Abstract
Rationale
Agonists of the kappa opioid receptor (KOR) have been shown to block the rewarding effects of drugs of abuse, but with negative side effects. The antipruritic drug nalfurafine, approved in Japan in 2009, is a potent, selective KOR agonist that does not cause significant side effects in humans. Nalfurafine has not been extensively tested for its effect on drug reward and reinforcement in preclinical models.
Objectives
The goal of this study was to compare the effects of nalfurafine and a reference KOR agonist for a variety of KOR-mediated endpoints in male C57BL6 mice. Specifically, we aimed to evaluate the “therapeutic window”—doses of agonists lower than those eliciting negative side effects, while still effective for desired therapeutic effects.
Methods
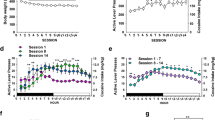
In this study, several low doses of nalfurafine and U50,488 were tested for serum prolactin release, rotarod-mediated sedation, and place-conditioning in male C57BL6 mice. These agonists were also tested for effects on intravenous cocaine self-administration, both on an FR1 schedule and on a progressive ratio schedule for 0.5 mg/kg/infusion cocaine.
Results
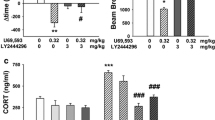
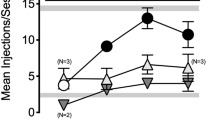
Serum prolactin levels increased following doses of both nalfurafine (3 μg/kg and 10 μg/kg) and U50,488 (3 mg/kg). These doses did not cause sedation in the rotarod assay or aversion in a place-conditioning assay, but blocked conditioned place preference for cocaine. Immediate pretreatment of mice with 10 μg/kg nalfurafine and 3 mg/kg U50,488, however, potentiated cocaine self-administration. Further 10 μg/kg nalfurafine was also observed to potentiate cocaine-seeking behavior as demonstrated by increased progressive ratio break point.
Conclusions
Both nalfurafine and U50,488 showed a separation of negative side effects and the modulation of cocaine reward, suggesting this effect of KOR agonists at low doses may be characteristic of the KOR system in general. At higher doses, nalfurafine had similar effects to traditional KOR agonists like U50,488, indicating that its relative potency, rather than differences in KOR signaling, may be responsible for its unique effects in humans.









Similar content being viewed by others
References
Albert-Vartanian A, Boyd MR, Hall AL et al (2016) Will peripherally restricted kappa-opioid receptor agonists (pKORAs) relieve pain with less opioid adverse effects and abuse potential? J Clin Pharm Ther 41:371–382. https://doi.org/10.1111/jcpt.12404
Balcombe JP, Barnard ND, Sandusky C (2004) Laboratory routines cause animal stress. Contemp Top Lab Anim Sci 43(6):42–51
Bardo MT, Bevins RA (2000) Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology 153:31–43
Bourgeois C, Werfel E, Galla F et al (2014) Synthesis and pharmacological evaluation of 5-pyrrolidinylquinoxalines as a novel class of peripherally restricted κ-opioid receptor agonists. J Med Chem 57:6845–6860. https://doi.org/10.1021/jm500940q
Brust TF, Morgenweck J, Kim SA et al (2016) Biased agonists of the kappa opioid receptor suppress pain and itch without causing sedation or dysphoria. Sci Signal 9:ra117–ra117. https://doi.org/10.1126/scisignal.aai8441
Caine SB, Koob GF (1994) Effects of dopamine D-1 and D-2 antagonists on cocaine self-administration under different schedules of reinforcement in the rat. J Pharmacol Exp Ther 270
Cervo L, Samanin R (1995) Effects of dopaminergic and glutamatergic receptor antagonists on the acquisition and expression of cocaine conditioning place preference. Brain Res 673:242–250. https://doi.org/10.1016/0006-8993(94)01420-M
