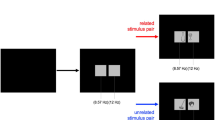
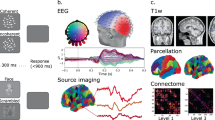
Abstract
In cognitive neuroscience, interest in the neuronal basis underlying the processing of human bodies is steadily increasing. Based on functional magnetic resonance imaging studies, it is assumed that the processing of pictures of human bodies is anchored in a network of specialized brain areas comprising the extrastriate and the fusiform body area (EBA, FBA). An alternative to examine the dynamics within these networks is electroencephalography, more specifically so-called steady-state visually evoked potentials (SSVEPs). In SSVEP tasks, a visual stimulus is presented repetitively at a predefined flickering rate and typically elicits a continuous oscillatory brain response at this frequency. This brain response is characterized by an excellent signal-to-noise ratio—a major advantage for source reconstructions. The main goal of present study was to demonstrate the feasibility of this method to study human body perception. To that end, we presented pictures of bodies and contrasted the resulting SSVEPs to two control conditions, i.e., non-objects and pictures of everyday objects (chairs). We found specific SSVEPs amplitude differences between bodies and both control conditions. Source reconstructions localized the SSVEP generators to a network of temporal, occipital and parietal areas. Interestingly, only body perception resulted in activity differences in middle temporal and lateral occipitotemporal areas, most likely reflecting the EBA/FBA.




Similar content being viewed by others
Notes
Statistical comparisons in source space were carried out by means of Hotelling t 2 tests. This approach does not provide information regarding the direction of significant effects. Nonetheless, it seems a valid assumption that the amplitudes as measures with scalp electrodes are equivalent to the strength of the current densities in source space.
References
Atkinson AP, Adolphs R (2011) The neuropsychology of face perception: beyond simple dissociations and functional selectivity. Philos Trans R Soc Lond B Biol Sci 366:1726–1738. doi:10.1098/rstb.2010.0349
Berlucchi G, Aglioti SM (2010) The body in the brain revisited. Exp Brain Res 200:25–35. doi:10.1007/s00221-009-1970-7
Bertrand O, Pantev C (1994) Stimulus frequency dependence of the transient oscillatory auditory evoked response (40 Hz) studied by electric and magnetic recordings in human. In: Pantev C, Elbert T, Lütkenhöner B (eds) Oscillatory event-related brain dynamics. Plenum, New York, pp 231–242
Binder JR, Desai RH, Graves WW, Conant LL (2009) Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex 19:2767–2796. doi:10.1093/cercor/bhp055
Bona S, Cattaneo Z, Silvanto J (2015) The causal role of the occipital face area (OFA) and lateral occipital (LO) cortex in symmetry perception. J Neurosci 35:731–738. doi:10.1523/jneurosci.3733-14.2015
Bosch-Bayard J, Valdes-Sosa P, Virues-Alba T, Aubert-Vazquez E, John ER, Harmony T, Riera-Diaz J, Trujillo-Barreto N (2001) 3D statistical parametric mapping of EEG source spectra by means of variable resolution electromagnetic tomography (VARETA). Clin Electroencephalogr 32:47–61
Busch NA, Herrmann CS, Müller MM, Lenz D, Gruber T (2006) A cross-laboratory study of event-related gamma activity in a standard object recognition paradigm. NeuroImage 33:1169–1177. doi:10.1016/j.neuroimage.2006.07.034
Capilla A, Pazo-Alvarez P, Darriba A, Campo P, Gross J (2011) Steady-state visual evoked potentials can be explained by temporal superposition of transient event-related responses. PLoS One 6:e14543. doi:10.1371/journal.pone.0014543
