Abstract
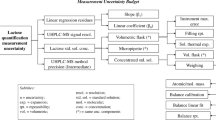
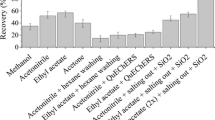
A robust liquid chromatography-tandem mass spectrometry method was developed and comprehensively validated for the quantification of cefquinome considering the changing matrix composition from bovine colostrum to raw milk. Sample preparation consisted of addition of isotopically labeled cefquinome internal standard prior to protein precipitation of 2 g colostrum or milk followed by solid-phase extraction. A wide concentration range from 1 to 5000 ng cefquinome per gram of colostrum or milk was quantified using a 3200 QTRAP tandem mass spectrometer in positive ionization mode with electrospray ionization. Validation was performed according to the European Commission Decision 2002/657/EC guidelines. Matrix-comprehensive in-house validation included analytical limits CCα and CCβ, recovery, precision and calibration curves with prediction intervals, storage conditions, and evaluation of robustness based on factorial effect analysis. The detection limit was 0.2 ng cefquinome per gram of colostrum or milk. Recovery was between 98.4 and 99.4% for cefquinome concentrations from 4 to 240 ng/g. None of the investigated validation factors (matrix, storage of extracts, lot of SPE cartridges, and operators) exerted an influence higher than ± 3.2%, indicating that these factors make relatively low contributions to the respective combined measurement uncertainties. The comprehensively validated method enables routine residue control purposes and to monitor the pharmacokinetics of cefquinome in bovine colostrum and raw milk. In particular, residue depletion curves of cefquinome from high concentrations in first milking after treatment to concentrations far below the maximum residue limit can be measured.




Similar content being viewed by others
References
Prescott JF. Beta-lactam antibiotics: cephalosporins. In: Giguère S, Prescott JF, Baggot JD, Walker RD, Dowling PM, editors. Antimicrobial therapy in veterinary medicine. 4th ed. Oxford: Wiley-Blackwell; 2006. p. 139–57.
WHO. WHO model list of essential medicines. In: 20; 2017.
Commission Regulation (EU) No. 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin.
Bachmann J, Helmschrodt C, Richter A, Heuwieser W, Bertulat S. Residue concentration of cefquinome after intramammary dry cow therapy and short dry periods. J Dairy Sci. https://doi.org/10.3168/jds.2017-13826.
Samanidou VF, Karageorgou EG. An overview of the use of monoliths in sample preparation and analysis of milk. J Sep Sci. 2011;34(16–17):2013–25. https://doi.org/10.1002/jssc.201100101.
Trufelli H, Palma P, Famiglini G, Cappiello A. An overview of matrix effects in liquid chromatography-mass spectrometry. Mass Spectrom Rev. 2011;30(3):491–509. https://doi.org/10.1002/mas.20298.
Rejczak T, Tuzimski T. QuEChERS-based extraction with dispersive solid phase extraction clean-up using PSA and ZrO2-based sorbents for determination of pesticides in bovine milk samples by HPLC-DAD. Food Chem. 2017;217:225–33. https://doi.org/10.1016/j.foodchem.2016.08.095.
Stockler RM, Morin DE, Lantz RK, Hurley WL, Constable PD. Effect of milk fraction on concentrations of cephapirin and desacetylcephapirin in bovine milk after intramammary infusion of cephapirin sodium. J Vet Pharmacol Ther. 2009;32(4):345–52. https://doi.org/10.1111/j.1365-2885.2008.01048.x.
Becker M, Zittlau E, Petz M. Residue analysis of 15 penicillins and cephalosporins in bovine muscle, kidney and milk by liquid chromatography–tandem mass spectrometry. Anal Chim Acta. 2004;520(1):19–32. https://doi.org/10.1016/j.aca.2004.04.022.
Hou XL, Wu YL, Lv Y, Xu XQ, Zhao J, Yang T. Development and validation of an ultra high performance liquid chromatography tandem mass spectrometry method for determination of 10 cephalosporins and desacetylcefapirin in milk. J Chromatogr B Analyt Technol Biomed Life Sci. 2013;931:6–11. https://doi.org/10.1016/j.jchromb.2013.05.006.
Commission Decision 2002/657/EC of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results (2002). Off J Eur Commun L221:8.
