Abstract
Rationale
Cocaine can increase inflammatory neuroimmune markers, including chemokines and cytokines characteristic of innate inflammatory responding. Prior work indicates that the Toll-like receptor 4 (TLR4) initiates this response, and administration of TLR4 antagonists provides mixed evidence that TLR4 contributes to cocaine reward and reinforcement.
Objective
These studies utilize (+)-naltrexone, the TLR4 antagonist, and mu-opioid inactive enantiomer to examine the role of TLR4 on cocaine self-administration and cocaine seeking in rats.
Methods
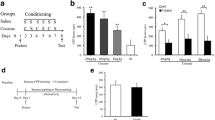
(+)-Naltrexone was continuously administered via an osmotic mini-pump during the acquisition or maintenance of cocaine self-administration. The motivation to acquire cocaine was assessed using a progressive ratio schedule following either continuous and acute (+)-naltrexone administration. The effects of (+)-naltrexone on cocaine seeking were assessed using both a cue craving model and a drug-primed reinstatement model. The highly selective TLR4 antagonist, lipopolysaccharide from Rhodobacter sphaeroides (LPS-Rs), was administered into the nucleus accumbens to determine the effectiveness of TLR4 blockade on cocaine-primed reinstatement.
Results
(+)-Naltrexone administration did not alter the acquisition or maintenance of cocaine self-administration. Similarly, (+)-naltrexone was ineffective at altering the progressive ratio responding. Continuous administration of (+)-naltrexone during forced abstinence did not impact cued cocaine seeking. Acute systemic administration of (+)-naltrexone dose-dependently decreased cocaine-primed reinstatement of previously extinguished cocaine seeking, and administration of LPS-Rs into the nucleus accumbens shell also reduced cocaine-primed reinstatement of cocaine seeking.
Discussion
These results complement previous studies suggesting that the TLR4 plays a role in cocaine-primed reinstatement of cocaine seeking, but may have a more limited role in cocaine reinforcement.







Similar content being viewed by others
References
Araos P, Pedraz M, Serrano A et al (2015) Plasma profile of pro-inflammatory cytokines and chemokines in cocaine users under outpatient treatment: influence of cocaine symptom severity and psychiatric co-morbidity. Addict Biol 20:756–772. https://doi.org/10.1111/adb.12156
Arezoomandan R, Haghparast A (2016) Administration of the glial cell modulator, minocycline, in the nucleus accumbens attenuated the maintenance and reinstatement of morphine-seeking behavior. Can J Physiol Pharmacol 94:257–264. https://doi.org/10.1139/cjpp-2015-0209
Arezoomandan R, Riahi E, Haghparast A (2018) Minocycline increases firing rates of accumbal neurons and modifies the effects of morphine on neuronal activity. Addict Biol 23:1055–1066. https://doi.org/10.1111/adb.12557
Attarzadeh-Yazdi G, Arezoomandan R, Haghparast A (2014) Minocycline, an antibiotic with inhibitory effect on microglial activation, attenuates the maintenance and reinstatement of methamphetamine-seeking behavior in rat. Prog Neuropsychopharmacol Biol Psychiatry 53:142–148. https://doi.org/10.1016/j.pnpbp.2014.04.008
Bachtell RK, Jones JD, Heinzerling KG et al (2017) Glial and neuroinflammatory targets for treating substance use disorders. Drug Alcohol Depend 180:156–170. https://doi.org/10.1016/j.drugalcdep.2017.08.003
Beardsley PM, Shelton KL, Hendrick E, Johnson KW (2010) The glial cell modulator and phosphodiesterase inhibitor, AV411 (ibudilast), attenuates prime- and stress-induced methamphetamine relapse. Eur J Pharmacol 637:102–108. https://doi.org/10.1016/j.ejphar.2010.04.010
