Abstract
Artesunate (AS), a hemisuccinate derivative of artemisinin, is readily soluble in water and can easily be used in formulations for parenteral treatment of severe malaria. AS is rapidly hydrolyzed to the active metabolite dihydroartemisinin (DHA) and primarily eliminated by biliary excretion after glucuronidation. To investigate systematically the AS metabolism and pharmacokinetics, a novel liquid chromatography–tandem mass spectrometry (LC-MS/MS) method for the simultaneous quantification of AS and its metabolites DHA and DHA glucuronide (DHAG) in human plasma samples was developed. Compared to previous methods, our method includes for the first time the quantification of the glucuronide metabolite using a newly synthesized stable isotope-labeled analogue as internal standard. Sample preparation was performed with only 50 μL plasma by high-throughput solid-phase extraction in the 96-well plate format. Separation of the analytes was achieved on a Poroshell 120 EC-C18 column (50*2.1 mm, 2.7 μm, Agilent Technologies, Waldbronn, Germany). The method was validated according to FDA guidelines. Calibration curves were linear over the entire range from 1 to 2,500 nM (0.4–961.1 ng/mL), 165 to 16,500 nM (46.9–4,691.8 ng/mL), and 4 to 10,000 nM (1.8–4,604.7 ng/mL) for AS, DHA, and DHAG, respectively. Intra- and interbatch accuracy, determined as a deviation between nominal and measured values, ranged from −5.7 to 3.5 % and from 2.7 to 5.8 %, respectively. The assay variability ranged from 1.5 to 10.9 % for intra- and interbatch approaches. All analytes showed extraction recoveries above 85 %. The method was successfully applied to plasma samples from patients under AS treatment.

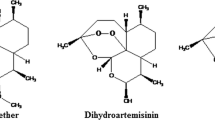
Chemical structures and combined MRM chromatograms of the analytes AS, DHA and DHAG





Similar content being viewed by others
References
Murray CJL, Rosenfeld LC, Lim SS, Andrews KG, Foreman KJ, Haring D, Fullman N, Naghavi M, Lozano R, Lopez AD (2012) Lancet 379(9814):413–431
Kremsner PG, Valim C, Missinou MA, Olola C, Krishna S, Issifou S, Kombila M, Bwanaisa L, Mithwani S, Newton CR, Agbenyega T, Pinder M, Bojang K, Wypij D, Taylor T (2009) J Infect Dis 199(1):142–150
Helbok R, Kendjo E, Issifou S, Lackner P, Newton CR, Kombila M, Agbenyega T, Bojang K, Dietz K, Schmutzhard E, Kremsner PG (2009) J Infect Dis 200(12):1834–1841
Newton CR, Krishna S (1998) Pharmacol Ther 79(1):1–53
World Health Organization (2012) Management of severe malaria: a practical handbook 3rd edition
Kremsner PG, Krishna S (2004) Lancet 364(9430):285–294
Ramu K, Baker JK (1995) J Med Chem 38(11):1911–1921
Ilett KF, Ethell BT, Maggs JL, Davis TME, Batty KT, Burchell B, Binh TQ, Thu LTA, Hung NC, Pirmohamed M, Park BK, Edwards G (2002) Drug Metabol Dispos 30(9):1005–1012
Rolling T, Wichmann D, Schmiedel S, Burchard GD, Kluge S, Cramer JP (2013) Malar J 12:241
Centers for Disease Control and Prevention (CDC) (2013) MMWR Morb Mortal Wkly Rep 62(1):5–8
Kreeftmeijer-Vegter AR, van Genderen PJ, Visser LG, Bierman WF, Clerinx J, van Veldhuizen CK, de Vries PJ (2012) Malar J 11:102
Kerb R, Fux R, Mörike K, Kremsner PG, Gil JP, Gleiter CH, Schwab M (2009) Lancet Infect Dis 9(12):760–774
Hanpithakpong W, Kamanikom B, Dondorp AM, Singhasivanon P, White NJ, Day NPJ, Lindegardh N (2008) J Chromatogr, B 876(1):61–68
Van Quekelberghe SAA, Soomro SA, Cordonnier JA, Jansen FH (2008) J Anal Tox 32(2):133–139
Duthaler U, Keiser J, Huwyler J (2011) J Mass Spectrom 46(2):172–181
Lindegardh N, Hanpithakpong W, Kamanikom B, Singhasivanon P, Socheat D, Yi P, Dondorp AM, McGready R, Nosten F, White NJ, Day NPJ (2008) J Chromatogr, B 876(1):54–60
U.S. Food and Drug Administration (FDA) (2001) Guidance for Industry: Bioanalytical Method Validation, http://www.fda.gov/downloads/Drugs/…/Guidances/ucm070107.pdf
O’Neill PM, Scheinmann F, Stachulski AV, Maggs JL, Park BK (2001) J Med Chem 44(9):1467–1470
Newton P, Suputtamongkol Y, Teja-Isavadharm P, Pukrittayakamee S, Navaratnam V, Bates I, White N (2000) Antimicrob Agents Chemother 44(4):972–977
Acknowledgments
This work was carried out with support of the Robert Bosch Foundation (Stuttgart, Germany), the European and Developing Countries Clinical Trials Partnership (EDCTP) grant # 2004.01.M.d2, the BMBF (Germany) grant 01KA1011, the Deutsche Forschungsgemeinschaft (Germany) grant KE 1629/1-1, and the Interfaculty Center for Pharmacogenomics and Drug Research (ICEPHA grant 10-0-0). Agilent Technologies (Waldbronn, Germany) is gratefully acknowledged for providing their LC-MS/MS system for sample measurements and herewith in particular Lázló Tölgyesi and Volker Gnau.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Geditz, M.C.K., Heinkele, G., Ahmed, A. et al. LC-MS/MS method for the simultaneous quantification of artesunate and its metabolites dihydroartemisinin and dihydroartemisinin glucuronide in human plasma. Anal Bioanal Chem 406, 4299–4308 (2014). https://doi.org/10.1007/s00216-014-7820-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-7820-x