Abstract
Rationale
The neuropeptide galanin has been implicated in a wide range of pathological conditions in which frontal and temporal structures are compromised. It works through three subtypes of G-protein-coupled receptors. One of these, the galanin receptor 1 (Gal-R1) subtype, is densely expressed in the ventral hippocampus (vHC) and ventral prefrontal cortex (vPFC); two brain structures that have similar actions on behavioral control. We hypothesize that Gal-R1 contributes to cognitive-control mechanisms that require hippocampal-prefrontal cortical circuitry.
Objective
To examine the effect of local vHC and vPFC infusions of M617, a Gal-R1 agonist, on inhibitory mechanisms of response control.
Methods
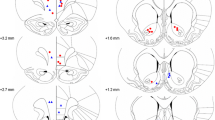
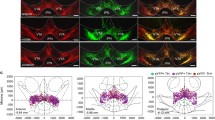
Different cohorts of rats were implanted with bilateral guide cannulae targeting the vPFC or the vHC. Following infusion of the Gal-R1 agonist, we examined the animals’ behavior using a touchscreen version of the 5-choice reaction time task (5-choice task).
Results
The Gal-R1 agonist produced opposing behaviors in the vPFC and vHC, leading to disruption of impulse control when infused in the vPFC but high impulse control when infused into the vHC. This contrast between areas was accentuated when we added variability to the timing of the stimulus, which led to long decision times and reduced accuracy in the vPFC group but a general improvement in performance accuracy in the vHC group.
Conclusions
These results provide the first evidence of a selective mechanism of Gal-R1–mediated modulation of impulse control in prefrontal-hippocampal circuitry.





Similar content being viewed by others
References
Abela AR, Chudasama Y (2013) Dissociable contributions of the ventral hippocampus and orbitofrontal cortex to decision-making with a delayed or uncertain outcome. Eur J Neurosci 37:640–647
Abela AR, Chudasama Y (2014) Noradrenergic alpha2A-receptor stimulation in the ventral hippocampus reduces impulsive decision-making. Psychopharmacology 231:521–531
Abela AR, Dougherty SD, Fagen ED, Hill CJ, Chudasama Y (2013) Inhibitory control deficits in rats with ventral hippocampal lesions. Cereb Cortex 23:1396–1409
Aghajanian GK, Cedarbaum JM, Wang RY (1977) Evidence for norepinephrine-mediated collateral inhibition of locus coeruleus neurons. Brain Res 136:570–577
Aston-Jones G, Bloom FE (1981) Activity of norepinephrine-containing locus coeruleus neurons in behaving rats anticipates fluctuations in the sleep-waking cycle. J Neurosci 1:876–886
Aston-Jones G, Rajkowski J, Cohen J (1999) Role of locus coeruleus in attention and behavioral flexibility. Biol Psychiatry 46:1309–1320
Bai YF, Ma HT, Liu LN, Li H, Li XX, Yang YT, Xue B, Wang D, Xu ZD (2018) Activation of galanin receptor 1 inhibits locus coeruleus neurons via GIRK channels. Biochem Biophys Res Commun 503:79–85
Bannerman DM, Yee BK, Good MA, Heupel MJ, Iversen SD, Rawlins JN (1999) Double dissociation of function within the hippocampus: a comparison of dorsal, ventral, and complete hippocampal cytotoxic lesions. Behav Neurosci 113:1170–1188
Beal MF, MacGarvey U, Swartz KJ (1990) Galanin immunoreactivity is increased in the nucleus basalis of Meynert in Alzheimer’s disease. Ann Neurol 28:157–161
Bellani M, Hatch JP, Nicoletti MA, Ertola AE, Zunta-Soares G, Swann AC, Brambilla P, Soares JC (2012) Does anxiety increase impulsivity in patients with bipolar disorder or major depressive disorder? J Psychiatr Res 46:616–621
Berkun KE, Messanvi F, Wang HA, Tejeda H, Fields R, Chudasama Y (2019) Galanin receptor 1 in the ventral prefrontal cortex and ventral hippocampus: Implications for fronto-temporal modulation of impulse control. Soc. Neurosci. Abstr. #774.11, Chicago, IL, USA
Bowser R, Kordower JH, Mufson EJ (1997) A confocal microscopic analysis of galaninergic hyperinnervation of cholinergic basal forebrain neurons in Alzheimer’s disease. Brain Pathol 7:723–730
Branchek TA, Smith KE, Gerald C, Walker MW (2000) Galanin receptor subtypes. Trends Pharmacol Sci 21:109–117
Carli M, Robbins TW, Evenden JL, Everitt BJ (1983) Effects of lesions to ascending noradrenergic neurones on performance of a 5-choice serial reaction task in rats; implications for theories of dorsal noradrenergic bundle function based on selective attention and arousal. Behav Brain Res 9:361–380
Cardinal RN, Aitken MRF (2010) Whisker: a client-server high-performance multimedia research control system. Behav Res Methods 42:1059–1071.
