Abstract
Rationale
Exposure of male rats to an inaccessible receptive female and copulation increases dopamine (DA) levels in the nucleus accumbens (NAcc). Males copulating to satiety become sexually inhibited and most of them do not display sexual activity when presented with a sexually receptive female 24 h later. This inhibitory state can be pharmacologically reversed. There are no studies exploring NAcc DA levels during this sexual inhibitory state.
Objectives
To characterize changes in NAcc DA and its metabolites’ levels during sexual satiety development, during the well-established sexual inhibitory state 24 h later, and during its pharmacological reversal.
Methods
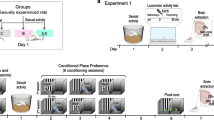
Changes in NAcc DA and its metabolites were measured in sexually experienced male rats, using in vivo microdialysis, during copulation to satiety, when presented to a new sexually receptive female 24 h later, and during the pharmacological reversal of the sexual inhibition by anandamide.
Results
NAcc DA levels remained increased during copulation to satiety. DA basal levels were significantly reduced 24 h after copulation to satiety, as compared to the initial basal levels. Presenting a receptive female behind a barrier 24 h after satiety did not induce the typical NAcc DA elevation in the sexually satiated males but there was a decrease that persisted when they got access to the female, with which they did not copulate. Anandamide injection slightly increased NAcc DA levels coinciding with sexual satiety reversal.
Conclusions
Reduced NAcc DA concentrations coincide with the inhibition of an instinctive, natural rewarding behavior suggesting that there might be a DA concentration threshold needed to be responsive to a rewarding stimulus.






Similar content being viewed by others
Data availability
Raw data will be provided upon request.
References
Ågmo A (2003) Unconditioned sexual incentive motivation in the male Norway Rat (Rattus norvegicus). J Comp Psychol 117:3–14. https://doi.org/10.1037/0735-7036.117.1.3
Ågmo A, Berenfeld R (1990) Reinforcing properties of ejaculation in the male rat: Role of opioids and dopamine. Behav Neurosci 104:177–182. https://doi.org/10.1037/0735-7044.104.1.177
Ågmo A, Turi AL, Ellingsen E, Kaspersen H (2004) Preclinical models of sexual desire: conceptual and behavioural analysis. Pharmacol Biochem Behav 78:379–404. https://doi.org/10.1016/j.pbb.2004.04.013
Ahn S, Phillips AG (1999) Dopaminergic correlates of sensory-specific satiety in the medial prefrontal cortex and nucleus accumbens of the rat. J Neurosci 19:RC29. https://doi.org/10.1523/JNEUROSCI.19-19-j0003.1999
Balfour ME, Yu L, Coolen LM (2004) Sexual behavior and sex associated environmental cues activate the mesolimbic system in male rats. Neuropsychopharmacology 29:718–730. https://doi.org/10.1038/sj.npp.1300350
Bassareo V, Di Chiara G (1997) Differential influence of associative and nonassociative learning mechanisms on the responsiveness of prefrontal and accumbal dopamine transmission to food stimuli in rats fed ad libitum. J Neurosci 17:851–861. https://doi.org/10.1523/JNEUROSCI.17-02-00851.1997
Beach F, Jordan L (1956) Sexual exhaustion and recovery in the male rat. Q J Exp Psychol 8:121–133. https://doi.org/10.1080/17470215608416811
Beaulieu JM, Gainetdinov RR (2011) The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol Rev 63:182–217. https://doi.org/10.1124/pr.110.002642
