Abstract
The combination of dynamic pH junction, sweeping (using borate complexation), and large volume sample stacking (LVSS) is investigated as three consecutive steps for on-line focusing in the sensitive quantitation of urinary nucleosides by CE-UVD. A low conductivity aqueous sample matrix free from borate and a high conductivity BGE (containing borate, pH 9.25) are needed to fulfill the required conditions for dynamic pH junction, LVSS, and sweeping. Parameters affecting the separation and the enrichment efficiency are studied such as buffer concentration, separation voltage, capillary temperature, sample composition, and sample injection volume. Prerequisite for the developed strategy is the extraction of the nucleosides from urine using a phenylboronate affinity gel, which is described to be a unique means for the selective enrichment of cis-diol metabolites under alkaline conditions. The impact of ionic constituents remaining in the eluate after extraction on focusing efficiency and resolution is investigated. The developed method is applied to the analysis of blank and spiked urine samples. Fundamental aspects underlying the proposed enrichment procedure are discussed. A detection limit as low as 10 ng mL−1 is achieved. To the best of our knowledge, this LOD represents the lowest LOD reported so far for the analysis of nucleosides using CE with UV detection and provides a comparable sensitivity to CE/MS. Because of the high sensitivity, the proposed method shows a great potential for the analysis of nucleosides in human urine and other types of biological fluids.
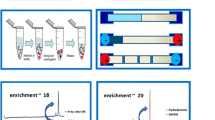
Schematic presentation of the developed three-step focusing mechanism (combining dynamic pHjunction, sweeping using borate complexation, and LVSS).











Similar content being viewed by others
References
Markuszewski MJ, Waszczuk-Jankowska M, Struck W, Koslinski P (2013) In: Buszewski B, Dziubakiewicz E, Szumski M (eds) Electromigration techniques: theory and practice. Springer, Berlin, p 274
Xu G, Liebich HM, Lehmann R, Müller-Hagedorn S (2001) Methods Mol Biol 162:459–474
Cho SH, Choi MH, Lee WY, Chung BC (2009) Clin Biochem 42:540–543
Rodriguez-Gonzalo E, Garcia-Gomez D, Carabias-Martinez R (2011) J Chromatogr A 1218:9055–9063
Sasco AJ, Rey F, Reynaud C, Bobin JY, Clavel M, Niveleau A (1996) Cancer Lett 108:157–162
Wang S, Zhao X, Mao Y, Cheng Y (2007) J Chromatogr A 1147:254–260
Helboe T, Hansen SH (1999) J Chromatogr A 836:315–324
Zhang M, El-Rassi Z (1999) Electrophoresis 20:31–36
Szymanska E, Markuszewski MJ, Bodzioch K, Kaliszan R (2007) J Pharm Biomed Anal 44:1118–1126
Zheng YF, Kong HW, Xiong JH, Shen L, Xu GW (2005) Clin Biochem 38:24–30
Liebich HM, Müller-Hagedorn S, Klaus F, Meziane K, Kim KR, Frickenschmidt A, Kammerer B (2005) J Chromatogr A 1071:271–275
Liebich HM, Lehmann R, Xu G, Wahl HG, Häring HU (2000) J Chromatogr B Biomed Sci Appl 745:189–196
Zheng YF, Xu GW, Liu DY, Xiong JH, Zhang PD, Zhang C, Yang Q, Shen L (2002) Electrophoresis 23:4104–4109
Rageh AH, Pyell U (2013) J Chromatogr A 1316:135–146
Haunschmidt M, Buchberger W, Klampfl CW (2008) J Chromatogr A 1213:88–92
Terabe S (2010) Procedia Chem 2:2–8
Quirino JP, Terabe S (1998) Science 282:465–468
Breadmore MC, Dawod M, Quirino JP (2011) Electrophoresis 32:127–148
Quirino JP, Terabe S (2001) Chromatographia 53:285–289
Liu J, Zhu F, Liu Z (2009) Talanta 80:544–550
Pyell U (2006) In: Pyell U (ed) Electrokinetic chromatography: theory, instrumentation & applications. Wiley, Chichester
Kazarian AA, Hilder EF, Breadmore MC (2011) J Sep Sci 34:2800–2821
Markuszewski MJ, Britz-McKibbin P, Terabe S, Matsuda K, Nishioka T (2003) J Chromatogr A 989:293–301