Chan-Palay V (1988) Galanin hyperinnervates surviving neurons of the human basal nucleus of Meynert in dementias of Alzheimer’s and Parkinson’s disease: a hypothesis for the role of galanin in accentuating cholinergic dysfunction in dementia. J Comp Neurol 273:543–557
Chudasama Y, Passetti F, Rhodes SE, Lopian D, Desai A, Robbins TW (2003) Dissociable aspects of performance on the 5-choice serial reaction time task following lesions of the dorsal anterior cingulate, infralimbic and orbitofrontal cortex in the rat: differential effects on selectivity, impulsivity and compulsivity. Behav Brain Res 146:105–119
Chudasama Y, Doobay VM, Liu Y (2012) Hippocampal-prefrontal cortical circuit mediates inhibitory response control in the rat. J Neurosci 32:10915–10924
Cole BJ, Robbins TW (1992) Forebrain norepinephrine: role in controlled information processing in the rat. Neuropsychopharmacology 7:129–142
Consolo S, Bertorelli R, Girotti P, La Porta C, Bartfai T, Parenti M, Zambelli M (1991) Pertussis toxin-sensitive G-protein mediates galanin’s inhibition of scopolamine-evoked acetylcholine release in vivo and carbachol-stimulated phosphoinositide turnover in rat ventral hippocampus. Neurosci Lett 126:29–32
Counts SE, He B, Che S, Ginsberg SD, Mufson EJ (2008) Galanin hyperinnervation upregulates choline acetyltransferase expression in cholinergic basal forebrain neurons in Alzheimer’s disease. Neurodegener Dis 5:228–231
Crawley JN (1993) Functional interactions of galanin and acetylcholine: relevance to memory and Alzheimer’s disease. Behav Brain Res 57:133–141
Crawley JN, Wenk GL (1989) Co-existence of galanin and acetylcholine: is galanin involved in memory processes and dementia? Trends Neurosci 12:278–282
de Wied D, de Kloet R (1988) Pro-opiomelanocortin and behavioral homeostasis. Hosp Pract (Off Ed) 23(83-8):90–91
Del Carlo A, Benvenuti M, Fornaro M, Toni C, Rizzato S, Swann AC, Dell’Osso L, Perugi G (2012) Different measures of impulsivity in patients with anxiety disorders: a case control study. Psychiatry Res 197:231–236
Elvander E, Schott PA, Sandin J, Bjelke B, Kehr J, Yoshitake T, Ogren SO (2004) Intraseptal muscarinic ligands and galanin: influence on hippocampal acetylcholine and cognition. Neuroscience 126:541–557
Fisone G, Wu CF, Consolo S, Nordstrom O, Brynne N, Bartfai T, Melander T, Hokfelt T (1987) Galanin inhibits acetylcholine release in the ventral hippocampus of the rat: histochemical, autoradiographic, in vivo, and in vitro studies. Proc Natl Acad Sci U S A 84:7339–7343
Genders SG, Scheller KJ, Djouma E (2018) Neuropeptide modulation of addiction: Focus on galanin. Neurosci Biobehav Rev
Gray JA, McNaughton N (1983) Comparison between the behavioural effects of septal and hippocampal lesions: a review. Neurosci Biobehav Rev 7:119–188
Groenewegen HJ, Wright CI, Uylings HBM (1997) The anatomical relationships of the prefrontal cortex with limbic structures and the basal ganglia. J Psychopharmacol 11:99–106
Hokfelt T, Millhorn D, Seroogy K, Tsuruo Y, Ceccatelli S, Lindh B, Meister B, Melander T, Schalling M, Bartfai T et al (1987) Coexistence of peptides with classical neurotransmitters. Experientia 43:768–780
Hokfelt T, Xu ZQ, Shi TJ, Holmberg K, Zhang X (1998) Galanin in ascending systems. Focus on coexistence with 5-hydroxytryptamine and noradrenaline. Ann N Y Acad Sci 863:252–263
Holets VR, Hokfelt T, Rokaeus A, Terenius L, Goldstein M (1988) Locus coeruleus neurons in the rat containing neuropeptide Y, tyrosine hydroxylase or galanin and their efferent projections to the spinal cord, cerebral cortex and hypothalamus. Neuroscience 24:893–906
Holmes A, Heilig M, Rupniak NM, Steckler T, Griebel G (2003) Neuropeptide systems as novel therapeutic targets for depression and anxiety disorders. Trends Pharmacol Sci 24:580–588
Hokfelt T, Barde S, Xu ZD, Kuteeva E, Ruegg J, Le Maitre E, Risling M, Kehr J, Ihnatko R, Theodorsson E, Palkovits M, Deakin W, Bagdy G, Juhasz G, Prud'homme HJ, Mechawar N, Diaz-Heijtz R, Ogren SO (2018) Neuropeptide and Small Transmitter Coexistence: Fundamental Studies and Relevance to Mental Illness. Front Neural Circuits 12: 106.
