Abstract
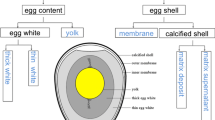
Structurally, egg white (EW) is composed of four distinct layers: outer thin white, thick white, inner thin white and chalaziferous layer (including chalaza cords). In addition, the thick white can be further separated into a gel and a liquid fraction by using ultracentrifugation. There were large differences in the amounts of various ovomucin complex forms between the EW layers, but more or less the order was about the same despite the ovomucin form. The three most ovomucin-rich fractions were: chalaziferous layer > gel fraction of thick EW layer > thick EW layer. Furthermore, each ovomucin fraction was also degraded by using enzymatic hydrolysis. Only three hydrolysed ovomucin fractions were found to contain hemagglutination inhibition activity against Newcastle disease virus, namely thick white, inner thin white and the gel fraction of thick white. Moreover, both thick EW and gel fraction ovomucin showed antiviral activity against avian influenza virus subtype H5. In addition, gel fraction ovomucin also inhibited avian influenza virus subtype H7. All three ovomucin hydrolysates having hemagglutination inhibition activity also contained the highest amount of sialic acid. Structural characterization of ovomucin revealed that it is composed of two subunits, α- and β-ovomucins, as EW protein formerly called α1-ovomucin seemed to be ovostatin. However, it seemed quite possible that ovostatin is associated within β- and α-ovomucins. This interaction might even have some effect on the physical nature of various EW layers.





Similar content being viewed by others
References
Li-Chan ECY, Kim H-O (2008) Structure and chemical composition of eggs. In: Mine Y (ed) Egg bioscience and biotechnology. Wiley, Hoboken, pp 1–95
Kato A, Nakamura R, Sato Y (1970) Studies on changes in stored shell eggs Part VI. Changes in the chemical composition of ovomucin during storage. Agric Biol Chem 34:1009–1013
Burley RW, Vadehra DV (1989) The albumen: chemistry. In: Burley RW, Vadehra DV (eds) The avian egg: chemistry and biology. Wiley, New York, pp 65–128
Hiidenhovi J (2007) Ovomucin. In: Huopalahti R, Lopez-Fandino R, Anton M, Schade R (eds) Bioactive egg compounds. Springer, Berlin, pp 60–68
Omana DA, Wang J, Wu J (2010) Ovomucin—a glycoprotein with promising potential. Trends Food Sci Technol 21:455–463
Ohami H, Ohishi H, Yokota T, Mori T, Watanabe K (1993) Cytotoxic effect of sialoglycoprotein derived from avian egg white ovomucin on the cultured tumor cell. Med Biol 126:19–23
Tsuge Y, Shimoyamada M, Watanabe K (1996) Binding of egg white proteins to viruses. Biosci Biotechnol Biochem 60:1503–1504
Tsuge Y, Shimoyamada M, Watanabe K (1996) Differences in hemagglutination inhibition activity against bovine rotavirus and hen Newcastle disease virus based on the subunits in hen egg white ovomucin. Biosci Biotechnol Biochem 60:1505–1506
Tanizaki H, Tanaka H, Iwata H, Kato A (1997) Activation of macrophages by sulfated glycopeptides in ovomucin, yolk membrane, and chalazae in chicken eggs. Biosci Biotechnol Biochem 61:1883–1889
Watanabe K, Tsuge Y, Shimoyamada M, Ogama N, Ebina T (1998) Antitumor effects of pronase-treated fragments, glycopeptides, from ovomucin in hen egg white in a double grafted tumor system. J Agric Food Chem 46:3033–3038
Brooks J, Hale HP (1961) The mechanical properties of the thick white of the hen’s egg II. The relation between rigidity and composition. Biochim Biophys Acta 46:289–301
Donovan JW, Davis JG, White LM (1970) Chemical and physical characterization of ovomucin, a sulfated glycoprotein complex from chicken eggs. Biochim Biophys Acta 207:190–201
Robinson DS, Monsey JB (1971) Studies on the composition of egg-white ovomucin. Biochem J 121:537–547
Guérin C, Brulé G (1992) Fractionnement de trois protéines du blanc d’oeuf. Sci Aliments 12:705–720
Omana DA, Wu J (2009) A new method of separating ovomucin from egg white. J Agric Food Chem 57:3596–3603
