Abstract
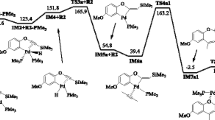
The transition metal-mediated coupling of allyl and alkyne ligands to yield cyclopentadienyl and pentadienyl compounds has been studied theoretically using M06 DFT calculations using CpRu(allyl)(acetylene) as a representative example. We find that the first step in the mechanism is the formation of a 16-electron α, π-vinyl olefin intermediate, whereas the alternative, least-motion 3+2 cycloaddition via a concerted process was calculated to lie at a prohibitively high energy and has the characteristics of a symmetry forbidden reaction. The 3+2 concerted path is shown to be unlikely for η3-allyl complexes in general. There are two competing mechanisms, which lead to Cp2Ru + H2 or CpRu(η5-pentadienyl). The pentadienyl product is predicted to form by either the rearrangement of the α, π-vinyl olefin complex or an electrocyclic ring opening of a cyclopentadiene intermediate. The alternative mechanism involves a ring closure of the α, π-vinyl olefin intermediate to yield CpRu(H)(cyclopentadiene). Four mechanistic scenarios were investigated for the reductive elimination of H2 to give Cp2Ru. The lowest energy process occurs via CpRu(H)2(η3-Cp). The other three possibilities are symmetry forbidden. It is shown that the partitioning of pentadienyl versus cyclopentadienyl products depends on the electronic characteristics of the metal. As the transition metal becomes more electron rich, the pathway to the pentadienyl product becomes more favored. Calculations on CpCo(π-allyl)(acetylene)+ and CpTc(π-allyl)(acetylene)− are in agreement with this hypothesis.








Similar content being viewed by others
References
Welker ME (1992) Chem Rev 92:97–112
Omae I (2008) Appl Organomet Chem 22:149–166
Ylijoki KEO, Stryker JM (2013) Chem Rev 113:2244–2266
Mazzoni R, Salmi M, Zanotti V (2012) Chem Eur J 18:10174–10194
Liyuan L, Astruc D (2011) Coord Chem Rev 255:2933–2945
Xing Y, Wang N.-X (2012) ibid 256:938–952
Chin CS, Won G, Chong D, Kim M, Lee H (2002) Acc Chem Res 35:218–225
Harmata M (2010) Chem Commun 46:8904–8922
Gothelf KV, Jorgensen KA (1998) Chem Rev 98:863–910
Goodall GW, Hayes W (2006) Chem Soc Rev 35:280–312
Harmata M (2010) Chem Commun 46:8886–8903
Moyano A, Rios R (2011) Chem Rev 111:4703–4832
Lutsenko ZL, Aleksandrov GG, Petrovskii PV, Shubina ES, Andrianov VG, Struchkov YT, Rubezhov AZ (1985) J Organomet Chem 281:349–364
Schwiebert KE, Stryker JM (1993) Organometallics 12:600–602
Older CM, Stryker JM (2000) Organometallics 19:2661–2663
Older CM, Stryker JM (2000) Organometallics 19:3266–3268
Older CM, McDonald R, Stryker JM (2005) J Am Chem Soc 127:14202–14203
Barrado G, Hricko MM, Miguel D, Riera V, Wally H (1998) Organometallics 17:820–826
Betz P, Jolly PW, Krüger C, Zakrzewski U (1991) Organometallics 10:3520–3525
Trost BM, Indolese I (1993) J Am Chem Soc 115:4361–4362
Nehl H (1993) Chem Ber 126:1519–1527
Fischer RA, Herrmann WA (1989) J Organomet Chem 377:275–279
Green M, Taylor SH (1975) J Chem Soc Dalton Trans 12:1142–1149
Sanchez-Castro ME, Ramirez-Monroy A, Paz-Sandoval MA (2005) Organometallics 24:2875–2888
Busetto L, Marchetti F, Mazzoni R, Salmi M, Zacchini S, Zanotti V (2009) Organometallics 28:3465–3472
Salmi M, Busetto L, Mazzoni R, Zacchini S, Zanotti V (2011) Organometallics 30:1175–1181
Mazzoni R, Salmi M, Zacchini S, Busetto L, Zanotti V (2014) J Organomet Chem 751:336–342
Older CM, Stryker JM (1998) Organometallics 17:5596–5598
Nicholls JC, Spencer JL (1994) Organometallics 13:1781–1787