Collins RJ, Weeks JR, Cooper MM et al (1983) Prediction of abuse liability of drugs using IV self-administration by rats. Psychopharmacology 82:6–13. https://doi.org/10.1007/BF00426372
Di Chiara G, Imperato A (1988) Opposite effects of mu and kappa opiate agonists on dopamine release in the nucleus accumbens and in the dorsal caudate of freely moving rats. J Pharmacol Exp Ther 244
Dunn AD, Reed B, Guariglia C et al (2018) Structurally related kappa opioid receptor agonists with substantial differential signaling bias: neuroendocrine and behavioral effects in C57BL6 Mice. Int J Neuropsychopharmacol 21:847–857. https://doi.org/10.1093/ijnp/pyy034
Dunn AD, Reed B, Erazo J et al (2019) Signaling properties of structurally diverse kappa opioid receptor ligands: toward in vitro models of in vivo responses. ACS Chem Neurosci 10:3590–3600. https://doi.org/10.1021/acschemneuro.9b00195
Hasebe K, Kawai K, Suzuki T et al (2004) Possible pharmacotherapy of the opioid κ receptor agonist for drug dependence. Ann N Y Acad Sci 1025:404–413. https://doi.org/10.1196/annals.1316.050
Kozono H, Yoshitani H, Nakano R (2018) Post-marketing surveillance study of the safety and efficacy of nalfurafine hydrochloride (Remitch® capsules 2.5 μg) in 3,762 hemodialysis patients with intractable pruritus. Int J Nephrol Renovasc Dis Volume 11:9–24. https://doi.org/10.2147/IJNRD.S145720
Kreek MJ (1997) Opiate and cocaine addictions: challenge for pharmacotherapies. Pharmacol Biochem Behav 57:551–569. https://doi.org/10.1016/S0091-3057(96)00440-6
Kreek MJ, LaForge KS, Butelman E (2002) Pharmacotherapy of addictions. Nat Rev Drug Discov 1:710–726. https://doi.org/10.1038/nrd897
Kumagai H, Ebata T, Takamori K et al (2012) Efficacy and safety of a novel ĸ-agonist for managing intractable pruritus in dialysis patients. Am J Nephrol 36:175–183. https://doi.org/10.1159/000341268
Kuzmin AV, Semenova S, Gerrits MAF et al (1997) κ-Opioid receptor agonist U50,488H modulates cocaine and morphine self-administration in drug-naive rats and mice. Eur J Pharmacol 321:265–271. https://doi.org/10.1016/S0014-2999(96)00961-2
Lew G, Parker LA (1998) Pentobarbital-induced place aversion learning. Anim Learn Behav 26:219–224. https://doi.org/10.3758/BF03199214
Liu JJ, Sharma K, Zangrandi L et al (2018) In vivo brain GPCR signaling elucidated by phosphoproteomics. Science (80- ) 360:eaao4927. https://doi.org/10.1126/science.aao4927
Liu JJ, Chiu Y-T, DiMattio KM et al (2019) Phosphoproteomic approach for agonist-specific signaling in mouse brains: mTOR pathway is involved in κ opioid aversion. Neuropsychopharmacology 44:939–949. https://doi.org/10.1038/s41386-018-0155-0
MacLean KA, Johnson MW, Reissig CJ et al (2013) Dose-related effects of salvinorin A in humans: dissociative, hallucinogenic, and memory effects. Psychopharmacology 226:381–392. https://doi.org/10.1007/s00213-012-2912-9
McLaughlin JP, Land BB, Li S et al (2006) Prior activation of kappa opioid receptors by U50,488 mimics repeated forced swim stress to potentiate cocaine place preference conditioning. Neuropsychopharmacology 31:787–794. https://doi.org/10.1038/sj.npp.1300860
Meehan SM, Schechter MD (1998) LSD produces conditioned place preference in male but not female fawn hooded rats. Pharmacol Biochem Behav 59:105–108. https://doi.org/10.1016/S0091-3057(97)00391-2
Mello NK, Negus SS (1998) Effects of kappa opioid agonists on cocaine- and food-maintained responding by rhesus monkeys. J Pharmacol Exp Ther 286:812–824
Mores KL, Cummins BR, Cassell RJ, van Rijn RM (2019) A review of the therapeutic potential of recently developed G protein-biased kappa agonists. Front Pharmacol 10:407. https://doi.org/10.3389/fphar.2019.00407