Benoit-Marand M, Ballion B, Borrelli E, Boraud T, Gonon F (2011) Inhibition of dopamine uptake by D2 antagonists: an in vivo study. J Neurochem 116:449–458. https://doi.org/10.1111/j.1471-4159.2010.07125.x
Berke JD (2018) What does dopamine mean? Nat Neurosci 21:787–793. https://doi.org/10.1038/s41593-018-0152-y
Berridge KC (2007) The debate over dopamine’s role in reward: the case for incentive salience. Psychopharmacology 191:391–431. https://doi.org/10.1007/s00213-006-0578-x
Cachope R, Mateo Y, Mathur BN, Irving J, Wang HL, Morales M, Lovinger DM, Cheer JF (2012) Selective activation of cholinergic interneurons enhances accumbal phasic dopamine release: setting the tone for reward processing. Cell Rep 2:33–41. https://doi.org/10.1016/j.celrep.2012.05.011
Canseco-Alba A, Rodríguez-Manzo G (2014) Low anandamide doses facilitate male rat sexual behaviour through the activation of CB1 receptors. Psychopharmacology 231:4071–4080. https://doi.org/10.1007/s00213-014-3547-9
Canseco-Alba A, Rodríguez-Manzo G (2016) Intra-VTA anandamide infusion produces dose-based biphasic effects on male rat sexual behavior expression. Pharmacol Biochem Behav 150–151:182–189. https://doi.org/10.1016/j.pbb.2016.11.004
Canseco-Alba A, Rodríguez-Manzo G (2019) Endocannabinoids interact with the dopaminergic system to increase sexual motivation: lessons from the sexual satiety phenomenon. Front Behav Neurosci 13:184. https://doi.org/10.3389/fnbeh.2019.00184
Collins AL, Aitken TJI, Huang W, Shieh C, Greenfield VY, Monbouquette HG, Ostlund SB, Wassum KM (2019) Nucleus accumbens cholinergic interneurons oppose cue-motivated behavior. Biol Psychiatry 86:388–396. https://doi.org/10.1016/j.biopsych.2019.02.014
Courtney NA, Mamaligas AA, Ford CP (2012) Species differences in somatodendritic dopamine transmission determine D2-autoreceptor mediated inhibition of ventral tegmental area neuron firing. J Neurosci 32:13520–13528. https://doi.org/10.1523/JNEUROSCI.2745-12.2012
Covey DP, Yocky AG (2021) Endocannabinoid modulation of nucleus accumbens microcircuitry and terminal dopamine release. Front Synaptic Neurosci 13:734975. https://doi.org/10.3389/fnsyn.2021.734975
Damsma G, Pfaus JG, Wenkstern D, Phillips AG, Fibiger HC (1992) Sexual behavior increases dopamine transmission in the nucleus accumbens and striatum of male rats: comparison with novelty and locomotion. Behav Neurosci 106:181–191. https://doi.org/10.1037//0735-7044.106.1.181
Egenrieder L, Mitricheva E, Spanagel R, Noori HR (2020) No basal or drug-induced sex differences in striatal dopaminergic levels: a cluster and meta-analysis of rat microdialysis studies. J Neurochem 152:482–492. https://doi.org/10.1111/jnc.14911
Everitt BJ (1990) Sexual motivation: a neural and behavioural analysis of the mechanisms underlying appetitive and copulatory responses of male rats. Neurosci Biobehav Rev 14:217–232. https://doi.org/10.1016/s0149-7634(05)80222-2
Everitt BJ, Stacey P (1987) Studies of instrumental behaviour with sexual reinforcement in male rats (Rattus norvegicus): II effects of preoptic area lesions, castration and testosterone. J Comp Psychol 101:407–419
Fiorino DF, Coury A, Phillips AG (1997) Dynamic changes in nucleus accumbens dopamine efflux during the Coolidge effect in male rats. J Neurosci 17:4849–4855. https://doi.org/10.1523/JNEUROSCI.17-12-04849.1997
Floresco S, West A, Ash B, Moore H, Grace AA (2003) Afferent modulation of dopamine neuron firing differentially regulates tonic and phasic dopamine transmission. Nat Neurosci 6:968–973. https://doi.org/10.1038/nn1103
