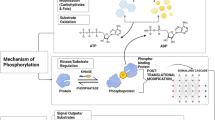
Abstract
While the molecular mechanisms underlying postmortem change have been exhaustively investigated, the establishment of an objective and reliable means for estimating postmortem interval (PMI) remains an elusive feat. In the present study, we exploit low molecular weight metabolites to estimate postmortem interval in mice. After sacrifice, serum and muscle samples were procured from C57BL/6J mice (n = 52) at seven predetermined postmortem intervals (0, 1, 3, 6, 12, 24, and 48 h). After extraction and isolation, low molecular weight metabolites were measured via gas chromatography/mass spectrometry (GC/MS) and examined via semi-quantification studies. Then, PMI prediction models were generated for each of the 175 and 163 metabolites identified in muscle and serum, respectively, using a non-linear least squares curve fitting program. A PMI estimation panel for muscle and serum was then erected which consisted of 17 (9.7 %) and 14 (8.5 %) of the best PMI biomarkers identified in muscle and serum profiles demonstrating statistically significant correlations between metabolite quantity and PMI. Using a single-blinded assessment, we carried out validation studies on the PMI estimation panels. Mean ± standard deviation for accuracy of muscle and serum PMI prediction panels was −0.27 ± 2.88 and −0.89 ± 2.31 h, respectively. Ultimately, these studies elucidate the utility of metabolomic profiling in PMI estimation and pave the path toward biochemical profiling studies involving human samples.



Similar content being viewed by others
References
Madea B, Henssge C, Honig W, Gerbracht A. References for determining the time of death by potassium in vitreous humor. Forensic Sci Int. 1989;40:231–43.
Bocaz-Beneventi G, Tagliaro F, Bortolotti F, Manetto G, Havel J. Capillary zone electrophoresis and artificial neural networks for estimation of the post-mortem interval (PMI) using electrolytes measurements in human vitreous humour. Int J Legal Med. 2002;116:5–11.
James RA, Hoadley PA, Sampson B. Determination of postmortem interval by sampling vitreous humour. Am J Forensic Med Pathol. 1997;18:158–62.
Henssge C, Althaus L, Bolt J, Freislederer A, Haffner H-T, Henssge CA, et al. Experiences with a compound method for estimating the time since death. I. Rectal temperature nomogram for time since death. Int J Legal Med. 2000;113:303–19.
Huang Q, Tan YX, Yin PY, Ye GZ, Gao P, Lu X, et al. Metabolic characterization of hepatocellular carcinoma using nontargeted tissue metabolomics. Cancer Res. 2013;73(16):4992–5002.
Schmidt TM, Wang ZJ, Keller S, Heinemann A, Acar S, Graessner J, et al. Postmortem 31P magnetic resonance spectroscopy of the skeletal muscle: α-ATP/Pi ratio as a forensic tool? Forensic Sci Int. 2014;242:172–6.
Hirakawa K, Koike K, Uekusa K, Nihira M, Yuta K, Ohno Y. Experimental estimation of postmortem interval using multivariate analysis of proton NMR metabolomic data. Legal Med. 2009;11:S282–5.
Prahlow J (2010) Forensic pathology for police, death investigators, attorneys, and forensic scientists. In: Press H, editor.
Mallach HJ. Zur frage der todeszeitbestimmung. Berl Med. 1964;18:577–82.
Kobayashi M, Takatori T, Nakajima M, Sakurada K, Hatanaka K, Ikegaya H, et al. Onset of rigor mortis is earlier in red muscle than in white muscle. Int J Leg Med. 2000;113:240–3.
Marshall TK, Hoare FE. The use of the body temperature in estimating the time of death. J Forensic Sci. 1962;7:211–21.
Vanezis P, Trujillo O. Evaluation of hypostasis using a colorimeter measuring system and its application to assessment of the post-mortem interval (time of death). Forensic Sci Int. 1996;78:19–28.
Dettmer K, Aronov PA, Hammock BD. Mass spectrometry-based metabolomics. Mass Spectrom Rev. 2007;26(1):51–78.
Lokhov PG, Dashtiev MI, Moshkovskii SA, Archakov AI. Metabolite profiling of blood plasma of patients with prostate cancer. Metabolomics. 2010;6(1):156–63.
Duarte IF, Rocha CM, Gil AM. Metabolic profiling of biofluids: potential in lung cancer screening and diagnosis. Expert Rev Mol Diagn. 2013;13(7):737–48.
Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009;457:910–4.
Hirayama A, Kami K, Sugimoto M, Sugawara M, Toki N, et al. Quantitative metabolome profiling of colon and stomach cancer microenvironment by capillary electrophoresis time-of-flight mass spectrometry. Cancer Res. 2009;69:4918–25.
Yoshida M, Hatano N, Nishiumi S, Irino Y, Izumi Y, et al. Diagnosis of gastroenterological diseases by metabolome analysis using gas chromatography–mass spectrometry. J Gastroenterol. 2012;47:9–20.
Nishiumi S, Shinohara M, Ikeda A, Yoshie T, Hatano N, et al. Serum metabolomics as a novel diagnostic approach for pancreatic cancer. Metabolomics. 2010;6:518–28.
Tsugawa H, Bamba T, Shinohara M, et al. Practical non-targeted gas chromatography/mass spectrometry-based metabolomics platform for metabolic phenotype analysis. J Biosci Bioeng. 2011;112:292–8.