Cracknell RB, Nicholls JC, Spencer JL (1996) Organometallics 15:446–448
Schwiebert KE, Stryker JM (1995) J Am Chem Soc 117:8275–8276
Dzwiniel TL, Etkin N, Stryker JM (1999) J Am Chem Soc 121:10640–10641
Etkin N, Dzwiniel TL, Schwiebert KE, Stryker JM (1998) J Am Chem Soc 120:9702–9703
Ylijoki KEO, Stryker JM (2012) Chem Rev 113:2244–2266
Ramful CD, Konway ZE, Boudreau S, Areephong J, Robertson KN, Ylijoki KEO (2016) J Organomet Chem 824:166–171
Ylijoki KEO, Witherell RD, Kirk AD, Böcklein S, Lofstrand VA, McDonald R, Ferguson MJ, Stryker JM (2009) Organometallics 28:6807–6822
Witherell RD, Ylijoki KEO, Stryker JM (2008) J Am Chem Soc 130:2176–2177
Ylijoki KEO, Kirk AD, Böcklein S, Witherell RD, Stryker JM (2015) Organometallics 34:3335–3357
Ylijoki KEO, Budzelaar PHM, Stryker JM (2012) Chem Eur J 18:9894–9900
Becke AD (1993) J Chem Phys 98:5648–5652
Stephens PJ, Devlin FJ, Chabalowski CF, Frisch MJ (1994) J Phys Chem 98:11623–11627
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Hay JR, Wadt WR (1985) J Chem Phys 82:299–310
Roy LE, Hay JP, Martin RL (2008) J Chem Theory Comput 4:1029–1031
Hehre WJ, Ditchfield R, Pople JA (1972) J Chem Phys 56:2257–2261
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215–241
Weigend F, Ahlrichs R (2005) Phys Chem Chem Phys 7:3297–3305
Grimme S, Antony J, Ehrlich S, Krieg H (2010) J Chem Phys 132:154104–154122
Hratchian HP, Schlegel HB (2005) J Chem Theory Comput 1:61–69
Gaussian 09, Revision B.01 (2010) Gaussian, Inc., Wallingford CT, 2010. The full citation for this reference is given in the Supplementary Material section
Schilling BER, Hoffmann R, Lichtenberger DL (1979) J Am Chem Soc 101:585–591
Silvestre J, Hoffmann R (1985) Helv Chim Acta 68:1461–1506
Padwa A (1984) 1,3-dipolar cycloaddition chemistry, 1st edn. Wiley, New York
Bigot B, Devaquet A, Turro NJ (1981) J Am Chem Soc 103:6
Kreiter CG, Koch EC, Frank W, Reiss G (1994) Inorg Chim Acta 220:77–83
Kreiter CG, Fiedler C, Frank W, Reiss GJ (1995) Chem Ber 128:515–518
Kreiter CG, Fiedler C, Frank W, Reiss GJ (1995) J Organomet Chem 490:125–131
Kreiter CG, Koch EC, Frank W, Reiss GJ (1996) Z Naturforsch 51B:1473–1485
Chen W, Chung HJ, Wang C, Sheridan JB, Cote ML, Lalancette RA (1996) Organometallics 15:3337–3344
Chung HJ, Sheridan JB, Cote ML, Lalancette RA (1996) Organometallics 15:4575–4585
Wang C, Sheridan JB, Chung HJ, Cote ML, Lalancette RA, Rheingold AL (1994) J Am Chem Soc 116:8966–8972
Albright TA, Burdett JK, Whangbo MH (2013) Orbital interactions in chemistry, 2nd edn. Wiley, New York, pp 549–552
Pinhas AR, Carpenter BK (1980) J Chem Soc Chem Commun 1:15–17
Tantillo DJ, Hoffmann R (2001) J Am Chem Soc 123:9855–9859
Tantillo DJ, Hoffmann R (2001) Helv Chim Acta 84:1396–1404
Hirshfeld FL (1977) Theor Chim Act 44:129–138
Davidson ER, Chakravorty S (1992) Theor Chim Acta 83:319–330
Guerra CF, Handgraaf JW, Baerends EJ, Bickelhaupt FM (2004) J Comput Chem 25:189–210
Kirss RU (1992) Organometallics 11:497–499
Kirss RU, Quazi A, Lake CH, Churchill MR (1993) Organometallics 12:4145–4150
Mann BE, Manning PW, Spencer CM (1986) J Organomet Chem 312:C64–C66
Acknowledgements
Computational support from the University of St. Thomas and discussions with Professor Thomas Albright are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Frangi, N.M., Soubra-Ghaoui, C. The [3+2] cycloaddition reaction in CpRu(allyl)(acetylene). Theor Chem Acc 136, 83 (2017). https://doi.org/10.1007/s00214-017-2111-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-017-2111-1