Ford CP (2014) The role of D2-autoreceptors in regulating dopamine neuron activity and transmission. Neuroscience 282:13–22. https://doi.org/10.1016/j.neuroscience.2014.01.025
Fumero B, Fernandez-Vera JR, Gonzalez-Mora JL, Mas M (1994) Changes in monoamine turnover in forebrain areas associated with masculine sexual behavior: a microdialysis study. Brain Res 662:233–239. https://doi.org/10.1016/0006-8993(94)90817-6
Gardner EL (2005) Endocannabinoid signaling system and brain reward: emphasis on dopamine. Pharmacol Biochem Behav 81:263–284. https://doi.org/10.1016/j.pbb.2005.01.032
Garduño-Gutiérrez R, León-Olea M, Rodríguez-Manzo G (2013) The mesolimbic system participates in the naltrexone-induced reversal of sexual exhaustion: opposite effects of intra-VTA opioid receptor antagonists on copulation of sexually experienced and sexually exhausted male rats. Behav Brain Res 256:64–71. https://doi.org/10.1016/j.bbr.2013.07.056
Grace AA (1991) Phasic versus tonic dopamine release and the modulation of dopamine system responsivity: a hypothesis for the etiology of schizophrenia. Neuroscience 41:1–24. https://doi.org/10.1016/0306-4522(91)90196-u
Grace AA (2016) Dysregulation of the dopamine system in the pathophysiology of schizophrenia and depression. Nat Rev Neurosci 17:524–532. https://doi.org/10.1038/nrn.2016.57
Grace AA, Floresco SB, Goto Y, Lodge DJ (2007) Regulation of firing of dopaminergic neurons and control of goal-directed behaviors. Trends Neurosci 30:220–227. https://doi.org/10.1016/j.tins.2007.03.003
Guadarrama-Bazante IL, Rodríguez-Manzo G (2019) Nucleus accumbens dopamine increases sexual motivation in sexually satiated male rats. Psychopharmacology 236:1303–1312. https://doi.org/10.1007/s00213-018-5142-y
Guadarrama-Bazante IL, Canseco-Alba A, Rodríguez-Manzo G (2014) Dopamine receptors play distinct roles in sexual behavior expression of rats with a different sexual motivational tone. Behav Pharmacol 25:684–694. https://doi.org/10.1097/FBP.0000000000000086
Haj-Dahmane S, Shen RY (2010) Regulation of plasticity of glutamate synapses by endocannabinoids and the cyclic-AMP/protein kinase A pathway in midbrain dopamine neurons. J Physiol 588:2589–2604. https://doi.org/10.1113/jphysiol.2010.190066
Hernandez-Gonzalez M, Guevara MA, Morali G, Cervantes M (1997) Subcortical multiple unit activity changes during rat male sexual behavior. Physiol Behav 61:285–291. https://doi.org/10.1016/S0031-9384(96)00367-8
Hull EM, Warner RK, Bazzett TJ, Eaton RC, Thompson JT, Scaletta LL (1989) D2/D1 ratio in the medial preoptic area affects copulation of male rats. J Pharmacol Exp Ther 251:422–427
Hull EM, Rodríguez-Manzo G (2017) Male sexual behavior. In: Pfaff DW, Joëls M (eds.) Hormones, Brain and Behavior, vol 1, chap. 1, 3rd edn. Academic Press, Oxford, 1–57
Kelley AE, Berridge KC (2002) The neuroscience of natural rewards: relevance to addictive drugs. J Neurosci 22:3306–3311. https://doi.org/10.1523/JNEUROSCI.22-09-03306.2002
Lupica CR, Riegel AC, Hoffman AF (2004) Marijuana and cannabinoid regulation of brain reward circuits. Br J Pharmacol 143:227–234. https://doi.org/10.1038/sj.bjp.0705931
Mas M, Gonzalez-Mora JL, Louilot A, Solé C, Guadalupe T (1990) Increased dopamine release in the nucleus accumbens of copulating male rats as evidenced by in vivo voltammetry. Neurosci Lett 110:303–308. https://doi.org/10.1016/0304-3940(90)90864-6
Mateo Y, Lack CM, Morgan D, Roberts DC, Jones SR (2005) Reduced dopamine terminal function and insensitivity to cocaine following cocaine binge self-administration and deprivation. Neuropsychopharmacology 30:1455–1463. https://doi.org/10.1038/sj.npp.1300687
