Abstract
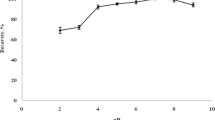
A simple, rapid, and efficient ultrasound-assisted dispersive liquid–liquid microextraction (US-DLLME) method was developed for extraction of tetracycline residues from egg supplement samples, with subsequent determination by flow injection analysis (FIA) coupled to a liquid waveguide capillary cell (LWCC) and a controlled temperature heating bath. Tetracyclines react with diazotized p-sulfanilic acid, in a slightly alkaline medium, to form azo compounds that can be measured at 435 nm. The reaction sensitivity improved substantially (5.12-fold) using an in-line heating temperature of 45 °C. Multivariate methodology was used to optimize the factors affecting the extraction efficiency, considering the volumes of extraction and disperser solvents, sonication time, extraction time, and centrifugation time. Good linearity in the range 30–600 μg L−1 was obtained for all the tetracyclines, with regression coefficients (r) higher than 0.9974. The limits of detection ranged from 6.4 to 11.1 μg L−1, and the recoveries were in the range 85.7–96.4 %, with relative standard deviation lower than 9.8 %. Analyte recovery was improved by approximately 6 % when the microextraction was assisted by ultrasound. The results obtained with the proposed US-DLLME-FIA method were confirmed by a reference HPLC method and showed that the egg supplement samples analyzed were suitable for human consumption.





Similar content being viewed by others
References
Bruno F, Curini R, Di-Corcia A, Nazzari M, Pallagrosi M. An original approach to determining traces of tetracycline antibiotics in milk and eggs by solid-phase extraction and liquid chromatography/mass spectrometry. Rapid Commun Mass Spectrom. 2002;16:1365–76.
Dasenaki ME, Thomaidis NS. Multi-residue determination of 115 veterinary drugs and pharmaceutical residues in milk powder, butter, fish tissue and eggs using liquid chromatography–tandem mass spectrometry. Anal Chim Acta. 2015;880:103–21.
Sczesny S, Nau H, Hamscher G. Residue analysis of tetracyclines and their metabolites in eggs and in the environment by HPLC coupled with a microbiological assay and tandem mass spectrometry. J Agric Food Chem. 2003;51:697–703.
Piatkowska M, Jedziniak P, Zmudzki J. Comparison of different sample preparation procedures for multiclass determination of selected veterinary drug, coccidiostat and insecticide residues in eggs by liquid chromatography-tandem mass spectrometry. Anal Methods. 2014;6:3034–44.
Frenich AG, Aguilera-Luiz M, Vidal JLM, Romero-González R. Comparison of several extraction techniques for multiclass analysis of veterinary drugs in eggs using ultra-high pressure liquid chromatography–tandem mass spectrometry. Anal Chim Acta. 2010;661:150–60.
Jiménez V, Rubies A, Centrich F, Companyó R, Guiteras J. Development and validation of a multiclass method for the analysis of antibiotic residues in eggs by liquid chromatography–tandem mass spectrometry. J Chromatogr A. 2011;1218:1443–51.
Song J, Zhang ZH, Zhang YQ, Feng C, Wang GN, Wang JP. Ionic liquid dispersive liquid–liquid microextraction combined with high performance liquid chromatography for determination of tetracycline drugs in eggs. Anal Methods. 2014;6:6459–66.
Canada, Food and Drugs Act SOR–87. List of maximum residue limits (MRLs) for veterinary drugs in foods. Canada Gazette Part II. 2013;147:1306.
Codex Alimentarius CAC/MRL–2. Maximum residues limits (MRLs) and risk management recommendations (RMRs) for residues of veterinary drugs in Foods. 2014:8.
Union E. Commission regulation No 37. On pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin. Off J Eur Union. 2010;L15:19–64.
Nikolaidou KI, Samanidou VF, Papadoyannis IN. Development and validation of an HPLC method for the determination of seven tetracycline antibiotics residues in chicken muscle and egg yolk according to 2002/657/EC. J Liq Chrom Relat Tech. 2008;31:2141–58.
Liu Y, Yang H, Yang S, Hu Q, Cheng H, Liu H, et al. High-performance liquid chromatography using pressurized liquid extraction for the determination of seven tetracyclines in egg, fish and shrimp. J Chromatogr B. 2013;917–918:11–7.
Jing T, Niu J, Xia H, Dai Q, Zheng H, Hao Q, et al. Online coupling of molecularly imprinted solid-phase extraction to HPLC for determination of trace tetracycline antibiotic residues in egg samples. J Sep Sci. 2001;34:1469–76.
Zurhelle G, Muller-Seitz E, Petz M. Automated residue analysis of tetracyclines and their metabolites in whole egg, egg white, egg yolk and hen’s plasma utilizing a modified ASTED system. J Chromatogr B. 2000;739:191–203.
Heller DN, Nochetto CB, Rummel NG, Thomas MH. Development of multiclass methods for drug residues in eggs: hydrophilic solid-phase extraction cleanup and liquid chromatography/tandem mass spectrometry analysis of tetracycline, fluoroquinolone, sulfonamide, and beta-lactam residues. J Agric Food Chem. 2006;54:5267–78.