Shan Y, Huang X, Ma M, Miao F (2012) Role of magnesium chloride on the purity and activity of ovomucin in during the isolation process. J Biol Macromol 50:421–427
Guerin-Dubiard C, Brule G (1994) Essai de solubilisation de l’ovomucine par voie enzymatique. Ind Aliment Agric 111:701–707
Moreau S (1996) Contribution a l’etude de la structure de l’ovomucine et sa valorisation. Ph.D. Dissertation, L’Ecole Nationale Superieure Agronomique de Rennes, France
Moreau S, Nau F, Piot M, Guerin C, Brule G (1997) Hydrolysis of hen egg white ovomucin. Z Lebensm Unters Forsch A 205:329–334
Hiidenhovi J, Hietanen A, Mäkinen J, Huopalahti R, Ryhänen E-L (2000) Enzymatic hydrolysis of ovomucin: comparison of different proteases. In: Fourteenth forum for applied biotechnology proceedings part II. Brugge, Belgium, pp 535–538
Hiidenhovi J, Rinta-Koski M, Hietanen A, Mantere-Alhonen S, Huopalahti R, Ryhänen E-L (2000) Hen egg white ovomucin, a potential ingredient for functional foods. In: Buttriss J, Saltmarsh M (eds) Functional foods II: claims and evidence. The Royal Society of Chemistry, Cambridge, pp 197–199
Gottschalk A, Lind PE (1949) Ovomucin, a substrate for the enzyme of influenza virus I. Ovomucin as an inhibitor of haemagglutination by heated Lee virus. Br J Exp Pathol 30:85–92
Gottschalk A, Lind PE (1949) Product of interaction between influenza virus enzyme and ovomucin. Science 64:232–233
Lanni F, Sharp DG, Eckert EA, Dillon ES, Beard D, Beard JW (1949) The egg white inhibitor of influenza virus hemagglutination I. Preparation and properties of semipurified inhibitor. J Biol Chem 179:1275–1287
Dan Z (2013) Research on the preparation, solubility enhancement and anti-infection of ovomucin from hen egg, Ph.D. Dissertation, Huazhong Agricultural University, China
Hiidenhovi J (2015) Isolation and characterization of ovomucin—a bioactive agent of egg white, Ph.D. Dissertation, University of Turku, Finland
Hiidenhovi J, Aro HS, Kankare V (1999) Separation of ovomucin subunits by gel filtration: enhanced resolution of subunits by using dual-column system. J Agric Food Chem 47:1004–1008
Hiidenhovi J, Mäkinen J, Huopalahti R, Ryhänen E-L (2002) Comparison of different egg albumen fractions as sources of ovomucin. J Agric Food Chem 50:2840–2845
Nielsen KL, Sottrup-Jensen L, Nagase H, Thøgersen HC, Etzerodt M (1994) Amino acid sequence of hen ovomacroglobulin (ovostatin) deduced from cloned cDNA. DNA Seq 5:111–119
Jourdian GW, Dean L, Roseman S (1971) The sialic acids XI. A perodate-resorcinol method for the quantitative estimation of free sialic acids and their glycosides. J Biol Chem 246:430–435
Church FC, Swaisgood HE, Porter DH, Catignáni GL (1983) Spectrophotometric assay using ο-phthaldialdehyde for determination of proteolysis in milk and isolated milk proteins. J Dairy Sci 66:1219–1227
Frister H, Meisel H, Schlimme E (1988) OPA method modified by use of N,N-dimethyl-2-mercaptoethylammonium chloride as thiol component. Fres J Anal Chem 330:631–633
Anonymous (1992) Directive 92/66/EEC Annex III, chapter 5 and 6
Kato A, Nakamura R, Sato Y (1971) Studies on changes in stored shell eggs: part VII. Changes in physicochemical properties of ovomucin solubilized by treatment with mercaptoethanol during storage. Agric Biol Chem 35:351–356
Itoh T, Miyaki J, Sugawara H, Adachi S (1987) Studies on the characterization of ovomucin and chalaza of the hen’s egg. J Food Sci 52:1518–1521
Hammershøj M, Nebel C, Carstens JH (2008) Enzymatic hydrolysis of ovomucin and effect on foaming properties. Food Res Int 41:522–531
Omana DA, Wu J (2009) Effect of different concentrations of calcium chloride and potassium chloride on egg white proteins during isoelectric precipitation of ovomucin. Poult Sci 88:2224–2234
Tsuge Y, Shimoyamada M, Watanabe K (1997) Structural features of Newcastle disease virus- and anti-ovomucin antibody-binding glycopeptides from pronase-treated ovomucin. J Agric Food Chem 45:2393–2398
Wu J, Omana DA (2009) The methods for isolating high purity ovomucin. WO Patent, WO 2009/138875 A1
