examined the putative approaches to reduce such impairments. This study tested the hypotheses that sevoflurane exposure can decrease NR2B subunit-containing NMDA receptor activity in hippocampus of aged mice, and voluntary exercise may counteract the declining hippocampal functions. We found that long exposure (3 h/day for 3 days), but not short exposure (1 h/day for 3 days), to 3 % sevoflurane produced a long-lasting spatial memory deficits up to 3 weeks in aged mice, and such an effect was not due to the neuronal loss in the hippocampus, but was correlated with a long-term decrease in Fyn kinase expression and NR2B subunit phosphorylation in the hippocampus. Furthermore, voluntary exercise rescued sevoflurane-induced spatial memory deficits in aged mice and restored Fyn kinase expression and NR2B subunit phosphorylation in the hippocampus to a level comparable to control animals. Generally, our results suggested that Fyn-mediated NR2B subunit phosphorylation may play a critical role in sevoflurane-induced impairment in cognitive functions in aged animals, and voluntary exercise might be an important non-pharmacological approach to treatment of inhaled anesthetics-induced postoperative cognitive impairment in clinical settings.





Similar content being viewed by others
References
Alkire MT, Gorski LA (2004) Relative amnesic potency of five inhalational anesthetics follows the Meyer–Overton rule. Anesthesiology 101:417–429
Alkire MT, Nathan SV (2005) Does the amygdala mediate anesthetic-induced amnesia? Basolateral amygdala lesions block sevoflurane-induced amnesia. Anesthesiology 102:754–760. doi:10.1097/00000542-200504000-00010
Alkire MT, Nathan SV, McReynolds JR (2005) Memory enhancing effect of low-dose sevoflurane does not occur in basolateral amygdala-lesioned rats. Anesthesiology 103:1167–1173. doi:10.1097/00000542-200512000-00010
Alkire MT, Gruver R, Miller J, McReynolds JR, Hahn EL, Cahill L (2008a) Neuroimaging analysis of an anesthetic gas that blocks human emotional memory. Proc Natl Acad Sci USA 105:1722–1727
Alkire MT, Hudetz AG, Tononi G (2008b) Consciousness and anesthesia. Science 322:876–880
Angulo MC, Kozlov AS, Charpak S, Audinat E (2004) Glutamate released from glial cells synchronizes neuronal activity in the hippocampus. J Neurosci 24:6920–6927. doi:10.1523/JNEUROSCI.0473-04.2004
Ba M, Kong M, Ma G (2015) Postsynaptic density protein 95-regulated NR2B tyrosine phosphorylation and interactions of Fyn with NR2B in levodopa-induced dyskinesia rat models. Drug Des Dev Ther 9:199–206. doi:10.2147/DDDT.S75495
Bibb JA, Mayford MR, Tsien JZ, Alberini CM (2010) Cognition enhancement strategies. J Neurosci 30:14987–14992. doi:10.1523/JNEUROSCI.4419-10.2010
Brim BL, Haskell R, Awedikian R et al (2013) Memory in aged mice is rescued by enhanced expression of the GluN2B subunit of the NMDA receptor. Behav Brain Res 238:211–226. doi:10.1016/j.bbr.2012.10.026
Chen BS, Roche KW (2007) Regulation of NMDA receptors by phosphorylation. Neuropharmacology 53:362–368. doi:10.1016/j.neuropharm.2007.05.018
Collingridge GL, Volianskis A, Bannister N et al (2013) The NMDA receptor as a target for cognitive enhancement. Neuropharmacology 64:13–26. doi:10.1016/j.neuropharm.2012.06.051
Corcoran KA, Donnan MD, Tronson NC et al (2011) NMDA receptors in retrosplenial cortex are necessary for retrieval of recent and remote context fear memory. J Neurosci 31:11655–11659. doi:10.1523/JNEUROSCI.2107-11.2011