Caspari N, Popivanov ID, De Maziere PA, Vanduffel W, Vogels R, Orban GA, Jastorff J (2014) Fine-grained stimulus representations in body selective areas of human occipito-temporal cortex. NeuroImage 102(Pt 2):484–497. doi:10.1016/j.neuroimage.2014.07.066
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134:9–21. doi:10.1016/j.jneumeth.2003.10.009
Downing PE, Peelen MV (2011) The role of occipitotemporal body-selective regions in person perception. Cogn Neurosci 2:186–203. doi:10.1080/17588928.2011.582945
Downing PE, Peelen MV (2015) Body selectivity in occipitotemporal cortex: causal evidence. Neuropsychologia. doi:10.1016/j.neuropsychologia.2015.05.033
Downing PE, Jiang Y, Shuman M, Kanwisher N (2001) A cortical area selective for visual processing of the human body. Science 293:2470–2473
Evans AC, Collins DL, Mills SR, Brown ED, Kelly RI, Peters TM (1993) 3D statistical neuroanatomical models from 305 MRI volumes. In: Proceedings of the IEEE-nuclear science symposium and medical imaging conference, vol 95, pp 1813–1817
Giabbiconi CM, Dancer C, Zopf R, Gruber T, Müller MM (2004) Selective spatial attention to left or right hand flutter sensation modulates the steady-state somatosensory evoked potential. Brain Res Cogn Brain Res 20:58–66. doi:10.1016/j.cogbrainres.2004.01.004
Giabbiconi CM, Trujillo-Barreto NJ, Gruber T, Müller MM (2007) Sustained spatial attention to vibration is mediated in primary somatosensory cortex. NeuroImage 35:255–262. doi:10.1016/j.neuroimage.2006.11.022
Grill-Spector K, Kourtzi Z, Kanwisher N (2001) The lateral occipital complex and its role in object recognition. Vision Res 41:1409–1422
Gruber T, Müller MM (2005) Oscillatory brain activity dissociates between associative stimulus content in a repetition priming task in the human EEG. Cereb Cortex 15:109–116. doi:10.1093/cercor/bhh113
Gruber T, Trujillo-Barreto NJ, Giabbiconi CM, Valdes-Sosa PA, Müller MM (2006) Brain electrical tomography (BET) analysis of induced gamma band responses during a simple object recognition task. NeuroImage 29:888–900. doi:10.1016/j.neuroimage.2005.09.004
Gruber T, Maess B, Trujillo-Barreto NJ, Muller MM (2008) Sources of synchronized induced Gamma-Band responses during a simple object recognition task: a replication study in human MEG. Brain Res 1196:74–84. doi:10.1016/j.brainres.2007.12.037
Gruss LF, Wieser MJ, Schweinberger SR, Keil A (2012) Face-evoked steady-state visual potentials: effects of presentation rate and face inversion. Front Hum Neurosci 6:1–10
Hindi Attar C, Müller MM (2012) Selective attention to task-irrelevant emotional distractors is unaffected by the perceptual load associated with a foreground task. PLoS One 7:e37186. doi:10.1371/journal.pone.0037186
Hodzic A, Muckli L, Singer W, Stirn A (2009) Cortical responses to self and others. Hum Brain Mapp 30:951–962
Jasper HH (1958) The ten-twenty electrode system of the International Federation. Electroencephalogr Clin Neurophysiol 10:371–375
Kanwisher N (2000) Domain specificity in face perception. Nat Neurosci 3:759–763. doi:10.1038/77664
Kanwisher N, McDermott J, Chun MM (1997) The fusiform face area: a module in human extrastriate cortex specialized for face perception. J Neurosci 17:4302–4311
Kaspar K, Hassler U, Martens U, Trujillo-Barreto N, Gruber T (2010) Steady-state visually evoked potential correlates of object recognition. Brain Res 1343:112–121. doi:10.1016/j.brainres.2010.04.072
Keil A, Gruber T, Müller MM, Moratti S, Stolarova M, Bradley MM, Lang PJ (2003) Early modulation of visual perception by emotional arousal: evidence from steady-state visual evoked brain potentials. Cogn Affect Behav Neurosci 3:195–206