Junza A, Amatya R, Barron D, Barbosa J. Comparative study of the LC-MS/MS and UPLC-MS/MS for the multi-residue analysis of quinolones, penicillins and cephalosporins in cow milk, and validation according to the regulation 2002/657/EC. J Chromatogr B Analyt Technol Biomed Life Sci. 2011;879(25):2601–10. https://doi.org/10.1016/j.jchromb.2011.07.018.
Jank L, Martins MT, Arsand JB, Hoff RB, Barreto F, Pizzolato TM. High-throughput method for the determination of residues of beta-lactam antibiotics in bovine milk by LC-MS/MS. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2015;32(12):1992–2001. https://doi.org/10.1080/19440049.2015.1099745.
Baeza AN, Urraca JL, Chamorro R, Orellana G, Castellari M, Moreno-Bondi MC. Multiresidue analysis of cephalosporin antibiotics in bovine milk based on molecularly imprinted polymer extraction followed by liquid chromatography-tandem mass spectrometry. J Chromatogr A. 2016;1474:121–9. https://doi.org/10.1016/j.chroma.2016.10.069.
Aust V, Knappstein K, Kunz HJ, Kaspar H, Wallmann J, Kaske M. Feeding untreated and pasteurized waste milk and bulk milk to calves: effects on calf performance, health status and antibiotic resistance of faecal bacteria. J Anim Physiol Anim Nutr (Berl). 2013;97(6):1091–103. https://doi.org/10.1111/jpn.12019.
Donaldson SC, Straley BA, Hegde NV, Sawant AA, DebRoy C, Jayarao BM. Molecular epidemiology of ceftiofur-resistant Escherichia coli isolates from dairy calves. Appl Environ Microbiol. 2006;72(6):3940–8. https://doi.org/10.1128/aem.02770-05.
Julicher B, Gowik P, Uhlig S. Assessment of detection methods in trace analysis by means of a statistically based in-house validation concept. Analyst. 1998;123(2):173–9. https://doi.org/10.1039/A707281C.
Julicher B, Gowik P, Uhlig S. A top-down in-house validation based approach for the investigation of the measurement uncertainty using fractional factorial experiments. Analyst. 1999;124(4):537–45. https://doi.org/10.1039/A807077F.
Hellmann K, Radeloff I. Guidance for industry: good clinical practice. International Cooperation on Harmonisation of Technical Requirements for Registration of Veterinary Medicinal Products (VICH). VICH, Brussels, Belgium. 2000.
Schmidt KS, Stachel CS. In-house validation and factorial effect analysis of a liquid chromatography–tandem mass spectrometry method for the determination of corticosteroids in bovine and porcine muscle tissue. Anal Bioanal Chem. 2013;405(19):6287–97. https://doi.org/10.1007/s00216-013-7052-5.
Schmidt KS. In-house validation and factorial effect analysis of a liquid chromatography–tandem mass spectrometry method for the determination of thyreostats in bovine blood plasma. Anal Bioanal Chem. 2014;406(3):735–43. https://doi.org/10.1007/s00216-013-7517-6.
Schmidt KS, Mankertz J. In-house validation of a liquid chromatography–tandem mass spectrometry method for the determination of selective androgen receptor modulators (SARMS) in bovine urine. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2018;35(7):1292–304. https://doi.org/10.1080/19440049.2018.1471222.
Bogialli S, Curini R, Di Corcia A, Lagana A, Mele M, Nazzari M. Simple confirmatory assay for analyzing residues of aminoglycoside antibiotics in bovine milk: hot water extraction followed by liquid chromatography-tandem mass spectrometry. J Chromatogr A. 2005;1067(1–2):93–100.
Bogialli S, D’Ascenzo G, Di Corcia A, Innocenti G, Lagana A, Pacchiarotta T. Monitoring quinolone antibacterial residues in bovine tissues: extraction with hot water and liquid chromatography coupled to a single- or triple-quadrupole mass spectrometer. Rapid Commun Mass Spectrom. 2007;21(17):2833–42. https://doi.org/10.1002/rcm.3155.
Bohm DA, Stachel CS, Gowik P. Multi-method for the determination of antibiotics of different substance groups in milk and validation in accordance with Commission Decision 2002/657/EC. J Chromatogr A. 2009;1216(46):8217–23. https://doi.org/10.1016/j.chroma.2009.06.058.
