Abstract
Rationale
The smallest difference in nicotine that can change a smoker’s cigarette preference is not clearly known.
Objective
A procedure to efficiently identify the difference in nicotine needed to change cigarette preference could help inform research to gauge effects of a nicotine reduction policy.
Methods
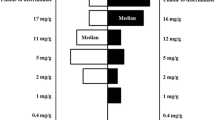
Using a within-subject design, we assessed preference for research cigarettes varying in nicotine contents (NIC; 18.7, 10.8, 5.3, 2.3, and 1.3 mg/g of tobacco), relative to a very low nicotine cigarette (VLNC; 0.4 mg/g), in 17 adult-dependent non-menthol smokers abstinent overnight. Only one NIC was compared vs. the VLNC per session, with order of the five NIC contents randomized across sessions on five separate days. Preference for each NIC vs. VLNC was determined by validated forced choice procedure, with those NIC chosen more than VLNC indicating greater reinforcement due to greater nicotine per se. Secondarily, less preference for lower NIC (vs. VLNC), relative to choice for the highest NIC, 18.7 mg/g (vs. VLNC), indexed reduced reinforcement.
Results
Overall, NIC choices increased as their nicotine increased, as anticipated. Relative to the 0.4 mg/g VLNC, choice was greater for NIC ≥ 5.3 mg/g but not ≤ 2.3 mg/g. Correspondingly, relative to choice for 18.7 mg/g, choice was less for NIC ≤ 2.3 mg/g but not ≥ 5.3 mg/g.
Conclusions
Although replication with larger samples and longer access is needed, results indicate that nicotine reduction to ≤ 2.3 mg/g in cigarettes would attenuate reinforcement. This choice procedure may efficiently inform future clinical trials to assess relative reinforcing effects of smoking reduced nicotine cigarettes.

Similar content being viewed by others
References
Ai J, Taylor KM, Lisko JG, Tran H, Watson CH, Holman MR (2015) Menthol content in US marketed cigarettes. Nicotine Tob Res 18:1575–1580. https://doi.org/10.1093/ntr/ntv162
American Psychiatric Association (APA) (2013) Diagnostic and statistical manual-V. American Psychiatric Association, Washington DC ISBN 978-0-89042-556-5
Benowitz NL, Henningfield JE (2013) Reducing the nicotine content to make cigarettes less addictive. Tob Control 22:i14–i17. https://doi.org/10.1136/tobaccocontrol-2012-050860
Berman ML, Glasser AM (2019) Nicotine reduction in cigarettes: literature review and gap analysis. Nicotine Tob Res 21(suppl 1):S133–SS44. https://doi.org/10.1093/ntr/ntz162
Blendy JA, Strasser A, Walters CL, Perkins KA, Patterson F, Berkowitz R, Lerman C (2005) Reduced nicotine reward in obesity: cross comparison in human and mouse. Psychopharmacol. 180(2):306–315. https://doi.org/10.1007/s00213-005-2167-9
Carmines E, Gillman IG (2019) Comparison of the yield of very low nicotine content cigarettes to the top 100 United States brand styles. Beiträge zur Tabakforschung International/Contributions to Tobacco Research 28:253–266. https://doi.org/10.2478/cttr-2019-0005
Carter LP, Stitzer ML, Henningfield JE, O’Connor RJ, Cummings KM, Hatsukami DK (2009) Abuse liability assessment of tobacco products including potential reduced exposure products. Cancer Epidemiol Biomark Prev 18:3241–4262. https://doi.org/10.1158/1055-9965.EPI-09-0948
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. L. Erlbaum Associates, Hillsdale
Dermody SS, Donny EC, Hertsgaard LA, Hatsukami DK (2015) Greater reductions in nicotine exposure while smoking very low nicotine content cigarettes predict smoking cessation. Tob Control 24:536–539. https://doi.org/10.1136/tobaccocontrol-2014-051797
Donny EC, Denlinger RL, Tidey JW, Koopmeiners JS, Benowitz NL, Vandrey RG, al’Absi M, Carmella SG, Cinciripini PM, Dermody SS, Drobes DJ, Hecht SS, Jensen J, Lane T, le CT, McClernon F, Montoya ID, Murphy SE, Robinson JD, Stitzer ML, Strasser AA, Tindle H, Hatsukami DK (2015) Randomized trial of reduced-nicotine standards for cigarettes. N Engl J Med 373:1340–1349. https://doi.org/10.1056/NEJMsa1502403
