Abstract
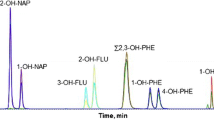
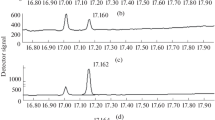
A new gas chromatography–tandem mass spectrometry method for the determination of mono- and dihydroxylated polycyclic aromatic hydrocarbon metabolites (OH-PAHs and diol-PAHs) in urine was developed and validated. Various sample preparation procedures were compared, namely liquid–liquid extraction (LLE), dispersive solid-phase extraction (dSPE), and SPE, alone or combined. A novel two-stage derivatization approach using 2 silylation reagents was developed, and an experimental procedure design was used to optimize the programmed temperature vaporization–solvent vent injection (PTV-SV) GC parameters. The method focused on 11 target compounds resulting from four- to five-ring suspected carcinogenic PAHs. SPE was identified as an acceptable and more convenient extraction method for all tested metabolites, with extraction rates ranging from 63 to 86% and relative standard deviations lower than 20%. The two-stage derivatization approach successfully allowed first the derivatization of OH-PAHs by MTBSTFA (N-tert-butyldimethylsilyl-N-methyltrifluoroacetamide) and then diol-PAHs by BSTFA (N,O-bis(trimethylsilyl)trifluoroacetamide) in a single run. The limits of quantification were in the range of 0.01–0.02 μg l−1 for OH-PAHs and 0.02–0.2 μg l−1 for diol-PAHs. The intra- and interday precisions were lower than 10%. The method was applied to determine PAH metabolites in urine collected at the beginning and at the end of the working week from 6 workers involved in aluminum production. The mean diol-PAH levels at the end of the week were 10 to 20 times higher (0.86–2.34 μg g−1 creatinine) than those of OH-PAHs (0.03–0.30 μg g−1). These results confirmed the usefulness of this new analytical technique for detecting and characterizing metabolic patterns of PAHs in urine and assessing carcinogenic occupational exposures.
Graphical abstract





Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this article will be made available by the authors within respect of General Data Protection Regulation.
References
Menzie CA, Potocki BB, Santodonato J. Exposure to carcinogenic PAHs in the environment. Environ Sci Technol. 1992;26(7):1278–84. https://doi.org/10.1021/es00031a002.
Zhong Y, Carmella SG, Upadhyaya P, Hochalter JB, Rauch D, Oliver A, et al. Immediate consequences of cigarette smoking: rapid formation of polycyclic aromatic hydrocarbon diol epoxides. Chem Res Toxicol. 2011;24(2):246–52. https://doi.org/10.1021/tx100345x.
Bandowe BAM, Meusel H. Nitrated polycyclic aromatic hydrocarbons (nitro-PAHs) in the environment - a review. Sci Total Environ. 2017;581–582:237–57. https://doi.org/10.1016/j.scitotenv.2016.12.115.
Mueller A, Ulrich N, Hollmann J, Zapata Sanchez CE, Rolle-Kampczyk UE, von Bergen M. Characterization of a multianalyte GC-MS/MS procedure for detecting and quantifying polycyclic aromatic hydrocarbons (PAHs) and PAH derivatives from air particulate matter for an improved risk assessment. Environ Pollut. 2019;255(Pt 2):112967. https://doi.org/10.1016/j.envpol.2019.112967.
Olsson AC, Fevotte J, Fletcher T, Cassidy A, Mannetje TA, Zaridze D, et al. Occupational exposure to polycyclic aromatic hydrocarbons and lung cancer risk: a multicenter study in Europe. Occup Environ Med. 2010;67(2):98–103. https://doi.org/10.1136/oem.2009.046680.
Bosetti C, Boffetta P, La Vecchia C. Occupational exposures to polycyclic aromatic hydrocarbons, and respiratory and urinary tract cancers: a quantitative review to 2005. Ann Oncol. 2007;18(3):431–46. https://doi.org/10.1093/annonc/mdl172.