Bell RL, Lopez MF, Cui C et al (2015) Ibudilast reduces alcohol drinking in multiple animal models of alcohol dependence. Addict Biol 20:38–42. https://doi.org/10.1111/adb.12106
Birath JB, Briones M, Amaya S et al (2017) Ibudilast may improve attention during early abstinence from methamphetamine. Drug Alcohol Depend 178:386–390. https://doi.org/10.1016/j.drugalcdep.2017.05.016
Brown KT, Levis SC, O’Neill CE et al (2018) Innate immune signaling in the ventral tegmental area contributes to drug-primed reinstatement of cocaine seeking. Brain Behav Immun 67:130–138. https://doi.org/10.1016/j.bbi.2017.08.012
Burkovetskaya ME, Small R, Guo L et al (2020) Cocaine self-administration differentially activates microglia in the mouse brain. Neurosci Lett 728:134951. https://doi.org/10.1016/j.neulet.2020.134951
Calipari ES, Godino A, Peck EG et al (2018) Granulocyte-colony stimulating factor controls neural and behavioral plasticity in response to cocaine. Nat Commun 9:9. https://doi.org/10.1038/s41467-017-01881-x
Chen H, Uz T, Manev H (2009) Minocycline affects cocaine sensitization in mice. Neurosci Lett 452:258–261. https://doi.org/10.1016/j.neulet.2009.01.078
Cooper ZD, Johnson KW, Pavlicova M et al (2016) The effects of ibudilast, a glial activation inhibitor, on opioid withdrawal symptoms in opioid-dependent volunteers. Addict Biol 21:895–903. https://doi.org/10.1111/adb.12261
Cooper ZD, Johnson KW, Vosburg SK et al (2017) Effects of ibudilast on oxycodone-induced analgesia and subjective effects in opioid-dependent volunteers. Drug Alcohol Depend 178:340–347. https://doi.org/10.1016/j.drugalcdep.2017.04.029
Crews FT, Walter TJ, Coleman LG, Vetreno RP (2017) Toll-like receptor signaling and stages of addiction. Psychopharmacology (Berl) 234:1483–1498. https://doi.org/10.1007/s00213-017-4560-6
Fox HC, D’Sa C, Kimmerling A et al (2012) Immune system inflammation in cocaine dependent individuals: implications for medications development. Hum Psychopharmacol 27:156–166. https://doi.org/10.1002/hup.1251
Fujita Y, Kunitachi S, Iyo M, Hashimoto K (2012) The antibiotic minocycline prevents methamphetamine-induced rewarding effects in mice. Pharmacol Biochem Behav 101:303–306. https://doi.org/10.1016/j.pbb.2012.01.005
Gao S-Q, Zhang H, He J-G et al (2020) Neuronal HMGB1 in nucleus accumbens regulates cocaine reward memory. Addict Biol 25:e12739. https://doi.org/10.1111/adb.12739
García-Marchena N, Barrera M, Mestre-Pintó JI et al (2019) Inflammatory mediators and dual depression: potential biomarkers in plasma of primary and substance-induced major depression in cocaine and alcohol use disorders. PLoS One 14:e0213791. https://doi.org/10.1371/journal.pone.0213791
Guo M-L, Liao K, Periyasamy P et al (2015) Cocaine-mediated microglial activation involves the ER stress-autophagy axis. Autophagy 11:995–1009. https://doi.org/10.1080/15548627.2015.1052205
Hill KG, Sable HJK, Ferraro Iii FM, Kiefer SW (2010) Chronic naltrexone treatment and ethanol responsivity in outbred rats. Alcohol Clin Exp Res 34:272–279. https://doi.org/10.1111/j.1530-0277.2009.01090.x
Hofford RS, Russo SJ, Kiraly DD (2019) Neuroimmune mechanisms of psychostimulant and opioid use disorders. Eur J Neurosci 50:2562–2573. https://doi.org/10.1111/ejn.14143
Hutchinson MR, Northcutt AL, Hiranita T et al (2012) Opioid activation of Toll-like receptor 4 contributes to drug reinforcement. J Neurosci 32:11187–11200. https://doi.org/10.1523/JNEUROSCI.0684-12.2012