Mateo Y, Johnson KA, Covey DP, Atwood BK, Wang HL, Zhang S et al (2017) Endocannabinoid actions on cortical terminals orchestrate local modulation of dopamine release in the nucleus accumbens. Neuron 96:1112-1126.e5. https://doi.org/10.1016/j.neuron.2017.11.012
Mayfield RD, Zahniser NR (2001) Dopamine D2 receptor regulation of the dopamine transporter expressed in Xenopus laevis oocytes is voltage-independent. Mol Pharmacol 59:113–121. https://doi.org/10.1124/mol.59.1.113
Melis MR, Argiolas A (1995) Dopamine and sexual behavior. Neurosci Biobehav Rev 19:19–38. https://doi.org/10.1016/j.physbeh.2004.08.018
Melis MR, Succu S, Mascia MS, Cortis L, Argiolas A (2003) Extracellular dopamine increases in the paraventricular nucleus of male rats during sexual activity. Eur J Neurosci 17:1266–1272. https://doi.org/10.1046/j.1460-9568.2003.02558.x
Mogenson GJ, Jones DL, Yim CY (1980) From motivation to action: functional interface between the limbic system and the motor system. Prog Neurobiol 14:69–97. https://doi.org/10.1016/0301-0082(80)90018-0
Mohebi A, Pettibone JR, Hamid AA, Wong JT, Vinson LT, Patriarchi T, Tian L, Kennedy RT, Berke JD (2019) Dissociable dopamine dynamics for learning and motivation. Nature 570:65–70. https://doi.org/10.1038/s41586-019-1235-y
Ostlund SB, Wassum KM, Murphy NP, Balleine BW, Maidment NT (2011) Extracellular dopamine levels in striatal subregions track shifts in motivation and response cost during instrumental conditioning. J Neurosci 31:200–207. https://doi.org/10.1523/JNEUROSCI.4759-10.2011
Paxinos G, Watson C (2009) The rat brain in stereotaxic coordinates. Academic Press
Pehek EA, Warner RK, Bazzett TJ, Bitran D, Band LC, Eaton RC, Hull EM (1988) Microinjections of cis-flupenthixol, a dopamine antagonist, into the medial preoptic area impairs sexual behavior of male rats. Brain Res 443:70–76. https://doi.org/10.1016/0006-8993(88)91599-5
Pfaus JG (2009) Pathways of sexual desire. J Sex Med 6:1506–1533. https://doi.org/10.1111/j.1743-6109.2009.01309.x
Pfaus JG, Phillips AG (1989) Differential effects of dopamine receptor antagonists on the sexual behavior of male rats. Psychopharmacology 98:363–368. https://doi.org/10.1007/BF00451688
Pfaus JG, Phillips AG (1991) Role of dopamine in anticipatory and consummatory aspects of sexual behavior in the male rat. Behav Neurosci 105:727–743. https://doi.org/10.1037/0735-7044.105.5.727
Pfaus JG, Damsma G, Nomikos GG, Wenkstern DG, Blaha CD, Phillips AG, Fibiger HC (1990) Sexual behavior enhances central dopamine transmission in the male rat. Brain Res 530:345–348. https://doi.org/10.1016/0006-8993(90)91309-5
Pitman KA, Puil E, Borgland SL (2014) GABA(B) modulation of dopamine release in the nucleus accumbens core. Eur J Neurosci 40:3472–3480. https://doi.org/10.1111/ejn.12733
Pleim ET, Matochik JA, Barfield RJ, Auerbach SB (1990) Correlation of dopamine release in the nucleus accumbens with masculine sexual behavior in rats. Brain Res 524:160–163. https://doi.org/10.1016/0006-8993(90)90507-8
Rada P, Avena NM, Hoebel BG (2005) Daily bingeing on sugar repeatedly releases dopamine in the accumbens shell. Neuroscience 134:737–744. https://doi.org/10.1016/j.neuroscience.2005.04.043
Robinson DL, Phillips PE, Budygin EA, Trafton BJ, Garris PA, Wightman RM (2001) Sub-second changes in accumbal dopamine during sexual behavior in male rats. NeuroReport 12:2549–2552. https://doi.org/10.1097/00001756-200108080-00051