Boffetta P, Jourenkova N, Gustavsson P. Cancer risk from occupational and environmental exposure to polycyclic aromatic hydrocarbons. Cancer Causes Control. 1997;8(3):444–72. https://doi.org/10.1023/a:1018465507029.
Xu X, Cook RL, Ilacqua VA, Kan H, Talbott EO, Kearney G. Studying associations between urinary metabolites of polycyclic aromatic hydrocarbons (PAHs) and cardiovascular diseases in the United States. Sci Total Environ. 2010;408(21):4943–8. https://doi.org/10.1016/j.scitotenv.2010.07.034.
Han Y, Xia Y, Zhu P, Qiao S, Zhao R, Jin, et al. Reproductive hormones in relation to polycyclic aromatic hydrocarbon (PAH) metabolites among non-occupational exposure of males. Sci Total Environ. 2010;408(4):768–73. https://doi.org/10.1016/j.scitotenv.2009.11.021.
Zhang H, Han Y, Qiu X, Wang Y, Li W, Liu J, et al. Association of internal exposure to polycyclic aromatic hydrocarbons with inflammation and oxidative stress in prediabetic and healthy individuals. Chemosphere. 2020;253:126748. https://doi.org/10.1016/j.chemosphere.2020.126748.
Farzan SF, Chen Y, Trachtman H, Trasande L. Urinary polycyclic aromatic hydrocarbons and measures of oxidative stress, inflammation and renal function in adolescents: NHANES 2003–2008. Environ Res. 2016;144(Pt A):149–57. https://doi.org/10.1016/j.envres.2015.11.012.
Kim KH, Jahan SA, Kabir E, Brown RJ. A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ Int. 2013;60:71–80. https://doi.org/10.1016/j.envint.2013.07.019.
Cathey AL, Watkins DJ, Rosario ZY, Vélez Vega CM, Loch-Caruso R, Alshawabkeh AN, et al. Polycyclic aromatic hydrocarbon exposure results in altered CRH, reproductive, and thyroid hormone concentrations during human pregnancy. Sci Total Environ. 2020;749:141581. https://doi.org/10.1016/j.scitotenv.2020.141581.
Grimmer G, Jacob J, Dettbarn G, Naujack KW. Determination of urinary metabolites of polycyclic aromatic hydrocarbons (PAH) for the risk assessment of PAH-exposed workers. Int Arch Occup Environ Health. 1997;69(4):231–9. https://doi.org/10.1007/s004200050141.
Luo K, Hochalter JB, Carmella SG, Hecht SS. Quantitation of phenanthrene dihydrodiols in the urine of smokers and non-smokers by gas chromatography-negative ion chemical ionization-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2020;1141:122023. https://doi.org/10.1016/j.jchromb.2020.122023.
Smith CJ, Huang W, Walcott CJ, Turner W, Grainger J, Patterson DG Jr. Quantification of monohydroxy-PAH metabolites in urine by solid-phase extraction with isotope dilution-GC-MS. Anal Bioanal Chem. 2002;372(1):216–20. https://doi.org/10.1007/s00216-001-1123-8.
Romanoff LC, Li Z, Young KJ, Blakely NC 3rd, Patterson DG Jr, Sandau CD. Automated solid-phase extraction method for measuring urinary polycyclic aromatic hydrocarbon metabolites in human biomonitoring using isotope-dilution gas chromatography high-resolution mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2006;835(1–2):47–54. https://doi.org/10.1016/j.jchromb.2006.03.004.
Campo L, Rossella F, Fustinoni S. Development of a gas chromatography/mass spectrometry method to quantify several urinary monohydroxy metabolites of polycyclic aromatic hydrocarbons in occupationally exposed subjects. J Chromatogr B Analyt Technol Biomed Life Sci. 2008;875(2):531–40. https://doi.org/10.1016/j.jchromb.2008.10.017.
Grova N, Salquèbre G, Appenzeller BM. Gas chromatography-tandem mass spectrometry analysis of 52 monohydroxylated metabolites of polycyclic aromatic hydrocarbons in hairs of rats after controlled exposure. Anal Bioanal Chem. 2013;405(27):8897–911. https://doi.org/10.1007/s00216-013-7317-z.