Coultrap SJ, Nixon KM, Alvestad RM, Valenzuela CF, Browning MD (2005) Differential expression of NMDA receptor subunits and splice variants among the CA1, CA3 and dentate gyrus of the adult rat. Brain Res Mol Brain Res 135:104–111. doi:10.1016/j.molbrainres.2004.12.005
Culley DJ, Baxter M, Yukhananov R, Crosby G (2003) The memory effects of general anesthesia persist for weeks in young and aged rats. Anesth Analg 96:1004–1009 (table of contents)
Culley DJ, Baxter MG, Crosby CA, Yukhananov R, Crosby G (2004a) Impaired acquisition of spatial memory 2 weeks after isoflurane and isoflurane–nitrous oxide anesthesia in aged rats. Anesth Analg 99:1393–1397. doi:10.1213/01.ANE.0000135408.14319.CC (table of contents)
Culley DJ, Baxter MG, Yukhananov R, Crosby G (2004b) Long-term impairment of acquisition of a spatial memory task following isoflurane–nitrous oxide anesthesia in rats. Anesthesiology 100:309–314
Eckenhoff RG, Johansson JS, Wei H et al (2004) Inhaled anesthetic enhancement of amyloid-beta oligomerization and cytotoxicity. Anesthesiology 101:703–709
Evered L, Scott DA, Silbert B, Maruff P (2011) Postoperative cognitive dysfunction is independent of type of surgery and anesthetic. Anesth Analg 112:1179–1185. doi:10.1213/ANE.0b013e318215217e
Fang F, Xue Z, Cang J (2012) Sevoflurane exposure in 7-day-old rats affects neurogenesis, neurodegeneration and neurocognitive function. Neurosci Bull 28:499–508. doi:10.1007/s12264-012-1260-4
Farmer J, Zhao X, van Praag H, Wodtke K, Gage FH, Christie BR (2004) Effects of voluntary exercise on synaptic plasticity and gene expression in the dentate gyrus of adult male Sprague-Dawley rats in vivo. Neuroscience 124:71–79. doi:10.1016/j.neuroscience.2003.09.029
Fellini L, Florian C, Courtey J, Roullet P (2009) Pharmacological intervention of hippocampal CA3 NMDA receptors impairs acquisition and long-term memory retrieval of spatial pattern completion task. Learn Mem 16:387–394. doi:10.1101/lm.1433209
Feng X, Liu JJ, Zhou X et al (2012) Single sevoflurane exposure decreases neuronal nitric oxide synthase levels in the hippocampus of developing rats. Br J Anaesth 109:225–233. doi:10.1093/bja/aes121
Hardingham GE, Bading H (2010) Synaptic versus extrasynaptic NMDA receptor signalling: implications for neurodegenerative disorders. Nat Rev Neurosci 11:682–696. doi:10.1038/nrn2911
Harri M, Lindblom J, Malinen H, Hyttinen M, Lapvetelainen T, Eskola S, Helminen HJ (1999) Effect of access to a running wheel on behavior of C57BL/6 J mice. Lab Anim Sci 49:401–405
Haseneder R, Starker L, Berkmann J et al (2013) Sevoflurane anesthesia improves cognitive performance in mice, but does not influence in vitro long-term potentation in hippocampus CA1 stratum radiatum. PLoS ONE 8:e64732. doi:10.1371/journal.pone.0064732
Hogue CW, Lillie R, Hershey T, Birge S, Nassief AM, Thomas B, Freedland KE (2003) Gender influence on cognitive function after cardiac operation. Ann Thorac Surg 76:1119–1125
Jamali-Raeufy N, Nasehi M, Zarrindast MR (2011) Influence of N-methyl d-aspartate receptor mechanism on WIN55,212-2-induced amnesia in rat dorsal hippocampus. Behav Pharmacol 22:645–654. doi:10.1097/FBP.0b013e32834aff1f
Jarrard LE (1993) On the role of the hippocampus in learning and memory in the rat. Behav Neural Biol 60:9–26