Jacobowitz DM, Kresse A, Skofitsch G (2004) Galanin in the brain: chemoarchitectonics and brain cartography–a historical review. Peptides 25:433–464
Jakuszkowiak-Wojten K, Landowski J, Wiglusz MS, Cubala WJ (2015) Impulsivity and panic disorder: an exploratory study of psychometric correlates. Psychiatr Danub 27(Suppl 1):S456–S458
Jarrard LE, Isaacson RL (1965) Hippocampal ablation in rats - effects of intertrial interval. Nature 207: 109-&.
Jay TM, Thierry AM, Wiklund L, Glowinski J (1992) Excitatory amino acid pathway from the hippocampus to the prefrontal cortex. Contribution of AMPA receptors in hippocampo-prefrontal cortex transmission. Eur J Neurosci 4:1285–1295
Jhamandas JH, Harris KH, MacTavish D, Jassar BS (2002) Novel excitatory actions of galanin on rat cholinergic basal forebrain neurons: implications for its role in Alzheimer’s disease. J Neurophysiol 87:696–704
Jones DN, Higgins GA (1995) Effect of scopolamine on visual attention in rats. Psychopharmacology 120:142–149
Juhasz G, Hullam G, Eszlari N, Gonda X, Antal P, Anderson IM, Hokfelt TG, Deakin JF, Bagdy G (2014) Brain galanin system genes interact with life stresses in depression-related phenotypes. Proc Natl Acad Sci U S A 111:E1666–E1673
Karlsson RM, Holmes A (2006) Galanin as a modulator of anxiety and depression and a therapeutic target for affective disease. Amino Acids 31: 231-9.
Kjelstrup KG, Tuvnes FA, Steffenach HA, Murison R, Moser EI, Moser MB (2002) Reduced fear expression after lesions of the ventral hippocampus. Proc Natl Acad Sci U S A 99:10825–10830
Kong Q, Yu LC (2013) Antinociceptive effects induced by intra-periaqueductal grey injection of the galanin receptor 1 agonist M617 in rats with morphine tolerance. Neurosci Lett 550:125–128
Kovac S, Walker MC (2013) Neuropeptides in epilepsy. Neuropeptides 47:467–475
Lang R, Gundlach AL, Holmes FE, Hobson SA, Wynick D, Hokfelt T, Kofler B (2015) Physiology, signaling, and pharmacology of galanin peptides and receptors: three decades of emerging diversity. Pharmacol Rev 67:118–175
Laplante F, Crawley JN, Quirion R (2004) Selective reduction in ventral hippocampal acetylcholine release in awake galanin-treated rats and galanin-overexpressing transgenic mice. Regul Pept 122:91–98
Luppi PH, Aston-Jones G, Akaoka H, Chouvet G, Jouvet M (1995) Afferent projections to the rat locus coeruleus demonstrated by retrograde and anterograde tracing with cholera-toxin B subunit and Phaseolus vulgaris leucoagglutinin. Neuroscience 65:119–160
Ma X, Tong YG, Schmidt R, Brown W, Payza K, Hodzic L, Pou C, Godbout C, Hokfelt T, Xu ZQ (2001) Effects of galanin receptor agonists on locus coeruleus neurons. Brain Res 919:169–174
Mariano TY, Bannerman DM, McHugh SB, Preston TJ, Rudebeck PH, Rudebeck SR, Rawlins JN, Walton ME, Rushworth MF, Baxter MG, Campbell TG (2009) Impulsive choice in hippocampal but not orbitofrontal cortex-lesioned rats on a nonspatial decision-making maze task. Eur J Neurosci 30:472–484
Mazarati A, Langel U, Bartfai T (2001) Galanin: an endogenous anticonvulsant? Neuroscientist 7:506–517
McDonald MP, Gleason TC, Robinson JK, Crawley JN (1998) Galanin inhibits performance on rodent memory tasks. Ann N Y Acad Sci 863:305–322
McGaughy J, Dalley JW, Morrison CH, Everitt BJ, Robbins TW (2002) Selective behavioral and neurochemical effects of cholinergic lesions produced by intrabasalis infusions of 192 IgG-saporin on attentional performance in a five-choice serial reaction time task. J Neurosci 22:1905–1913
Moser MB, Moser EI (1998) Functional differentiation in the hippocampus. Hippocampus 8:608–619