Motorykin O, Schrlau J, Jia Y, Harper B, Harris S, Harding A, et al. Determination of parent and hydroxy PAHs in personal PM2.s and urine samples collected during Native American fish smoking activities. Sci Total Environ. 2015;505:694–703. https://doi.org/10.1016/j.scitotenv.2014.10.051.
Lankova D, Urbancova K, Sram RJ, Hajslova J, Pulkrabova J. A novel strategy for the determination of polycyclic aromatic hydrocarbon monohydroxylated metabolites in urine using ultra-high-performance liquid chromatography with tandem mass spectrometry. Anal Bioanal Chem. 2016;408(10):2515–25. https://doi.org/10.1007/s00216-016-9350-1.
Barbeau D, Lutier S, Marques M, Persoons R, Maitre A. Comparison of gaseous polycyclic aromatic hydrocarbon metabolites according to their specificity as biomarkers of occupational exposure: selection of 2-hydroxyfluorene and 2-hydroxyphenanthrene. J Hazard Mater. 2017;332:185–94. https://doi.org/10.1016/j.jhazmat.2017.03.011.
Seidel A, Spickenheuer A, Straif K, Rihs HP, Marczynski B, Scherenberg M, et al. New biomarkers of occupational exposure to polycyclic aromatic hydrocarbons. J Toxicol Environ Health A. 2008;71(11–12):734–45. https://doi.org/10.1080/15287390801985265.
Jonsson G, Taban IC, Jørgensen KB, Sundt RC. Quantitative determination of de-conjugated chrysene metabolites in fish bile by HPLC-fluorescence and GC--MS. Chemosphere. 2004;54(8):1085–97. https://doi.org/10.1016/j.chemosphere.2003.09.026.
Moreau M, Bouchard M. Comparison of the kinetics of various biomarkers of benzo[a]pyrene exposure following different routes of entry in rats. J Appl Toxicol. 2015;35(7):781–90. https://doi.org/10.1002/jat.3070.
Schummer C, Delhomme O, Appenzeller BM, Wennig R, Millet M. Comparison of MTBSTFA and BSTFA in derivatization reactions of polar compounds prior to GC/MS analysis. Talanta. 2009;77(4):1473–82. https://doi.org/10.1016/j.talanta.2008.09.043.
Pleil JD. Imputing defensible values for left-censored “below level of quantification” (LoQ) biomarker measurements. J. Breath Res. 2016;10(4):045001. https://doi.org/10.1088/1752-7155/10/4/045001.
Li Z, Romanoff LC, Trinidad DA, Hussain N, Jones RS, Porter EN, et al. Measurement of urinary monohydroxy polycyclic aromatic hydrocarbons using automated liquid-liquid extraction and gas chromatography/isotope dilution high-resolution mass spectrometry. Anal Chem. 2006;78(16):5744–51. https://doi.org/10.1021/ac0606094.
Urbancova K, Dvorakova D, Gramblicka T, Sram RJ, Hajslova J, Pulkrabova J. Comparison of polycyclic aromatic hydrocarbon metabolite concentrations in urine of mothers and their newborns. Sci Total Environ. 2020;723:138116. https://doi.org/10.1016/j.scitotenv.2020.138116.
Woudneh MB, Benskin JP, Grace R, Hamilton MC, Magee BH, Hoeger GC, et al. Quantitative determination of hydroxy polycylic aromatic hydrocarbons as a biomarker of exposure to carcinogenic polycyclic aromatic hydrocarbons. J Chromatogr A. 2016;1454:93–100. https://doi.org/10.1016/j.chroma.2016.05.057.
Li Z, Romanoff LC, Trinidad DA, Pittman EN, Hilton D, Hubbard K, et al. Quantification of 21 metabolites of methylnaphthalenes and polycyclic aromatic hydrocarbons in human urine. Anal Bioanal Chem. 2014;406(13):3119–29. https://doi.org/10.1007/s00216-014-7676-0.