Liebich HM, Xu G, Di S, Lehmann R, Häring HU, Lu P, Zhang Y (1997) Chromatographia 45:396–401
Beckman Coulter (2003) P/ACE Setter 7:1–4
Jiang Y, Ma Y (2009) Anal Chem 81:6474–6480
Iqbal J, Müller CE (2011) J Chromatogr A 1218:4764–4771
Rodriguez-Gonzalo E, Hernandez-Prieto R, Garcia-Gomez D, Carabias-Martinez R (2014) J Pharm Biomed Anal 88:489–496
Britz-McKibbin P, Bebault GM, Chen DDY (2000) Anal Chem 72:1729–1735
Gehrke CW, Kuo KC, Davis GE, Suits RD, Waalkes TP, Borek E (1978) J Chromatogr 150:455–476
ICH Harmonised Tripartite Guidelines, Validation of analytical procedures: text and methodology Q2(R1) (1996) http://www.ich.org/products/guidelines/quality/article/quality-guidelines.html. (Accessed 16 Jun 2014)
El-Awady M, Huhn C, Pyell U (2012) J Chromatogr A 1264:124–136
Landers JP (2008) Handbook of capillary and microchip electrophoresis and associated microtechniques, 3rd edn. CRC, Taylor and Francis Group, Boca Raton
Riekkola ML, Joensson JA, Smith RM (2004) Pure Appl Chem 76:443–451
van de Merbel NC (2008) Trends Anal Chem 27:924–933
Kuo KC, Phan DT, Williams N, Gehrke CW (1990) In: Gehrke CW, Kuo KC (eds) Chromatography and modification of Nucleosides. Part c modified nucleosides in cancer and normal metabolism methods and applications. Elsevier, Oxford, p C41
IUPAC-IUB (1974) Pure Appl Chem 40:277–290
Lundblad RL, MacDonald F (2010) Handbook of biochemistry and molecular biology, 4th edn. CRC, Taylor and Francis Group, Boca Raton
Wang P, Ren J (2004) J Pharm Biomed Anal 34:277–283
Xiang TX, Anderson BD (1990) Int J Pharm 59:45–55
Kawamura K (1998) J Chromatogr A 802:167–177
Tadey T, Purdy WC (1994) J Chromatogr B Biomed Sci Appl 657:365–372
Peng X, Bebault GM, Sacks SL, Chen DDY (1997) Can J Chem 75:507–517
Oestergaard J, Jensen H, Holm R (2009) J Sep Sci 32:1712–1721
Britz-McKibbin P, Chen DDY (2002) Electrophoresis 23:880–888
Van Loco J, Elskens M, Croux C, Beernaert H (2002) Accred Qual Assur 7:281–285
Sonnergaard JM (2006) Int J Pharm 321:12–17
Wätzig H, Degenhardt M, Kunkel A (1998) Electrophoresis 19:2695–2752
Reichenbaecher M, Einax JW (2011) Challenges in analytical quality assurance. Springer, Berlin
Wätzig H (1995) J Chromatogr A 700:1–7
European Pharmacopoeia (7.8), 7th edn. (2013) Online Version, European directorate for the quality of medicines & healthcare (EDQM), Strasbourg
Chun MS, Chung DS (2003) Anal Chim Acta 491:173–179
Simpson SL, Quirino JP, Terabe S (2008) J Chromatogr A 1184:504–541
Pawel K, Edward B (2013) In: Boguslaw B, Ewelina D, Michal S (eds) Electromigration techniques: theory and practice. Springer, Berlin, p 217
Miller JC, Miller JN (2005) Statistics and chemometrics for analytical chemistry, 5th edn. Pearson, Prentice Hall
Acknowledgments
A.H. Rageh thanks the Egyptian ministry of higher education and state for scientific research and the Deutscher Akademischer Austauschdienst (DAAD) for funding her PhD scholarship through German Egyptian Research Long-Term Scholarship program (GERLS). We thank Prof. M. Marahiel for providing the lyophilizer. We thank the workshops of the Department of Chemistry for the development of the data-recording unit.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 391 kb)
Rights and permissions
About this article
Cite this article
Rageh, A.H., Kaltz, A. & Pyell, U. Determination of urinary nucleosides via borate complexation capillary electrophoresis combined with dynamic pH junction-sweeping-large volume sample stacking as three sequential steps for their on-line enrichment. Anal Bioanal Chem 406, 5877–5895 (2014). https://doi.org/10.1007/s00216-014-8022-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-8022-2