Hutchinson MR, Watkins LR (2014) Why is neuroimmunopharmacology crucial for the future of addiction research? Neuropharmacology 76(Pt B):218–227. https://doi.org/10.1016/j.neuropharm.2013.05.039
Hutchinson MR, Zhang Y, Brown K et al (2008) Non-stereoselective reversal of neuropathic pain by naloxone and naltrexone: involvement of Toll-like receptor 4 (TLR4). Eur J Neurosci 28:20–29. https://doi.org/10.1111/j.1460-9568.2008.06321.x
Jacobsen JHW, Watkins LR, Hutchinson MR (2014) Discovery of a novel site of opioid action at the innate immune pattern-recognition receptor TLR4 and its role in addiction. Int Rev Neurobiol 118:129–163. https://doi.org/10.1016/B978-0-12-801284-0.00006-3
Kashima DT, Grueter BA (2017) Toll-like receptor 4 deficiency alters nucleus accumbens synaptic physiology and drug reward behavior. Proc Natl Acad Sci USA 114:8865–8870. https://doi.org/10.1073/pnas.1705974114
Kawai T, Akira S (2007) Signaling to NF-kappaB by Toll-like receptors. Trends Mol Med 13:460–469. https://doi.org/10.1016/j.molmed.2007.09.002
Korpi ER, Linden A-M, Hytönen HR et al (2017) Continuous delivery of naltrexone and nalmefene leads to tolerance in reducing alcohol drinking and to supersensitivity of brain opioid receptors. Addict Biol 22:1022–1035. https://doi.org/10.1111/adb.12393
Lacagnina MJ, Rivera PD, Bilbo SD (2017) Glial and neuroimmune mechanisms as critical modulators of drug use and abuse. Neuropsychopharmacology 42:156–177. https://doi.org/10.1038/npp.2016.121
Levandowski ML, Viola TW, Prado CH et al (2016) Distinct behavioral and immunoendocrine parameters during crack cocaine abstinence in women reporting childhood abuse and neglect. Drug Alcohol Depend 167:140–148. https://doi.org/10.1016/j.drugalcdep.2016.08.010
Li MJ, Briones MS, Heinzerling KG et al (2020) Ibudilast attenuates peripheral inflammatory effects of methamphetamine in patients with methamphetamine use disorder. Drug Alcohol Depend 206:107776. https://doi.org/10.1016/j.drugalcdep.2019.107776
Liao K, Guo M, Niu F et al (2016) Cocaine-mediated induction of microglial activation involves the ER stress-TLR2 axis. J Neuroinflammation 13:33. https://doi.org/10.1186/s12974-016-0501-2
Linker KE, Gad M, Tawadrous P et al (2020) Microglial activation increases cocaine self-administration following adolescent nicotine exposure. Nat Commun 11:306. https://doi.org/10.1038/s41467-019-14173-3
Liu HQ, Zhang WY, Luo XT et al (2006) Paeoniflorin attenuates neuroinflammation and dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease by activation of adenosine A1 receptor. Br J Pharmacol 148:314–325. https://doi.org/10.1038/sj.bjp.0706732
Lo Iacono L, Catale C, Martini A et al (2018) From traumatic childhood to cocaine abuse: the critical function of the immune system. Biol Psychiatry 84:905–916. https://doi.org/10.1016/j.biopsych.2018.05.022
Metz VE, Jones JD, Manubay J et al (2017) Effects of ibudilast on the subjective, reinforcing, and analgesic effects of oxycodone in recently detoxified adults with opioid dependence. Neuropsychopharmacology. https://doi.org/10.1038/npp.2017.70
Moreira FP, Medeiros JRC, Lhullier AC et al (2016) Cocaine abuse and effects in the serum levels of cytokines IL-6 and IL-10. Drug Alcohol Depend 158:181–185. https://doi.org/10.1016/j.drugalcdep.2015.11.024
Northcutt AL, Hutchinson MR, Wang X et al (2015) DAT isn’t all that: cocaine reward and reinforcement requires toll like receptor 4 signaling. Mol Psychiatry. https://doi.org/10.1038/mp.2014.177
Paxinos G, Watson, C (2013) The Rat Brain in Stereotaxic Coordinates. Netherlands: Elsevier Science.