Rodríguez-Manzo G (1999) Yohimbine interacts with the dopaminergic system to reverse sexual satiation: further evidence for a role of sexual motivation in sexual exhaustion. Eur J Pharmacol 372:1–8. https://doi.org/10.1016/s0014-2999(99)00140-5
Rodríguez-Manzo G (2015) Glutamatergic transmission is involved in the long lasting sexual inhibition of sexually exhausted male rats. Pharmacol Biochem Behav 131:64–70. https://doi.org/10.1016/j.pbb.2015.02.002
Rodríguez-Manzo G, Canseco-Alba A (2017) A new role for GABAergic transmission in the control of male rat sexual behavior expression. Behav Brain Res 320:21–29. https://doi.org/10.1016/j.bbr.2016.11.041
Rodríguez-Manzo G, Fernández-Guasti A (1994) Reversal of sexual exhaustion by serotonergic and noradrenergic agents. Behav Brain Res 62:127–134. https://doi.org/10.1016/0166-4328(94)90019-1
Rodríguez-Manzo G, Fernández-Guasti A (1995) Opioid antagonists and the sexual satiation phenomenon. Psychopharmacology 122:131–136. https://doi.org/10.1007/BF02246087
Rodríguez-Manzo G, Guadarrama-Bazante IL, Morales-Calderón A (2011) Recovery from sexual exhaustion-induced copulatory inhibition and drug hypersensitivity follow a same time course: Two expressions of a same process? Behav Brain Res 217:253–260. https://doi.org/10.1016/j.bbr.2010.09.014
Rodríguez-Manzo G, González-Morales E, Garduño-Gutiérrez R (2021) Endocannabinoids released in the ventral tegmental area during copulation to satiety modulate changes in glutamate receptors associated with synaptic plasticity processes. Front Synaptic Neurosci 13:701290. https://doi.org/10.3389/fnsyn.2021.701290
Sachs BD, Akasofu K, Citro JK, Daniels SB, Natoli JH (1994) Noncontact stimulation from estrus females evokes penile erection in rats. Physiol Behav 55:1073–1079. https://doi.org/10.1016/0031-9384(94)90390-5
Salamone JD, Correa M (2012) The mysterious motivational functions of mesolimbic dopamine. Neuron 76:470–485. https://doi.org/10.1016/j.neuron.2012.10.021
Salamone JD, Pardo M, Yohn SE, López-Cruz L, San Miguel N, Correa M (2016) Mesolimbic dopamine and the regulation of motivated behavior. Curr Top Behav Neurosci 27:231–257. https://doi.org/10.1007/7854_2015_383
Sanna F, Bratzu J, Serra MP, Leo D, Quartu M, Boi M, Espinoza S, Gainetdinov RR, Melis MR, Argiolas A (2020) Altered sexual behavior in dopamine transporter (DAT) knockout male rats: a behavioral, neurochemical and intracerebral microdialysis study. Front Behav Neurosci 14:58. https://doi.org/10.3389/fnbeh.2020.00058
Schultz W (1998) Predictive reward signal of dopamine neurons. J Neurophysiol 80:1–27. https://doi.org/10.1152/jn.1998.80.1.1
Segovia KN, Correa M, Salamone JD (2011) Slow phasic changes in nucleus accumbens dopamine release during fixed ratio acquisition: a microdialysis study. Neuroscience 196:178–188. https://doi.org/10.1016/j.neuroscience.2011.07.078
Sokolowski JD, Conlan AN, Salamone JD (1998) A microdialysis study of nucleus accumbens core and shell dopamine during operant responding in the rat. Neuroscience 86:1001–1009. https://doi.org/10.1016/s0306-4522(98)00066-9
Solinas M, Justinova Z, Goldberg SR, Tanda G (2006) Anandamide administration alone and after inhibition of fatty acid amide hydrolase (FAAH) increases dopamine levels in the nucleus accumbens shell in rats. J Neurochem 98:408–419. https://doi.org/10.1111/j.1471-4159.2006.03880.x