Keil A, Moratti S, Sabatinelli D, Bradley MM, Lang PJ (2005) Additive effects of emotional content and spatial selective attention on electrocortical facilitation. Cereb Cortex 15(8):1187–1197
Keil A, Stolarova M, Moratti S, Ray WJ (2007) Adaptation in visual cortex as a mechanism for rapid discrimination of aversive stimuli. Neuroimage. 36:472–479
Keil A, Costa V, Smith JC, Sabatinelli D, McGinnis EM, Bradley MM, Lang PJ (2012) Tagging cortical networks in emotion: a topographical analysis. Hum Brain Mapp 33:2920–2931
Klem GH, Luders HO, Jasper HH, Elger C (1999) The ten-twenty electrode system of the International Federation. The International Federation of Clinical Neurophysiology. Electroencephalogr Clin Neurophysiol Suppl 52:3–6
Kourtzi Z, Kanwisher N (2000) Cortical regions involved in perceiving object shape. J Neurosci 20:3310–3318
Lang PJ, Bradley MM, Cuthbert BN (2005) International affective picture system (IAPS): affective ratings of pictures and instruction manual. Technical Report A-6. University of Florida, Gainesville, FL
Leder H, Bruce V (2000) When inverted faces are recognized: the role of configural information in face recognition. Q J Exp Psychol A 53:513–536. doi:10.1080/713755889
Martens U, Gruber T (2012) Sharpening and formation: two distinct neuronal mechanisms of repetition priming. Eur J Neurosci 36:2989–2995. doi:10.1111/j.1460-9568.2012.08222.x
Martens U, Trujillo-Barreto N, Gruber T (2011) Perceiving the tree in the woods: segregating brain responses to stimuli constituting natural scenes. J Neurosci 31:17713–17718. doi:10.1523/jneurosci.4743-11.2011
Martens U, Gert AL, Gruber T (2012a) Influences of encoding and retrieval on the steady-state visual evoked potential. NeuroReport 23:337–341. doi:10.1097/WNR.0b013e3283516542
Martens U, Wahl P, Hassler U, Friese U, Gruber T (2012b) Implicit and explicit contributions to object recognition: evidence from rapid perceptual learning. PLoS One 7:e47009. doi:10.1371/journal.pone.0047009
Maurer D, Grand RL, Mondloch CJ (2002) The many faces of configural processing. Trends Cogn Sci 6:255–260
Minnebusch DA, Daum I (2009) Neuropsychological mechanisms of visual face and body perception. Neurosci Biobehav Rev 33:1133–1144
Müller MM, Teder-Salejarvi W, Hillyard SA (1998) The time course of cortical facilitation during cued shifts of spatial attention. Nat Neurosci 1:631–634. doi:10.1038/2865
Müller MM, Malinowski P, Gruber T, Hillyard SA (2003) Sustained division of the attentional spotlight. Nature 424:309–312. doi:10.1038/nature01812
Müller MM, Andersen S, Trujillo NJ, Valdes-Sosa P, Malinowski P, Hillyard SA (2006) Feature-selective attention enhances color signals in early visual areas of the human brain. Proc Natl Acad Sci USA 103:14250–14254. doi:10.1073/pnas.0606668103
Nolan H, Whelan R, Reilly RB (2010) FASTER: fully automated statistical thresholding for EEG artifact rejection. J Neurosci Methods 192:152–162. doi:10.1016/j.jneumeth.2010.07.015
Peelen MV, Downing PE (2005) Selectivity for the human body in the fusiform gyrus. J Neurophysiol 93:603–608. doi:10.1152/jn.00513.2004
Peelen MV, Downing PE (2007) The neural basis of visual body perception. Nat Neurosci 8:636–648
Picton TW, Bentin S, Berg P, Donchin E, Hillyard SA, Johnson R Jr, Miller GA, Ritter W, Ruchkin DS, Rugg MD, Taylor MJ (2000) Guidelines for using human event-related potentials to study cognition: recording standards and publication criteria. Psychophysiology 37:127–152
Popivanov ID, Jastorff J, Vanduffel W, Vogels R (2014) Heterogeneous single-unit selectivity in an fMRI-defined body-selective patch. J Neurosci 34:95–111. doi:10.1523/jneurosci.2748-13.2014