Pedraz M, Martín-Velasco AI, García-Marchena N et al (2015) Plasma concentrations of BDNF and IGF-1 in abstinent cocaine users with high prevalence of substance use disorders: relationship to psychiatric comorbidity. PLoS One 10:e0118610. https://doi.org/10.1371/journal.pone.0118610
Poltorak A, He X, Smirnova I et al (1998) Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282:2085–2088. https://doi.org/10.1126/science.282.5396.2085
Soder HE, Berumen AM, Gomez KE et al (2020) Elevated neutrophil to lymphocyte ratio in older adults with cocaine use disorder as a marker of chronic inflammation. Clin Psychopharmacol Neurosci 18:32–40. https://doi.org/10.9758/cpn.2020.18.1.32
Stamatovich S, Lopez-Gamundi P, Suchting R et al (2021) Plasma pro- and anti-inflammatory cytokines may relate to cocaine use, cognitive functioning, and depressive symptoms in cocaine use disorder. Am J Drug Alcohol Abuse 47. https://doi.org/10.1080/00952990.2020.1828439
Tanda G, Mereu M, Hiranita T et al (2016) Lack of specific involvement of (+)-naloxone and (+)-naltrexone on the reinforcing and neurochemical effects of cocaine and opioids. Neuropsychopharmacology 41:2772–2781. https://doi.org/10.1038/npp.2016.91
Theberge FR, Li X, Kambhampati S et al (2013) Effect of chronic delivery of the Toll-like receptor 4 antagonist (+)-naltrexone on incubation of heroin craving. Biological psychiatry 73:729–737. https://doi.org/10.1016/j.biopsych.2012.12.019
Tortorelli LS, Engelke DS, Lunardi P et al (2015) Cocaine counteracts LPS-induced hypolocomotion and triggers locomotor sensitization expression. Behav Brain Res 287:226–229. https://doi.org/10.1016/j.bbr.2015.03.054
Valdizán EM, Díaz A, Pilar-Cuéllar F et al (2012) Chronic treatment with the opioid antagonist naltrexone favours the coupling of spinal cord μ-opioid receptors to Gαz protein subunits. Neuropharmacology 62:757–764. https://doi.org/10.1016/j.neuropharm.2011.08.029
Vallender EJ, Goswami DB, Shinday NM et al (2017) Transcriptomic profiling of the ventral tegmental area and nucleus accumbens in rhesus macaques following long-term cocaine self-administration. Drug Alcohol Depend 175:9–23. https://doi.org/10.1016/j.drugalcdep.2017.01.030
Wang X, Northcutt AL, Cochran TA et al (2019) Methamphetamine activates Toll-like receptor 4 to induce central immune signaling within the ventral tegmental area and contributes to extracellular dopamine increase in the nucleus accumbens shell. ACS Chem Neurosci 10:3622–3634. https://doi.org/10.1021/acschemneuro.9b00225
Worley MJ, Heinzerling KG, Roche DJO, Shoptaw S (2016) Ibudilast attenuates subjective effects of methamphetamine in a placebo-controlled inpatient study. Drug Alcohol Depend 162:245–250. https://doi.org/10.1016/j.drugalcdep.2016.02.036
Wu R, Li J-X (2020) Toll-like receptor 4 signaling and drug addiction. Front Pharmacol 11:603445. https://doi.org/10.3389/fphar.2020.603445
Yue K, Tanda G, Katz JL, Zanettini C (2019) A further assessment of a role for Toll-like receptor 4 in the reinforcing and reinstating effects of opioids. Behav Pharmacol. https://doi.org/10.1097/FBP.0000000000000474
Zhang X, Cui F, Chen H et al (2018) Dissecting the innate immune recognition of opioid inactive isomer (+)-naltrexone derived Toll-like receptor 4 (TLR4) antagonists. J Chem Inf Model 58:816–825. https://doi.org/10.1021/acs.jcim.7b00717
Funding
This work was supported by institutional funds, US Public Health Service grant DA 033358, and the intramural research programs of the National Institute on Drug Abuse and the National Institute on Alcohol Abuse and Alcoholism, National Institute on Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brown, K.T., Levis, S.C., O’Neill, C.E. et al. Toll-like receptor 4 antagonists reduce cocaine-primed reinstatement of drug seeking. Psychopharmacology 240, 1587–1600 (2023). https://doi.org/10.1007/s00213-023-06392-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-023-06392-w