Kesner RP (2007) Behavioral functions of the CA3 subregion of the hippocampus. Learn Mem 14:771–781. doi:10.1101/lm.688207
Komatsu H, Nogaya J, Anabuki D, Yokono S, Kinoshita H, Shirakawa Y, Ogli K (1993) Memory facilitation by posttraining exposure to halothane, enflurane, and isoflurane in ddN mice. Anesth Analg 76:609–612
Lee KY, Chou AK, Yang LC, Buerkle H (2003) NMDA receptors offer more than one functionality. Anesth Analg 96:1533–1534 (author reply 1534)
Liu Y, Wong TP, Aarts M et al (2007) NMDA receptor subunits have differential roles in mediating excitotoxic neuronal death both in vitro and in vivo. J Neurosci 27:2846–2857. doi:10.1523/JNEUROSCI.0116-07.2007
Liu XS, Xue QS, Zeng QW, Li Q, Liu J, Feng XM, Yu BW (2010) Sevoflurane impairs memory consolidation in rats, possibly through inhibiting phosphorylation of glycogen synthase kinase-3beta in the hippocampus. Neurobiol Learn Mem 94:461–467. doi:10.1016/j.nlm.2010.08.011
Liu J, Zhang X, Zhang W, Gu G, Wang P (2015) Effects of sevoflurane on young male adult C57BL/6 mice spatial cognition. PLoS ONE 10:e0134217. doi:10.1371/journal.pone.0134217
Maher A, El-Sayed NS, Breitinger HG, Gad MZ (2014) Overexpression of NMDAR2B in an inflammatory model of Alzheimer’s disease: modulation by NOS inhibitors. Brain Res Bull 109:109–116. doi:10.1016/j.brainresbull.2014.10.007
Matus-Amat P, Higgins EA, Sprunger D, Wright-Hardesty K, Rudy JW (2007) The role of dorsal hippocampus and basolateral amygdala NMDA receptors in the acquisition and retrieval of context and contextual fear memories. Behav Neurosci 121:721–731. doi:10.1037/0735-7044.121.4.721
Melik E, Babar E, Ozen E, Ozgunen T (2006) Hypofunction of the dorsal hippocampal NMDA receptors impairs retrieval of memory to partially presented foreground context in a single-trial fear conditioning in rats. Eur Neuropsychopharmacol 16:241–247. doi:10.1016/j.euroneuro.2005.07.008
Nyiri G, Stephenson FA, Freund TF, Somogyi P (2003) Large variability in synaptic N-methyl-d-aspartate receptor density on interneurons and a comparison with pyramidal-cell spines in the rat hippocampus. Neuroscience 119:347–363
Pandis C, Sotiriou E, Kouvaras E, Asprodini E, Papatheodoropoulos C, Angelatou F (2006) Differential expression of NMDA and AMPA receptor subunits in rat dorsal and ventral hippocampus. Neuroscience 140:163–175. doi:10.1016/j.neuroscience.2006.02.003
Quinn JJ, Loya F, Ma QD, Fanselow MS (2005) Dorsal hippocampus NMDA receptors differentially mediate trace and contextual fear conditioning. Hippocampus 15:665–674. doi:10.1002/hipo.20088
Rammes G, Starker LK, Haseneder R et al (2009) Isoflurane anaesthesia reversibly improves cognitive function and long-term potentiation (LTP) via an up-regulation in NMDA receptor 2B subunit expression. Neuropharmacology 56:626–636. doi:10.1016/j.neuropharm.2008.11.002
Rasmussen LS, Johnson T, Kuipers HM et al (2003) Does anaesthesia cause postoperative cognitive dysfunction? A randomised study of regional versus general anaesthesia in 438 elderly patients. Acta Anaesthesiol Scand 47:260–266
Sakai EM, Connolly LA, Klauck JA (2005) Inhalation anesthesiology and volatile liquid anesthetics: focus on isoflurane, desflurane, and sevoflurane. Pharmacotherapy 25:1773–1788. doi:10.1592/phco.2005.25.12.1773