Capriotti AL, Cavaliere C, Piovesana S, Samperi R, Laganà A. Multiclass screening method based on solvent extraction and liquid chromatography–tandem mass spectrometry for the determination of antimicrobials and mycotoxins in egg. J Chromatogr A. 2012;1268:84–90.
Piatkowska M, Jedziniak P, Zmudzki J. Multiresidue method for the simultaneous determination of veterinary medicinal products, feed additives and illegal dyes in eggs using liquid chromatography–tandem mass spectrometry. Food Chem. 2016;197:571–80.
Chen L, Liu J, Zeng Q, Wang H, Yu A, Zhang H, et al. Preparation of magnetic molecularly imprinted polymer for the separation of tetracycline antibiotics from egg and tissue samples. J Chromatogr A. 2009;1216:3710–9.
Jia X, Shao B, Wu Y, Yang Y, Zhang J. Simultaneous determination of tetracyclines and quinolones antibiotics in egg by ultra-performance liquid chromatography-electrospray tandem mass spectrometry. J AOAC Int. 2008;91:461–8.
Jing T, Gao XD, Wang P, Wang Y, Lin YF, Hu XZ, et al. Determination of trace tetracycline antibiotics in foodstuffs by liquid chromatography–tandem mass spectrometry coupled with selective molecular-imprinted solid-phase extraction. Anal Bioanal Chem. 2009;393:2009–18.
Gałuszka A, Migaszewski Z, Namiesnik J. The 12 principles of green analytical chemistry and the SIGNIFICANCE mnemonic of green analytical practices. Trends Anal Chem. 2013;50:78–84.
Saraji M, Boroujeni MK. Recent developments in dispersive liquid–liquid microextraction. Anal Bioanal Chem. 2014;406:2027–66.
Saraji M, Marzban M. Determination of 11 priority pollutant phenols in wastewater using dispersive liquid–liquid microextraction followed by high-performance liquid chromatography–diode-array detection. Anal Bioanal Chem. 2010;396:2685–93.
Vázquez MMP, Vázquez PP, Galera MM, García MDG, Uclés A. Ultrasound-assisted ionic liquid dispersive liquid–liquid microextraction coupled with liquid chromatography-quadrupole-linear ion trap-mass spectrometry for simultaneous analysis of pharmaceuticals in wastewaters. J Chromatogr A. 2013;1291:19–26.
Farajzadeh MA, Bahram M, Vardast MR, Bamorowat M. Dispersive liquid-liquid microextraction for the analysis of three organophosphorus pesticides in real samples by high performance liquid chromatography-ultraviolet detection and its optimization by experimental design. Microchim Acta. 2011;172:465–70.
Diao C, Li C, Yang X, Sun A, Liu R. Magnetic matrix solid phase dispersion assisted dispersive liquid liquid microextraction of ultra trace polychlorinated biphenyls in water prior to GC-ECD determination. Microchim Acta. 2016;183:1261–8.
Kozani RR, Mofid-Nakhaei J, Jamali MR. Rapid spectrophotometric determination of trace amounts of palladium in water samples after dispersive liquid–liquid microextraction. Environ Monit Assess. 2013;185:6531–7.
Bidari A, Hemmatkhah P, Jafarvand S, Hosseini MRM, Assadi Y. Selenium analysis in water samples by dispersive liquid-liquid microextraction based on piazselenol formation and GC–ECD. Microchim Acta. 2008;163:243–9.
Jafarvand S, Shemirani F. Supramolecular-based dispersive liquid–liquid microextraction: a novel sample preparation technique for determination of inorganic species. Microchim Acta. 2011;173:353–9.
Frugeri PM, Lago AC, Wisniewski C, Luccas PO. A spectrophotometric flow injection system for streptomycin determination in veterinary samples. Spectrochim Acta A. 2014;117:304–8.
Rodríguez MP, Pezza HR, Pezza L. Simple and clean determination of tetracyclines by flow injection analysis. Spectrochim Acta A. 2016;153:386–92.
Mason RL, Gunst RF, Hess JL. Statistical design and analysis of experiments. 2nd ed. Hoboken, New Jersey: Wiley; 2003.
Currie L, Horwitz W. IUPAC recommendations for defining and measuring detection and quantification limits. Anal Magazine. 1994;22:M24–6.
Atkins P, Jones L. Chemical principles: questioning the modern life and environment. 5th ed. Bookman; 2011.
Codex Alimentarius CAC/GL–71. Guidelines for the design and implementation of national regulatory food safety assurance programme associated with the use of veterinary drugs in food producing animals. 2009:22.
Acknowledgments
The authors would like to thank CNPq for financial support, and the PAEDEX/AUIP/UNESP program for the awarding of a scholarship to M. P. Rodríguez.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 223 kb)
Rights and permissions
About this article
Cite this article
Rodríguez, M.P., Pezza, H.R. & Pezza, L. Ultrasound-assisted dispersive liquid–liquid microextraction of tetracycline drugs from egg supplements before flow injection analysis coupled to a liquid waveguide capillary cell. Anal Bioanal Chem 408, 6201–6211 (2016). https://doi.org/10.1007/s00216-016-9732-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9732-4