Offengenden M, Fentabil MA, Wu J (2011) N-glycosylation of ovomucin from egg white. Glycoconj J 28:113–123
Offengenden M (2011) N-glycosylation and gelling properties of ovomucin from egg white. M.Sc. Thesis, University of Alberta, Canada
Mann K (2007) The chicken egg white proteome. Proteomics 7:3558–3568
Mann K, Mann M (2011) In-depth analysis of the chicken egg white proteome using an LTQ Orbitrap Velos. Proteome Sci 9. doi:101186/1477-5956-9-7
Jay GD, Culp DJ, Jahnke MR (1990) Silver staining of extensively glycosylated proteins on sodium dodecyl sulfate-polyacrylamide gels: enhancement by carbohydrate-binding dyes. Anal Biochem 185:324–330
Watanabe K, Shimoyamada M, Onizuka T, Akiyama H, Niwa M, Ido T, Tsuge Y (2004) Amino acid sequence of α-ovomucin in hen egg white ovomucin deduced from cloned cDNA. DNA Seq 15:251–261
Miller HT, Feeney RE (1965) The physical and chemical properties of an immunologically cross-reacting protein from avian egg whites. Biochemistry 5:952–958
Donovan JW, Mapes CJ, Davis JG, Hamburg RD (1969) Dissociation of chicken egg-white macroglobulin into subunits in acid hydrodynamic, spectrophotometric, and optical rotatory measurements. Biochemistry 8:4190–4199
Kitamoto T, Nakashima M, Ikai A (1982) Hen egg white ovomacroglobulin has a protease inhibitory activity. J Biochem 92:1679–1682
Nagase H, Harris ED Jr, Woessner JF Jr, Brew K (1983) Ovostatin: a novel proteinase inhibitor from chicken egg white. I. Purification, physicochemical properties, and tissue distribution of ovostatin. J Biol Chem 258:7481–7489
Hayakawa S, Sato Y (1978) Subunit structures of sonicated α- and β-ovomucin and their molecular weights estimated by sedimentation equilibrium. Agric Biol Chem 42:957–961
Robinson DS, Monsey JB (1975) The composition and proposed subunit structure of egg-white β-ovomucin. The isolation of an unreduced soluble ovomucin. Biochem J 147:55–62
Watanabe K, Tsuge Y, Shimoyamada M (1998) Binding activities of pronase-treated fragments from egg white ovomucin with anti-ovomucin antibodies and Newcastle disease virus. J Agric Food Chem 46:4501–4506
Kato A, Imoto T, Yagishita K (1975) The binding groups in ovomucin-lysozyme interaction. Agric Biol Chem 39:541–544
Kato A, Yoshida K, Matsudomi N, Kobayashi K (1976) The interaction between ovomucin and egg white proteins. Agric Biol Chem 40:2361–2366
Damodaran S, Anand K, Razumovsky L (1998) Competitive adsorption of egg white proteins at the air-water interface: direct evidence for electrostatic complex formation between lysozyme and other egg white proteins in the interface. J Agric Food Chem 46:872–876
Garipaldi JA, Donovan JW, Davis JG, Cimino SL (1968) Heat denaturation of the ovomucin-lysozyme electrostatic complex-a source of damage to the whipping properties of pasteurized egg white. J Food Sci 33:514–524
Miller SM, Kato A, Nakai S (1982) Sedimentation equilibrium study of the interaction between egg white lysozyme and ovomucin. J Agric Food Chem 30:1127–1132
Rabouille C, Aon MA, Thomas D (1989) Interactions involved in ovomucin gel-forming properties: a rheological-biochemical approach. Arch Biochem Biophys 270:495–503
Zhang J (2012) Protein-protein interactions in salt solutions. In: Cai W (ed) Protein–protein interactions—computational and experimental tools. InTech, doi:10.5772/38056. Available from: http://www.intechopen.com/books/protein-protein-interactions-computational-and-experimental-tools/protein-protein-interactions-in-salt-solutions
Robinson DS (1972) Egg white glycoproteins and the physical properties of egg white. In: Freeman BM, Lake PE (eds) Egg formation and production. British Poultry Science Ltd, Edinburgh, pp 65–86
Sauveur B (1971) Répartion des électrolytes et du glucose dans l’oeuf conservé sous 0 ou 2 P. 100 de gaz carbonique. Ann Biol Anim Biochem Biophys 11:625–643
Sharp PF (1929) The pH of the white as an important factor influencing the keeping quality of hens’ eggs. Science 69:278–280