Sukoff-Rizzo SJ, Schechter LE, Rosenzweig-Lipson S (2008) A novel approach for predicting antidepressant-induced sexual dysfunction in rats. Psychopharmacology 195:459–467. https://doi.org/10.1007/s00213-007-0924-7
Tenk CM, Wilson H, Zhang Q, Pitchers KK, Coolen LM (2009) Sexual reward in male rats: effects of sexual experience on conditioned place preferences associated with ejaculation and intromissions. Horm Behav 55:93–97. https://doi.org/10.1016/j.yhbeh.2008.08.012
Wassum KM, Ostlund SB, Maidment NT (2012) Phasic mesolimbic dopamine signaling precedes and predicts performance of a self-initiated action sequence task. Biol Psychiatry 71:846–854. https://doi.org/10.1016/j.biopsych.2011.12.019
Wenkstern D, Pfaus JG, Fibiger HC (1993) Dopamine transmission increases in the nucleus accumbens of male rats during their first exposure to sexually receptive female rats. Brain Res 618:41–46. https://doi.org/10.1016/0006-8993(93)90426-n
Wightman RM, Robinson DL (2002) Transient changes in mesolimbic dopamine and their association with ‘reward.’ J Neurochem 82:721–735. https://doi.org/10.1046/j.1471-4159.2002.01005.x
Wilson C, Nomikos GG, Collu M, Fibiger HC (1995) Dopaminergic correlates of motivated behavior: importance of drive. J Neurosci 15:5169–5178. https://doi.org/10.1523/JNEUROSCI.15-07-05169.1995
Yang H, de Jong JW, Tak Y, Peck J, Bateup HS, Lammel S (2018) Nucleus accumbens subnuclei regulate motivated behavior via direct inhibition and disinhibition of VTA dopamine subpopulations. Neuron 97:434-449.e4. https://doi.org/10.1016/j.neuron.2017.12.022
Zhang H, Sulzer D (2004) Frequency-dependent modulation of dopamine release by nicotine. Nat Neurosci 7:581–582. https://doi.org/10.1038/nn1243
Zweifel LS, Parker JG, Lobb CJ, Rainwater A, Wall VZ, Fadok JP, Darvas M, Kim MJ, Mizumori SJ, Paladini CA, Phillips PE, Palmiter RD (2009) Disruption of NMDAR-dependent burst firing by dopamine neurons provides selective assessment of phasic dopamine-dependent behavior. Proc Natl Acad Sci USA 106:7281–7288. https://doi.org/10.1073/pnas.0813415106
Acknowledgements
The data here reported are part of the PhD dissertation of A. C-A, who received a doctoral fellowship (CONACYT grant 232728). We wish to thank Mrs. Ángeles Ceja Gálvez for animal care.
Funding
This work was supported by a grant from the Mexican Consejo Nacional de Ciencia y Tecnología (Conacyt) to G. R-M (Grant 220772).
Author information
Authors and Affiliations
Contributions
A. C-A. and G. R-M. conceived and designed the study. A. C-A., U. C., and O. J. performed the experiments and analyzed the data. A. C-A., G. R-M., and F. P. reviewed the intellectual content. A. C-A. and G. R-M. wrote the manuscript. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
We declare that we have complied with the ethical principles regarding care and use of animals in the conduct of the research presented in this manuscript. The Local Committees of Ethics on Animal Experimentation approved all experimental procedures (protocols NC093230.0 and CICUAL 0230–16), which followed the regulations established in the Mexican Official Norm for the use and care of laboratory animals NOM-062-ZOO-1999.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Canseco-Alba, A., Coffeen, U., Jaimes, O. et al. The nucleus accumbens dopamine increase, typically triggered by sexual stimuli in male rats, is no longer produced when animals are sexually inhibited due to sexual satiety. Psychopharmacology 239, 3679–3695 (2022). https://doi.org/10.1007/s00213-022-06240-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-022-06240-3