Satomoto M, Satoh Y, Terui K, Miyao H, Takishima K, Ito M, Imaki J (2009) Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in fear conditioning in mice. Anesthesiology 110:628–637. doi:10.1097/ALN.0b013e3181974fa2
Schenning KJ, Deiner SG (2015) Postoperative delirium in the geriatric patient. Anesthesiol Clin 33:505–516. doi:10.1016/j.anclin.2015.05.007
Serrano A, Robitaille R, Lacaille JC (2008) Differential NMDA-dependent activation of glial cells in mouse hippocampus. Glia 56:1648–1663. doi:10.1002/glia.20717
Shen X, Dong Y, Xu Z et al (2013) Selective anesthesia-induced neuroinflammation in developing mouse brain and cognitive impairment. Anesthesiology 118:502–515. doi:10.1097/ALN.0b013e3182834d77
Siegel SJ, Brose N, Janssen WG, Gasic GP, Jahn R, Heinemann SF, Morrison JH (1994) Regional, cellular, and ultrastructural distribution of N-methyl-d-aspartate receptor subunit 1 in monkey hippocampus. Proc Natl Acad Sci USA 91:564–568
Solt K, Forman SA (2007) Correlating the clinical actions and molecular mechanisms of general anesthetics. Curr Opin Anaesthesiol 20:300–306. doi:10.1097/ACO.0b013e32816678a5
Szabadits E, Cserep C, Szonyi A et al (2011) NMDA receptors in hippocampal GABAergic synapses and their role in nitric oxide signaling. J Neurosci 31:5893–5904. doi:10.1523/JNEUROSCI.5938-10.2011
Takumi Y, Matsubara A, Rinvik E, Ottersen OP (1999) The arrangement of glutamate receptors in excitatory synapses. Ann N Y Acad Sci 868:474–482
Tang YP, Shimizu E, Dube GR et al (1999) Genetic enhancement of learning and memory in mice. Nature 401:63–69. doi:10.1038/43432
Tejada-Simon MV, Villasana LE, Serrano F, Klann E (2006) NMDA receptor activation induces translocation and activation of Rac in mouse hippocampal area CA1. Biochem Biophys Res Commun 343:504–512. doi:10.1016/j.bbrc.2006.02.183
Todd KJ, Serrano A, Lacaille JC, Robitaille R (2006) Glial cells in synaptic plasticity. J Physiol Paris 99:75–83. doi:10.1016/j.jphysparis.2005.12.002
Volianskis A, Bannister N, Collett VJ et al (2013) Different NMDA receptor subtypes mediate induction of long-term potentiation and two forms of short-term potentiation at CA1 synapses in rat hippocampus in vitro. J Physiol 591:955–972. doi:10.1113/jphysiol.2012.247296
Wang YT, Salter MW (1994) Regulation of NMDA receptors by tyrosine kinases and phosphatases. Nature 369:233–235. doi:10.1038/369233a0
Wang D, Jacobs SA, Tsien JZ (2014a) Targeting the NMDA receptor subunit NR2B for treating or preventing age-related memory decline. Expert Opin Ther Targets 18:1121–1130. doi:10.1517/14728222.2014.941286
Wang WY, Jia LJ, Luo Y et al (2014b) Location- and subunit-specific NMDA receptors determine the developmental sevoflurane neurotoxicity through ERK1/2 signaling. Mol Neurobiol. doi:10.1007/s12035-014-9005-1
Wiklund A, Granon S, Faure P, Sundman E, Changeux JP, Eriksson LI (2009) Object memory in young and aged mice after sevoflurane anaesthesia. NeuroReport 20:1419–1423. doi:10.1097/WNR.0b013e328330cd2b
Williams JM, Mason-Parker SE, Abraham WC, Tate WP (1998) Biphasic changes in the levels of N-methyl-d-aspartate receptor-2 subunits correlate with the induction and persistence of long-term potentiation. Brain Res Mol Brain Res 60:21–27
Williams-Russo P, Sharrock NE, Mattis S, Szatrowski TP, Charlson ME (1995) Cognitive effects after epidural vs general anesthesia in older adults. A randomized trial. JAMA 274:44–50