Kato A, Ogata S, Matsudomi N, Kobayashi K (1981) A comparative study of aggregated and disaggregated ovomucin during egg white thinning. J Agric Food Chem 29:821–823
Lyndrup ML (1973) The isolation and fractionation of chicken egg white ovomucin. Prep Biochem 3:135–148
Toussant MJ, Latshaw JD (1999) Ovomucin content and composition in chicken eggs with different interior quality. J Sci Food Agric 79:1666–1670
Skala JH, Swanson MH (1962) Studies of variation in initial quality of chicken eggs 2. Chemical properties of the albumen. Poult Sci 41:1537–1545
Balls AK, Hoover SM (1940) Behavior of ovomucin in the liquefaction of egg white. Ind Eng Chem 32:594–596
Robinson DS, Monsey JB (1969) A reduced ovomucin-reduced lysozyme complex from egg white. Biochem J 115:64P
Robinson DS, Monsey JB (1972) Changes in the composition of ovomucin during liquefaction of thick egg white. J Sci Food Agr 23:29–38
Robinson DS, Monsey JB (1972) Changes in the composition of ovomucin during liquefaction of thick egg white: the effect of ionic strength and magnesium salts. J Sci Food Agric 23:893–904
Adachi N, Azuma J, Janado M, Onodera K (1973) Solubilisation and characterisation of ovomucin without chemical modification. Agric Biol Chem 37:2175–2180
Baliga BR, Kadkol SB, Lahiry NL (1970) Thinning of thick albumen in shell eggs—changes in ovomucin. Poult Sci 49:466–473
Sato Y, Hayakawa S (1977) Further inspection for the structure of thick egg white. J Agric Chem Soc Jpn 5:47–51
Nau F, Gestin L, Protais J, Awadé AC, Thapon JL (1996) Etude comparée des propriétés fonctionnelles et de la composition des fractions épaisse et liquide du blanc d´oeuf de poule. Ind Aliment Agric 113:5–10
Kato A, Nakamura R, Sato Y (1970) Studies on changes in stored shell eggs: Part V the difference in the chemical and physicochemical properties of ovomucin B between the thick and thin white. Agric Biol Chem 34:854–859
Baliga BR, Kadkol SB, Lahiry NL (1968) Changes in ovomucin concentration during thinning of thick white in eggs. J Food Sci Technol (Mysore) 5:71
Sato Y, Kato A (1980) Chemical composition of the chalazae and their deterioration during storage of egg. Poult Sci 59:1416–1422
Hayakawa S, Sato Y (1977) Physicochemical identity of α-ovomucins or β-ovomucins obtained from the sonicated insoluble and soluble ovomucins. Agric Biol Chem 41:1185–1191
Kato A, Sato Y (1971) The separation and characterization of carbohydrate rich component from ovomucin in chicken eggs. Agric Biol Chem 35:439–440
Kato A, Sato Y (1972) The release of carbohydrate rich component from ovomucin gel during storage. Agric Biol Chem 36:831–836
Hayakawa S, Sato Y (1976) Studies on dissociation of the soluble ovomucin by sonication. Agric Biol Chem 40:2397–2404
Kato A, Ogino K, Matsudomi N, Kobayashi K (1977) Separation of ovomucin into carbohydrate rich and poor components by chromatography on lysozyme-Sepharose 4B. Agric Biol Chem 41:1925–1929
Hayakawa S, Sato Y (1978) Viscometric behaviour of the soluble ovomucin. Agric Biol Chem 42:2025–2029
Redfield C, Dobson CM (1988) Sequential proton 1H NMR assignments and secondary structure of hen egg white lysozyme in solution. Biochem 27:122–136
Tsuge Y, Shimoyamada M, Watanabe K (1997) Bindings of ovomucin to Newcastle disease virus and anti-ovomucin antibodies and its heat stability based on binding abilities. J Agric Food Chem 45:4629–4634
Visvanatham K, Chandrasekaran A, Srinivasan A, Raman R, Sasisekharan V, Sasisekharan R (2010) Glycans as receptors for influenza pathogenesis. Glycoconj J 27:561–570
Acknowledgements
We are grateful to Mrs. Auli Lehtonen and Mrs. Marja Kallioinen for their skilful technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Rights and permissions
About this article
Cite this article
Hiidenhovi, J., Ek-Kommonen, C., Järvenpää, E. et al. Characterization of crude ovomucins obtained from various egg white layers. Eur Food Res Technol 242, 233–246 (2016). https://doi.org/10.1007/s00217-015-2535-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-015-2535-6