Drope J, Liber AC, Cahn Z, Stoklosa M, Kennedy R, Douglas CD, Henson R, Drope J (2018) Who’s still smoking? Disparities in adult cigarette smoking prevalence in the United States. CA Cancer J Clin 68:106–115
Faulkner P, Ghahremani DG, Tyndale RF, Cox CM, Kazanjian AS, Paterson N et al (2017) Reduced-nicotine cigarettes in young smokers: impact of nicotine metabolism on nicotine dose effects. Neuropsychopharmacol 42:1610–1618
Food and Drug Administration (FDA) (2018) Tobacco Product Standard for Nicotine Level of Combusted Cigarettes. A proposed rule. Document citation: 83 FR 11818, pages 11818–11843. Document number: 2018–05345 (accessed 10/29/2019)
Frieden TR (2017) Evidence for health decision making—beyond randomized, controlled trials. New Engl J Med 377:465–475
GBD 2015 Tobacco Collaborators (2017) Smoking prevalence and attributable disease burden in 195 countries and territories, 1990–2015: a systematic analysis from the Global Burden of Disease Study 2015. Lancet 389:1885–1906. https://doi.org/10.1016/S0140-6736(17)30819-X
Grebenstein PE, Burroughs D, Roiko SA, Pentel PR, LeSage MG (2015) Predictors of the nicotine reinforcement threshold, compensation, and elasticity of demand in a rodent model of nicotine reduction policy. Drug Alcohol Depend 151:181–193. https://doi.org/10.1016/j.drugalcdep.2015.03.030
Gu X, Lohrenz T, Salas R, Baldwin PR, Soltani A, Kirk U, Cinciripini PM, Montague PR (2015) Belief about nicotine selectively modulates value and reward prediction error signals in smokers. Proc Natl Acad Sci 112:2539–2544. https://doi.org/10.1073/pnas.1416639112
Hasenfratz M, Jacober A, Battig K (1993) Smoking-related subjective and physiological changes: pre- to postpuff and pre- to postcigarette. Pharmacol Biochem Behav 46:527–534. https://doi.org/10.1016/0091-3057(93)90540-A
Hatsukami DK, Heishman SJ, Vogel RI, Denlinger RL, Roper-Batker AN, Mackowick KM, Jensen J, Murphy SE, Thomas BF, Donny E (2013) Dose–response effects of spectrum research cigarettes. Nicotine Tob Res 15:1113–1121. https://doi.org/10.1093/ntr/nts247
Hatsukami DK, Luo X, Jensen JA, al’Absi M, Allen SS, Carmella SG et al (2018) Effect of immediate vs gradual reduction in nicotine content of cigarettes on biomarkers of smoke exposure: a randomized clinical trial. JAMA 320:880–891. https://doi.org/10.1001/jama.2018.11473
Heatherton TF, Kozlowski LT, Frecker RC, Fagerstrom K-O (1991) The Fagerstrom Test for Nicotine Dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict 86:1119–1127. https://doi.org/10.1111/j.1360-0443.1991.tb01879.x
Higgins ST, Heil SH, Sigmon SC, Tidey JW, Gaalema D, Hughes JR, Stitzer ML, Durand H, Bunn JY, Priest JS, Arger CA, Miller ME, Bergeria CL, Davis DR, Streck JM, Reed DD, Skelly JM, Tursi L (2017) Addiction potential of cigarettes with reduced nicotine content in populations with psychiatric disorders and other vulnerabilities to tobacco addiction. JAMA Psychiatry 74(10):1056–1064. https://doi.org/10.1001/jamapsychiatry.2017.2355
Kamens HM, Silva CP, Nye RT, Miller CN, Singh M, Sipko J et al (2020) Pharmacokinetic profile of Spectrum reduced nicotine cigarettes. Nicotine Tob Res 22(2):273–279. https://doi.org/10.1093/ntr/ntz045
Mathieu JE, Aguinis H, Culpepper SA, Chen G (2012) Understanding and estimating the power to detect cross-level interaction effects in multilevel modeling. J Appl Psychol 97:951–966. https://doi.org/10.1037/a0028380
Mooney M, Green C, Hatsukami D (2006) Nicotine self-administration: cigarettes versus nicotine gum diurnal topography. Hum Psychopharmacol Clin Exp 21(8):539–548. https://doi.org/10.1002/hup.80
Panlilio L, Goldberg SR (2017) Self-administration of drugs in animals and humans as a model and an investigative tool. Addiction 102:1863–1870
Perkins KA (2019) Research on behavioral discrimination of nicotine may inform FDA policy on setting a maximum nicotine content in cigarettes. Nicotine Tob Res 21(suppl 1):S5–S12. https://doi.org/10.1093/ntr/ntz136