Tsugawa H, Tsujimoto Y, Arita M, et al. GC/MS based metabolomics: development of a data mining system for metabolite identification by using soft independent modeling of class analogy (SIMCA). BMC Bioinformatics. 2001;12:131.
Ackermann K, Ballantyne KN, Kayser M. Estimating trace deposition time with circadian biomarkers: a prospective and versatile tool for crime scene reconstruction. Int J Legal Med. 2010;124(5):387–95.
Kimura A, Ishida Y, Hayashi T, Nosaka M, Kondo T. Estimating time of death based on the biological clock. Int J Legal Med. 2011;125:385–91.
Bojkowski CJ, Aldhous ME, English J, Franey C, Poulton AL, Skene DJ, et al. Suppression of nocturnal plasma melatonin and 6-sulphatoxymelatonin by bright and dim light in man. Horm Metab Res. 1987;19:437–40.
Hack LM, Lockley SW, Arendt J, Skene DJ. The effects of low-dose 0.5-mg melatonin on the free-running circadian rhythms of blind subjects. J Biol Rhythms. 2003;18:420–9.
Thompson C, Franey C, Arendt J, Checkley SA. A comparison of melatonin secretion in depressed patients and normal subjects. Br J Psychiatry. 1988;152:260–5.
Latil M, Rocheteau P, Châtre L, Sanulli S, Mémet S, Ricchetti M, et al. Skeletal muscle stem cells adopt a dormant cell state post mortem and retain regenerative capacity. Nat Commun. 2012;3:903.
Bauer M, Gramlich I, Polzin S, Patzelt D. Quantification of mRNA degradation as possible indicator of postmortem interval—a pilot study. Leg Med. 2003;5(4):220–7.
Sabucedo AJ, Furton KG. Estimation of postmortem interval using the protein marker cardiac troponin I. Forensic Sci Int. 2003;134(1):11–6.
Thaik-Oo M, Tanaka E, Tsuchiya T, Kominato Y, Honda K, Yamazaki K, et al. Estimation of postmortem interval from hypoxic inducible levels of vascular endothelial growth factor. J Forensic Sci. 2002;47(1):186–9.
Boy SC, Bernitz H, Van Heerden WF. Flow cytometric evaluation of postmortem pulp DNA degradation. Am J Forensic Med Pathol. 2003;24(2):123–7.
Singh D, Prashad R, Sharma S, Pandeya. Double logarithmic, linear relationship between postmortem vitreous sodium/potassium electrolytes concentration ratio and time since death in subjects of Chandigarh zone of north-west India’s IAFM. 2005;27(3):159–164 15.
Prasad BK, Choudhary A, Sincha TNA. Study of correlation between vitreous potassium level and postmortem interval. Kathmandu Univ Med J. 2003;1(2):132–4.
Amith M, Jawahar K. Vitreous humour biochemical constituents—evaluation of eye differences. Am J Forensic Med Pathol. 2005;26(2):146–9.
Green M, Wright JC. Post-mortem interval estimation using body temperature data only. Forensic Sci Int. 1985;28(1):53–62.
Nelson E. Estimation of short-term postmortem interval utilizing core body temperature: a new algorithm. Forensic Sci Int. 2000;109:31–8.
Bleavins MR, Carini C, Jurima-Romet M, Ramin R. Biomarkers in drug development: a handbook of practice, application and strategy. Hoboken: Wiley-Blackwell; 2010.
Sato et al. A preliminary study on postmortem interval estimation of suffocated rats by GC-MS/MS-based plasma metabolic profiling. Anal Bioanal Chem. 2015;407:3659–65.
Kaszynski RH, Nishiumi S, Kondo T, Takahashi M, Kuse A, Asano M, Yoshida M, Azuma T, Ueno Y. Estimation of post-mortem interval: a novel approach utilizing serum and muscle metabolomics profiling. Oral presentation at the 97th Congress of the Japanese Society of Legal Medicine, Sapporo, Japan; 2013.
Kaszynski RH, Nishiumi S, Kondo T, Takahashi M, Kuse A, Asano M, Yoshida M, Azuma T, Ueno Y. Muscle and serum metabolomics profiling: a novel method for estimating post-mortem interval. Oral presentation at 9th International Symposium on Advances in Legal Medicine, Fukuoka, Japan; 2014.
Sampaio-Silva F, Magalhães T, Carvalho F, Dinis-Oliveira RJ, Silvestre R, Kayser M. Profiling of RNA degradation for estimation of post mortem interval. PLoS One. 2013;8(2):e56507.
Acknowledgments
The authors wish to express their gratitude to Ms. Azumi Kuse (Department of Legal Medicine, Kobe University Graduate School of Medicine) and Dr. Koji Yamamoto for their valuable technical contributions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All of the animal experiments performed in this study were approved by the institutional animal care and use committee and carried out according to the Kobe University Animal Experimentation Regulations. Specimens collected for subsequent analysis were utilized in accordance with the guidelines set forth by the Kobe University Graduate School of Medicine.
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 237 kb)
Rights and permissions
About this article
Cite this article
Kaszynski, R.H., Nishiumi, S., Azuma, T. et al. Postmortem interval estimation: a novel approach utilizing gas chromatography/mass spectrometry-based biochemical profiling. Anal Bioanal Chem 408, 3103–3112 (2016). https://doi.org/10.1007/s00216-016-9355-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9355-9