Abstract
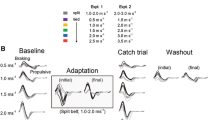
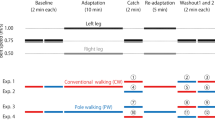
Movements learned in one set of conditions may not generalize to other conditions. For example, practicing walking on a split-belt treadmill subsequently changes coordination between the legs during normal (“tied-belt”) treadmill walking; however, there is limited generalization of these aftereffects to natural walking over the ground. We hypothesized that generalization of split-belt treadmill adaptation to over-ground walking would be improved by maintaining consistency in other task variables, specifically gait speed. This hypothesis was based on our previous finding that treadmill aftereffect size was sensitive to gait speed: Aftereffects were largest when tested on tied-belts running at the same speed as the slower belt during split-belt adaptation. In the present study, healthy adults were assigned to a “slow” or “fast” over-ground walking group. Both groups adapted to split-belts (0.7:1.4 m/s), and treadmill aftereffects were tested on tied-belts at the slow (0.7 m/s) and fast (1.4 m/s) speeds. All participants were subsequently transferred to the over-ground environment. The slow and fast groups walked over-ground at 0.7 and 1.4 m/s, respectively. As in previous work, we found that the size of aftereffects during treadmill walking was speed-dependent, with larger aftereffects occurring at 0.7 m/s compared with 1.4 m/s. However, over-ground walking aftereffects were less sensitive to changes in gait speed. We also found that aftereffects in spatial coordination generalized more to over-ground walking than aftereffects in temporal coordination across all speeds of walking. This suggests that different factors influence aftereffect size in different walking environments and for different measures of coordination.






Similar content being viewed by others
Abbreviations
- AE:
-
Aftereffect
- CF, CS :
-
Catch trial (fast, slow)
- DSF, DSS, DSD:
-
Double support (fast, slow, double support difference)
- OGF, OGS, OG PA:
-
Over-ground walking (fast, slow, post-adaptation)
- PT:
-
Percent transfer
- SLF, SLS, SLD:
-
Step length (fast, slow, step length difference)
- TMF, TMS, TMSB :
-
Treadmill walking (fast, slow, split belt)
References
Arshavsky YI, Gelfand IM, Orlovsky GN, Pavlova GA (1978a) Messages conveyed by descending tracts during scratching in the cat. I. Activity of vestibulospinal neurons. Brain Res 159:99–110
Arshavsky YI, Gelfand IM, Orlovsky GN, Pavlova GA (1978b) Messages conveyed by spinocerebellar pathways during scratching in the cat. I. Activity of neurons of the lateral reticular nucleus. Brain Res 151:479–491
Arshavsky YI, Gelfand IM, Orlovsky GN, Pavlova GA (1978c) Messages conveyed by spinocerebellar pathways during scratching in the cat. II. Activity of neurons of the ventral spinocerebellar tract. Brain Res 151:493–506
Arshavsky YI, Orlovsky GN, Pavlova GA, Perret C (1978d) Messages conveyed by descending tracts during scratching in the cat. II. Activity of rubrospinal neurons. Brain Res 159:111–123
Blau JJ, Stephen DG, Carello C, Turvey MT (2009) Prism adaptation of underhand throwing: rotational inertia and the primary and latent aftereffects. Neurosci Lett 456:54–58. doi:10.1016/j.neulet.2009.03.071
Boyd LA, Winstein CJ (2004) Cerebellar stroke impairs temporal but not spatial accuracy during implicit motor learning. Neurorehabil Neural Repair 18:134–143. doi:10.1177/0888439004269072
Choi JT, Bastian AJ (2007) Adaptation reveals independent control networks for human walking. Nat Neurosci 10:1055–1062. doi:10.1038/nn1930
Choi JT, Vining EP, Reisman DS, Bastian AJ (2009) Walking flexibility after hemispherectomy: split-belt treadmill adaptation and feedback control. Brain 132:722–733. doi:10.1093/brain/awn333