Gaudreau É, Bérubé R, Bienvenu JF, Fleury N. Stability issues in the determination of 19 urinary (free and conjugated) monohydroxy polycyclic aromatic hydrocarbons. Anal Bioanal Chem. 2016;408(15):4021–33. https://doi.org/10.1007/s00216-016-9491-2.
Kloos DP, Gay E, Lingeman H, Bracher F, Müller C, Mayboroda OA, et al. Comprehensive gas chromatography-electron ionisation mass spectrometric analysis of fatty acids and sterols using sequential one-pot silylation: quantification and isotopologue analysis. Rapid Commun Mass Spectrom. 2014;28(13):1507–14. https://doi.org/10.1002/rcm.6923.
Yoon HR. Determination of plasma dibasic amino acids following trimethylsilyl-trifluoroacyl derivatization using gas chromatography-mass spectrometry. Arch Pharm Res. 2013;36(3):366–73. https://doi.org/10.1007/s12272-013-0038-1.
Box GEP, Wilson KB. On the experimental attainment of optimum conditions. Journal of Royal Statistical Society B (Methodological). 1951;13(1):1–45.
Gómez-Ruiz JA, Cordeiro F, López P, Wenzl T. Optimisation and validation of programmed temperature vaporization (PTV) injection in solvent vent mode for the analysis of the 15+1 EU-priority PAHs by GC-MS. Talanta. 2009;80(2):643–50. https://doi.org/10.1016/j.talanta.2009.07.041.
Barbeau D, Maître A, Marques M. Highly sensitive routine method for urinary 3-hydroxybenzo[a]pyrene quantitation using liquid chromatography-fluorescence detection and automated off-line solid phase extraction. Analyst. 2011;136(6):1183–91. https://doi.org/10.1039/c0an00428f.
Barbeau D, Lutier S, Choisnard L, Marques M, Persoons R, Maitre A. Urinary trans-anti-7,8,9,10-tetrahydroxy-7,8,9,10-tetrahydrobenzo(a)pyrene as the most relevant biomarker for assessing carcinogenic polycyclic aromatic hydrocarbons exposure. Environ Int. 2018;112:147–55. https://doi.org/10.1016/j.envint.2017.12.012.
Grova N, Faÿs F, Hardy EM, Appenzeller BMR. New insights into urine-based assessment of polycyclic aromatic hydrocarbon-exposure from a rat model: identification of relevant metabolites and influence of elimination kinetics. Environ Pollut. 2017;228:484–95. https://doi.org/10.1016/j.envpol.2017.03.060.
Ramesh A, Walker SA, Hood DB, Guillen MD, Schneider K, Weyand EH. Bioavailability and risk assessment of orally ingested polycyclic aromatic hydrocarbons. Int J Toxicol. 2004;23:301–33.
Li Z, Sandau CD, Romanoff LC, Caudill SP, Sjodin A, Needham LL, Patterson DG Jr. Concentration and profile of 22 urinary polycyclic aromatic hydrocarbon metabolites in the US population. Environ Res. 2008;107(3):320–31. https://doi.org/10.1016/j.envres.2008.01.013.
Funding
This study was funded by the French sanitary agency ANSES (Agence nationale de sécurité sanitaire de l’alimentation, de l’environnement et du travail), Project n° 2014-1-176.
Author information
Authors and Affiliations
Contributions
Marie Marques and Renaud Persoons contributed to the analysis and interpretation of the results. All authors listed have made substantial, direct, and intellectual contributions to the work and approved it for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Informed consent
Informed consent was obtained from all participants for inclusion in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Marques, M., Maitre, A., Choisnard, L. et al. Simultaneous analysis of PAH urinary mono- and dihydroxylated metabolites by GC-MS-MS following SPE and two-stage derivatization. Anal Bioanal Chem 413, 6823–6835 (2021). https://doi.org/10.1007/s00216-021-03638-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-021-03638-4