Perkins KA, Karelitz JL (2019a) A forced choice procedure to assess the acute relative reinforcing effects of nicotine dose per se in humans. Nicotine Tob Res; in press. https://doi.org/10.1093/ntr/ntz224
Perkins KA, Karelitz JL (2019b) A procedure to standardize puff topography during evaluations of acute tobacco or electronic cigarette exposure. Nicotine Tob Res; in press. https://doi.org/10.1093/ntr/nty261
Perkins KA, Karelitz JL (2019c) Acute perceptions of preferred cigarettes when blinded to brand. Tob Control 28(3):311–316. https://doi.org/10.1136/tobaccocontrol-2018-054388
Perkins KA, Grobe JE, Weiss D, Fonte C, Caggiula A (1996) Nicotine preference in smokers as a function of smoking abstinence. Pharmacol Biochem Behav 55(2):257–263. https://doi.org/10.1016/S0091-3057(96)00079-2
Perkins KA, Karelitz JL, Giedgowd GE, Conklin CA (2012) The reliability of puff topography and subjective responses during ad lib smoking of a single cigarette. Nicotine Tob Res 14:490–494. https://doi.org/10.1093/ntr/ntr150
Perkins KA, Herb T, Karelitz JL (2019) Discrimination of nicotine content in electronic cigarettes. Addict Behav 91:106–111. https://doi.org/10.1016/j.addbeh.2018.05.027
Richter P, Pappas RS, Bravo R, Lisko JG, Damian M, Gonzalez-Jimenez N et al (2016) Characterization of SPECTRUM variable nicotine research cigarettes. Tob Regul Sci 2(2):94–105. https://doi.org/10.18001/TRS.2.2.1
Rose JE, Salley A, Behm FM, Bates JE, Westman EC (2010) Reinforcing effects of nicotine and non-nicotine components of cigarette smoke. Psychopharmacology 210:1–12. https://doi.org/10.1007/s00234-010-1810-2
Simonavicius E, McNeill A, Shahab L, Brose LS (2019) Heat-not-burn tobacco products: a systematic literature review. Tob Control 28:582–594. https://doi.org/10.1136/tobaccocontrol-2018-054419
Smith TT, Koopmeiners JS, Tessier KM, Davis EM, Conklin CA, Denlinger-Apte RL, Lane T, Murphy SE, Tidey JW, Hatsukami DK, Donny EC (2019) Randomized trial of low-nicotine cigarettes and transdermal nicotine. Am J Prev Med 57:515–524. https://doi.org/10.1016/j.amepre.2019.05.010
Snijders TB, Bosker RJ (2012) Multilevel analysis: an introduction to basic and advanced multilevel modeling. Sage, Los Angeles ISBN 9781849202008
Stolerman IP, Jarvis MJ (1995) The scientific case that nicotine is addictive. Psychopharmacology 117:2–10. https://doi.org/10.1007/BF02245088
Tucker MR, Laugesen M, Grace RC (2018) Estimating demand and cross-price elasticity for very low nicotine content (VLNC) cigarettes using a simulated demand task. Nicotine Tob Res 20:843–850. https://doi.org/10.1093/ntr/ntx051
Tunis SR, Stryer DB, Clancy CM (2003) Practical clinical trials: increasing the value of clinical research for decision making in clinical and health policy. JAMA 290:1624–1632. https://doi.org/10.1001/jama.290.12.1624
U.S. Department of Health and Human Services (USDHHS) (2014) The health consequences of smoking—50 years of progress: a report of the surgeon general. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, Atlanta
World Health Organization (WHO) (2015) Advisory note: global nicotine reduction strategy. WHO Study Group on Tobacco Product Regulation. WHO Press, Geneva
World Health Organization (WHO) (2019) WHO global report on trends in prevalence of tobacco use 2000–2025, third edn. WHO Press, Geneva
Funding
Research reported in this publication was supported by the National Institute on Drug Abuse of the National Institutes of Health under grant awards R21 DA035968 and R01 DA035774 (KAP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
These study procedures were approved by the University of Pittsburgh Institutional Review Board and are in accordance with the Helsinki Declaration of 1975.
Conflict of interest
The authors declare that they have no conflict of interest.
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Perkins, K.A., Karelitz, J.L. Differences in acute reinforcement across reduced nicotine content cigarettes. Psychopharmacology 237, 1885–1891 (2020). https://doi.org/10.1007/s00213-020-05509-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05509-9