Deweer B (1986) Pretest cuing after forgetting of a food-motivated maze task in rats: synergistic action of context and reinforcement. Anim Learn Behav 14:249–256
Dingwell JB, Marin LC (2006) Kinematic variability and local dynamic stability of upper body motions when walking at different speeds. J Biomech 39:444–452
Forssberg H, Grillner S, Halbertsma J, Rossignol S (1980) The locomotion of the low spinal cat. II. Interlimb coordination. Acta Physiol Scand 108:283–295
Francis JT (2008) Error generalization as a function of velocity and duration: human reaching movements. Exp Brain Res 186:23–37. doi:10.1007/s00221-007-1202-y
Gordon WC (1981) Mechanisms of cue-induced retention enhancement. In: Spear NE, Miller RR (eds) Information processing in animals: memory mechanisms. Erlbaum, Hillsdale, NJ, pp 319–340
Gordon WC, Mccracken KM, Dessbeech N, Mowrer RR (1981) Mechanisms for the cueing phenomenon—the addition of the cueing context to the training memory. Learn Motiv 12:196–211. doi:10.1016/0023-9690(81)90018-7
Grillner S, Halbertsma J, Nilsson J, Thorstensson A (1979) The adaptation to speed in human locomotion. Brain Res 165:177–182
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Stat 6:65–70
Hoogkamer W, Bruijn SM, Potocanac Z, Van Calenbergh F, Swinnen SP, Duysens J (2015) Gait asymmetry during early split-belt walking is related to perception of belt speed difference. J Neurophysiol 114:1705–1712. doi:10.1152/jn.00937.2014
Hwang EJ, Smith MA, Shadmehr R (2006) Adaptation and generalization in acceleration-dependent force fields. Exp Brain Res 169:496–506. doi:10.1007/s00221-005-0163-2
Kitazawa S, Kimura T, Uka T (1997) Prism adaptation of reaching movements: specificity for the velocity of reaching. J Neurosci 17:1481–1492
Krakauer JW, Pine ZM, Ghilardi MF, Ghez C (2000) Learning of visuomotor transformations for vectorial planning of reaching trajectories. J Neurosci 20:8916–8924
Malone LA, Bastian AJ (2010) Thinking about walking: effects of conscious correction versus distraction on locomotor adaptation. J Neurophysiol 103:1954–1962. doi:10.1152/jn.00832.2009
Malone LA, Bastian AJ (2014) Spatial and temporal asymmetries in gait predict split-belt adaptation behavior in stroke. Neurorehabil Neural Repair 28:230–240. doi:10.1177/1545968313505912
Malone LA, Vasudevan EV, Bastian AJ (2011) Motor adaptation training for faster relearning. J Neurosci 31:15136–15143. doi:10.1523/JNEUROSCI.1367-11.2011
Malone LA, Bastian AJ, Torres-Oviedo G (2012) How does the motor system correct for errors in time and space during locomotor adaptation? J Neurophysiol 108:672–683. doi:10.1152/jn.00391.2011
McLean DL, Fetcho JR (2009) Spinal interneurons differentiate sequentially from those driving the fastest swimming movements in larval zebrafish to those driving the slowest ones. J Neurosci 29:13566–13577. doi:10.1523/JNEUROSCI.3277-09.2009
McVea DA, Pearson KG (2007) Long-lasting, context-dependent modification of stepping in the cat after repeated stumbling-corrective responses. J Neurophysiol 97:659–669. doi:10.1152/jn.00921.2006
Mori S, Matsui T, Kuze B, Asanome M, Nakajima K, Matsuyama K (1999) Stimulation of a restricted region in the midline cerebellar white matter evokes coordinated quadrupedal locomotion in the decerebrate cat. J Neurophysiol 82:290–300
Musselman KE, Patrick SK, Vasudevan EV, Bastian AJ, Yang JF (2011) Unique characteristics of motor adaptation during walking in young children. J Neurophysiol 105:2195–2203. doi:10.1152/jn.01002.2010
Orlovskii GN, Severin FV, Shik ML (1966) Locomotion induced by stimulation of the mesencephalon. Dokl Akad Nauk SSSR 169:1223–1226
Reisman DS, Block HJ, Bastian AJ (2005) Interlimb coordination during locomotion: what can be adapted and stored? J Neurophysiol 94:2403–2415. doi:10.1152/jn.00089.2005