Ruiz-Viceo JA, Rosales-Conrado N, Guillén-Casla V, Pérez-Arribas LV, León-González ME, Polo-Díez LM. Fluoroquinolone antibiotic determination in bovine milk using capillary liquid chromatography with diode array and mass spectrometry detection. J Food Compost Anal. 2012;28(2):99–106. https://doi.org/10.1016/j.jfca.2012.08.003.
Horton RA, Randall LP, Bailey-Horne V, Heinrich K, Sharman M, Brunton LA, et al. Degradation of cefquinome in spiked milk as a model for bioremediation of dairy farm waste milk containing cephalosporin residues. J Appl Microbiol. 2015;118(4):901–10. https://doi.org/10.1111/jam.12765.
Hermo MP, Nemutlu E, Kir S, Barron D, Barbosa J. Improved determination of quinolones in milk at their MRL levels using LC-UV, LC-FD, LC-MS and LC-MS/MS and validation in line with regulation 2002/657/EC. Anal Chim Acta. 2008;613(1):98–107. https://doi.org/10.1016/j.aca.2008.02.045.
Tang Q, Yang T, Tan X, Luo J. Simultaneous determination of fluoroquinolone antibiotic residues in milk sample by solid-phase extraction-liquid chromatography-tandem mass spectrometry. J Agric Food Chem. 2009;57(11):4535–9. https://doi.org/10.1021/jf900513b.
Turnipseed SB, Andersen WC, Karbiwnyk CM, Madson MR, Miller KE. Multi-class, multi-residue liquid chromatography/tandem mass spectrometry screening and confirmation methods for drug residues in milk. Rapid Commun Mass Spectrom. 2008;22(10):1467–80. https://doi.org/10.1002/rcm.3532.
Wang J, Fan X, Liu Y, Du Z, Feng Y, Jia L, et al. Extraction optimization of sixteen cephalosporins in milk by filtered solid phase extraction and ultra high pressure liquid chromatography coupled to tandem mass spectrometry. Anal Methods. 2017;9(8):1282–9. https://doi.org/10.1039/C6AY03444F.
Spielmeyer A, Ahlborn J, Hamscher G. Simultaneous determination of 14 sulfonamides and tetracyclines in biogas plants by liquid-liquid-extraction and liquid chromatography tandem mass spectrometry. Anal Bioanal Chem. 2014;406(11):2513–24. https://doi.org/10.1007/s00216-014-7649-3.
Martins MT, Barreto F, Hoff RB, Jank L, Arsand JB, Motta TMC, et al. Multiclass and multi-residue determination of antibiotics in bovine milk by liquid chromatography–tandem mass spectrometry: combining efficiency of milk control and simplicity of routine analysis. I Int Dairy J. 2016;59(Supplement C):44–51. https://doi.org/10.1016/j.idairyj.2016.02.048.
Dołhań A, Urbaniak B, Manuszewska M, Klause R, Tomczak S, Muszalska I, et al. Critical parameters for the stability of cefquinome sulfate in aqueous solutions and solid phase. React Kinet Mech Cat. 2017;122(2):715–28. https://doi.org/10.1007/s11144-017-1257-0.
Berendsen BJ, Gerritsen HW, Wegh RS, Lameris S, van Sebille R, Stolker AA, et al. Comprehensive analysis of ss-lactam antibiotics including penicillins, cephalosporins, and carbapenems in poultry muscle using liquid chromatography coupled to tandem mass spectrometry. Anal Bioanal Chem. 2013;405(24):7859–74. https://doi.org/10.1007/s00216-013-6804-6.
Riediker S, Rytz A, Stadler RH. Cold-temperature stability of five beta-lactam antibiotics in bovine milk and milk extracts prepared for liquid chromatography-electrospray ionization tandem mass spectrometry analysis. J J Chromatogr A. 2004;1054(1–2):359–63.
Gowik P, Polzer J, Uhlig S. Reference material in residue control: assessment of matrix effects. Accred Qual Assur. 2007;12(3):161–6. https://doi.org/10.1007/s00769-006-0220-y.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 205 kb)
Rights and permissions
About this article
Cite this article
Helmschrodt, C., Schmidt, K., Bertulat, S. et al. Quantitative analysis of cefquinome considering different matrix compositions of bovine colostrum and raw milk. Anal Bioanal Chem 410, 7465–7475 (2018). https://doi.org/10.1007/s00216-018-1360-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-1360-8