Mori T, Nomura M, Nagase H et al (2002) Effects of a newly synthesized κ-opioid receptor agonist, TRK-820, on the discriminative stimulus and rewarding effects of cocaine in rats. Psychopharmacology 161:17–22. https://doi.org/10.1007/s00213-002-1028-z
Nagase H, Watanabe A, Nemoto T et al (2010) Drug design and synthesis of a novel kappa opioid receptor agonist with an oxabicyclo[2.2.2]octane skeleton and its pharmacology. Bioorg Med Chem Lett 20:121–124. https://doi.org/10.1016/j.bmcl.2009.11.027
Nemoto T, Yamamoto N, Wada N et al (2013) The effect of 17-N substituents on the activity of the opioid κ receptor in nalfurafine derivatives. Bioorg Med Chem Lett 23:268–272. https://doi.org/10.1016/J.BMCL.2012.10.100
Pande AC, Pyke RE, Greiner M et al (1996) Analgesic efficacy of enadoline versus placebo or morphine in postsurgical pain. Clin Neuropharmacol 19:451–456. https://doi.org/10.1097/00002826-199619050-00009
Schattauer SS, Kuhar JR, Song A, Chavkin C (2017) Nalfurafine is a G-protein biased agonist having significantly greater bias at the human than rodent form of the kappa opioid receptor. Cell Signal 32:59–65. https://doi.org/10.1016/j.cellsig.2017.01.016
Stevens Negus S (2004) Effects of the kappa opioid agonist U50,488 and the kappa opioid antagonist nor-binaltorphimine on choice between cocaine and food in rhesus monkeys. Psychopharmacology 176:204–213. https://doi.org/10.1007/s00213-004-1878-7
Togashi Y, Umeuchi H, Okano K et al (2002) Antipruritic activity of the kappa-opioid receptor agonist, TRK-820. Eur J Pharmacol 435:259–264. https://doi.org/10.1016/s0014-2999(01)01588-6
Wadenberg M-LG (2003) A review of the properties of spiradoline: a potent and selective kappa-opioid receptor agonist. CNS Drug Rev 9:187–198
Wang Y, Tang K, Inan S et al (2005) Comparison of pharmacological activities of three distinct kappa ligands (Salvinorin A, TRK-820 and 3FLB) on kappa opioid receptors in vitro and their antipruritic and antinociceptive activities in vivo. J Pharmacol Exp Ther 312:220–230. https://doi.org/10.1124/jpet.104.073668
White KL, Robinson JE, Zhu H et al (2015) The G protein-biased κ-opioid receptor agonist RB-64 is analgesic with a unique spectrum of activities in vivo. J Pharmacol Exp Ther 352:98–109
Zhang Y, Butelman ER, Schlussman SD et al (2004) Effect of the endogenous κ opioid agonist dynorphin A(1-17) on cocaine-evoked increases in striatal dopamine levels and cocaine-induced place preference in C57BL/6J mice. Psychopharmacology 172:422–429. https://doi.org/10.1007/s00213-003-1688-3
Zhou Y, Kreek MJ (2019) Combination of clinically utilized kappa-opioid receptor agonist nalfurafine with low-dose naltrexone reduces excessive alcohol drinking in male and female mice. Alcohol Clin Exp Res 43:1077–1090. https://doi.org/10.1111/acer.14033
Acknowledgments
We gratefully acknowledge Dr. Eduardo Butelman for intellectual contributions and Dr. Yong Zhang for valuable guidance on mouse self-administration experiments. We thank Dr. Linda Rorick-Kehn for facilitating the generous provision by Eli Lilly and Co. of a small portion of LY2444296. We thank the Rockefeller University Comparative Biosciences Center and Jonathon Negron for animal husbandry.
Conflict of interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by the Dr. Miriam and Sheldon Adelson Medical Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animals were housed and euthanized in a manner approved by The Rockefeller University Institutional Animal Care and Use Committee.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dunn, A., Windisch, K., Ben-Ezra, A. et al. Modulation of cocaine-related behaviors by low doses of the potent KOR agonist nalfurafine in male C57BL6 mice. Psychopharmacology 237, 2405–2418 (2020). https://doi.org/10.1007/s00213-020-05543-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05543-7