Xiong WX, Zhou GX, Wang B, Xue ZG, Wang L, Sun HC, Ge SJ (2013) Impaired spatial learning and memory after sevoflurane-nitrous oxide anesthesia in aged rats is associated with down-regulated cAMP/CREB signaling. PLoS ONE 8:e79408. doi:10.1371/journal.pone.0079408
Xue JG, Masuoka T, Gong XD, Chen KS, Yanagawa Y, Law SK, Konishi S (2011) NMDA receptor activation enhances inhibitory GABAergic transmission onto hippocampal pyramidal neurons via presynaptic and postsynaptic mechanisms. J Neurophysiol 105:2897–2906. doi:10.1152/jn.00287.2010
Yaka R, He DY, Phamluong K, Ron D (2003) Pituitary adenylate cyclase-activating polypeptide (PACAP(1–38)) enhances N-methyl-d-aspartate receptor function and brain-derived neurotrophic factor expression via RACK1. J Biol Chem 278:9630–9638. doi:10.1074/jbc.M209141200
Yoshihara T, Ichitani Y (2004) Hippocampal N-methyl-d-aspartate receptor-mediated encoding and retrieval processes in spatial working memory: delay-interposed radial maze performance in rats. Neuroscience 129:1–10. doi:10.1016/j.neuroscience.2004.07.030
Yu XM, Askalan R, Keil GJ 2nd, Salter MW (1997) NMDA channel regulation by channel-associated protein tyrosine kinase Src. Science 275:674–678
Yu X, Liu Y, Bo S, Qinghua L (2015) Effects of sevoflurane on learning, memory, and expression of pERK1/2 in hippocampus in neonatal rats. Acta Anaesthesiol Scand 59:78–84. doi:10.1111/aas.12433
Zadori D, Veres G, Szalardy L, Klivenyi P, Toldi J, Vecsei L (2014) Glutamatergic dysfunctioning in Alzheimer’s disease and related therapeutic targets. J Alzheimers Dis 42(Suppl 3):S177–S187. doi:10.3233/JAD-13262
Zhang F, Jia J, Wu Y, Hu Y, Wang Y (2010) The effect of treadmill training pre-exercise on glutamate receptor expression in rats after cerebral ischemia. Int J Mol Sci 11:2658–2669. doi:10.3390/ijms11072658
Zhang A, Bai Y, Hu Y et al (2012) The effects of exercise intensity on p-NR2B expression in cerebral ischemic rats. Can J Neurol Sci 39:613–618
Zhang XH, Liu SS, Yi F, Zhuo M, Li BM (2013) Delay-dependent impairment of spatial working memory with inhibition of NR2B-containing NMDA receptors in hippocampal CA1 region of rats. Mol Brain 6:13. doi:10.1186/1756-6606-6-13
Zhang J, Dong Y, Zhou C, Zhang Y, Xie Z (2015) Anesthetic sevoflurane reduces levels of hippocalcin and postsynaptic density protein 95. Mol Neurobiol 51:853–863. doi:10.1007/s12035-014-8746-1
Zhou M, Baudry M (2006) Developmental changes in NMDA neurotoxicity reflect developmental changes in subunit composition of NMDA receptors. J Neurosci 26:2956–2963. doi:10.1523/JNEUROSCI.4299-05.2006
Zhou X, Ding Q, Chen Z, Yun H, Wang H (2013) Involvement of the GluN2A and GluN2B subunits in synaptic and extrasynaptic N-methyl-d-aspartate receptor function and neuronal excitotoxicity. J Biol Chem 288:24151–24159. doi:10.1074/jbc.M113.482000
Acknowledgments
This study was funded by National Natural Science Foundation of Jilin Province, China (20160101092JC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Rights and permissions
About this article
Cite this article
Tian, D., Tian, M., Ma, Z. et al. Voluntary exercise rescues sevoflurane-induced memory impairment in aged male mice. Exp Brain Res 234, 3613–3624 (2016). https://doi.org/10.1007/s00221-016-4756-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4756-8