Regan D (1989) Human brain Electrophysiology: evoked potentials and evoked magnetic fields in science and medicine. Elsevier, New York
Rossion B, Boremanse A (2011) Robust sensitivity to facial identity in the right human occipito-temporal cortex as revealed by steady-state visual-evoked potentials. J Vis 11(2):1–21. doi:10.1167/11.2.16
Schönwald LI, Müller MM (2014) Slow biasing of processing resources in early visual cortex is preceded by emotional cue extraction in emotion-attention competition. Hum Brain Mapp 35:1477–1490. doi:10.1002/hbm.22267
Simons JS, Koutstaal W, Prince S, Wagner AD, Schacter DL (2003) Neural mechanisms of visual object priming: evidence for perceptual and semantic distinctions in fusiform cortex. NeuroImage 19:613–626
Soria Bauser DA, Suchan B (2013) Behavioral and electrophysiological correlates of intact and scrambled body perception. Clin Neurophysiol 124:686–696. doi:10.1016/j.clinph.2012.09.030
Soria Bauser D, Suchan B (2015) Is the whole the sum of its parts? Configural processing of headless bodies in the right fusiform gyrus. Behav Brain Res 281:102–110. doi:10.1016/j.bbr.2014.12.015
Supp GG, Schlogl A, Trujillo-Barreto N, Müller MM, Gruber T (2007) Directed cortical information flow during human object recognition: analyzing induced EEG gamma-band responses in brain’s source space. PLoS One 2:e684. doi:10.1371/journal.pone.0000684
Tanaka K (2003) Columns for complex visual object features in the inferotemporal cortex: clustering of cells with similar but slightly different stimulus selectivities. Cereb Cortex 13:90–99
Thierry G, Pegna AJ, Dodds C, Roberts M, Basan S, Downing PE (2006) An event-related potential component sensitive to images of the human body. NeuroImage 32:871–879
Thoma V, Henson RN (2011) Object representations in ventral and dorsal visual streams: fMRI repetition effects depend on attention and part-whole configuration. NeuroImage 57:513–525. doi:10.1016/j.neuroimage.2011.04.035
Vocks S, Busch M, Gronemeyer D, Schulte D, Herpertz S, Suchan B (2010a) Differential neuronal responses to the self and others in the extrastriate body area and the fusiform body area. Cogn Affect Behav Neurosci 10:422–429. doi:10.3758/cabn.10.3.422
Vocks S, Busch M, Gronemeyer D, Schulte D, Herpertz S, Suchan B (2010b) Neural correlates of viewing photographs of one’s own body and another woman’s body in anorexia and bulimia nervosa: an fMRI study. J Psychiatry Neurosci 35:163–176
Vocks S, Schulte D, Busch M, Gronemeyer D, Herpertz S, Suchan B (2011) Changes in neuronal correlates of body image processing by means of cognitive-behavioural body image therapy for eating disorders: a randomized controlled fMRI study. Psychol Med 41:1651–1663. doi:10.1017/s0033291710002382
Wiggs CL, Martin A (1998) Properties and mechanisms of perceptual priming. Curr Opin Neurobiol 8:227–233
Worsley KJ, Marrett S, Neelin P, Evans AC (1996) Searching scale space for activation in PET images. Hum Brain Mapp 4:74–90. doi:10.1002/(SICI)1097-0193(1996)4:1<74:AID-HBM5>3.0.CO;2-M
Yuval-Greenberg S, Tomer O, Keren AS, Nelken I, Deouell LY (2008) Transient induced gamma-band response in EEG as a manifestation of miniature saccades. Neuron 58:429–441. doi:10.1016/j.neuron.2008.03.027
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Giabbiconi, CM., Jurilj, V., Gruber, T. et al. Steady-state visually evoked potential correlates of human body perception. Exp Brain Res 234, 3133–3143 (2016). https://doi.org/10.1007/s00221-016-4711-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4711-8