Reisman DS, Wityk R, Silver K, Bastian AJ (2007) Locomotor adaptation on a split-belt treadmill can improve walking symmetry post-stroke. Brain 130:1861–1872. doi:10.1093/brain/awm035
Reisman DS, Wityk R, Silver K, Bastian AJ (2009) Split-belt treadmill adaptation transfers to overground walking in persons poststroke. Neurorehabil Neural Repair 23:735–744. doi:10.1177/1545968309332880
Reisman DS, McLean H, Keller J, Danks KA, Bastian AJ (2013) Repeated split-belt treadmill training improves poststroke step length asymmetry. Neurorehabil Neural Repair 27:460–468. doi:10.1177/1545968312474118
Reynolds RF, Bronstein AM (2004) The moving platform aftereffect: limited generalization of a locomotor adaptation. J Neurophysiol 91:92–100. doi:10.1152/jn.00495.2003
Russell DF, Zajac FE (1979) Effects of stimulating Deiters’ nucleus and medial longitudinal fasciculus on the timing of the fictive locomotor rhythm induced in cats by DOPA. Brain Res 177:588–592
Saibene F, Minetti AE (2003) Biomechanical and physiological aspects of legged locomotion in humans. Eur J Appl Physiol 88:297–316. doi:10.1007/s00421-002-0654-9
Shelhamer M, Clendaniel R (2002) Sensory, motor, and combined contexts for context-specific adaptation of saccade gain in humans. Neurosci Lett 332:200–204
Shik ML, Severin FV, Orlovskii GN (1966) Control of walking and running by means of electric stimulation of the midbrain. Biofizika 11:659–666
Taylor JA, Ivry RB (2013) Context-dependent generalization. Front Hum Neurosci 7:171. doi:10.3389/fnhum.2013.00171
Torres-Oviedo G, Bastian AJ (2010) Seeing is believing: effects of visual contextual cues on learning and transfer of locomotor adaptation. J Neurosci 30:17015–17022. doi:10.1523/JNEUROSCI.4205-10.2010
Torres-Oviedo G, Bastian AJ (2012) Natural error patterns enable transfer of motor learning to novel contexts. J Neurophysiol 107:346–356. doi:10.1152/jn.00570.2011
Tulving E, Pearlstone Z (1966) Availability versus accessibility of information in memory for words. J Verbal Learn Verbal Behav 5:381–391
Vasudevan EV, Bastian AJ (2010) Split-belt treadmill adaptation shows different functional networks for fast and slow human walking. J Neurophysiol 103:183–191. doi:10.1152/jn.00501.2009
Vasudevan EV, Bastian AJ, Torres-Oviedo G (2010) Emerging principles in the learning and generalization of new walking patterns. In: Danion F, Latash M (eds) Motor control: theories, experiments, and applications. Oxford University Press, Oxford
Vasudevan EV, Torres-Oviedo G, Morton SM, Yang JF, Bastian AJ (2011) Younger is not always better: development of locomotor adaptation from childhood to adulthood. J Neurosci 31:3055–3065. doi:10.1523/JNEUROSCI.5781-10.2011
Vasudevan EV, Glass RN, Packel AT (2014) Effects of traumatic brain injury on locomotor adaptation. J Neurol Phys Ther 38:172–182. doi:10.1097/NPT.0000000000000049
Acknowledgments
We would like to thank Rebecca Glass and Rachel Snyder for assistance with data collection and Dr. Lisa Muratori for helpful comments. R. J. Hamzey’s current affiliation is the Department of Mechanical Engineering, Boston University, Boston, MA. E. M. Kirk’s current affiliation is Department of Physical Therapy, MGH Institute of Health Professions, Boston, MA.
Funding
This work was funded by an American Heart Association Scientist Development Grant (#12SDG12200001) to E. Vasudevan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no potential conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Hamzey, R.J., Kirk, E.M. & Vasudevan, E.V.L. Gait speed influences aftereffect size following locomotor adaptation, but only in certain environments. Exp Brain Res 234, 1479–1490 (2016). https://doi.org/10.1007/s00221-015-4548-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-015-4548-6