Moustafa AA, Tindle R, Frydecka D, Misiak B (2017) Impulsivity and its relationship with anxiety, depression and stress. Compr Psychiatry 74:173–179
Murphy ER, Dalley JW, Robbins TW (2005) Local glutamate receptor antagonism in the rat prefrontal cortex disrupts response inhibition in a visuospatial attentional task. Psychopharmacology 179:99–107
Nishibori M, Oishi R, Itoh Y, Saeki K (1988) Galanin inhibits noradrenaline-induced accumulation of cyclic AMP in the rat cerebral cortex. J Neurochem 51:1953–1955
Nonneman AJ, Voigt J, Kolb BE (1974) Comparisons of behavioral effects of hippocampal and prefrontal cortex lesions in the rat. J Comp Physiol Psychol 87:249–260
O’Donnell D, Ahmad S, Wahlestedt C, Walker P (1999) Expression of the novel galanin receptor subtype GALR2 in the adult rat CNS: distinct distribution from GALR1. J Comp Neurol 409:469–481
Ogren SO, Pramanik A (1991) Galanin stimulates acetylcholine release in the rat striatum. Neurosci Lett 128:253–256
Ogren SO, Kehr J, Schott PA (1996) Effects of ventral hippocampal galanin on spatial learning and on in vivo acetylcholine release in the rat. Neuroscience 75:1127–1140
Ogren SO, Schott PA, Kehr J, Misane I, Razani H (1999) Galanin and learning. Brain Res 848:174–182
Ogren SO, Kuteeva E, Elvander-Tottie E, Hokfelt T (2010) Neuropeptides in learning and memory processes with focus on galanin. Eur J Pharmacol 626:9–17
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates, 5th edn. Elsevier Academic Press, Amsterdam; Boston
Pieribone VA, Xu ZQ, Zhang X, Grillner S, Bartfai T, Hokfelt T (1995) Galanin induces a hyperpolarization of norepinephrine-containing locus coeruleus neurons in the brainstem slice. Neuroscience 64:861–874
Prasad JA, Chudasama Y (2013) Viral tracing identifies parallel disynaptic pathways to the hippocampus. J Neurosci 33:8494–8503
Robinson JK (2004) Galanin and cognition. Behav Cogn Neurosci Rev 3:222–242
Robinson JK, Crawley JN (1994) Analysis of anatomical sites at which galanin impairs delayed nonmatching to sample in rats. Behav Neurosci 108:941–950
Robinson JK, Zocchi A, Pert A, Crawley JN (1996) Galanin microinjected into the medial septum inhibits scopolamine-induced acetylcholine overflow in the rat ventral hippocampus. Brain Res 709:81–87
Rodriguez-Puertas R, Pascual J, Vilaro T, Pazos A (1997) Autoradiographic distribution of M1, M2, M3, and M4 muscarinic receptor subtypes in Alzheimer’s disease. Synapse 26:341–350
Sabbagh JJ, Heaney CF, Bolton MM, Murtishaw AS, Ure JA, Kinney JW (2012) Administration of donepezil does not rescue galanin-induced spatial learning deficits. Int J Neurosci 122:742–747
Schott PA, Bjelke B, Ogren SO (1998) Time-dependent effects of intrahippocampal galanin on spatial learning. Relationship to distribution and kinetics. Ann N Y Acad Sci 863:454–456
Schott PA, Hokfelt T, Ogren SO (2000) Galanin and spatial learning in the rat. Evidence for a differential role for galanin in subregions of the hippocampal formation. Neuropharmacology 39:1386–1403
Svensson TH, Strombom U (1977) Discontinuation of chronic clonidine treatment: evidence for facilitated brain noradrenergic neurotransmission. Naunyn Schmiedeberg's Arch Pharmacol 299:83–87
Swanson LW (1981) A direct projection from Ammon’s horn to prefrontal cortex in the rat. Brain Res 217:150–154
Vertes RP (2006) Interactions among the medial prefrontal cortex, hippocampus and midline thalamus in emotional and cognitive processing in the rat. Neuroscience 142:1–20
Weinshenker D, Holmes PV (2016) Regulation of neurological and neuropsychiatric phenotypes by locus coeruleus-derived galanin. Brain Res 1641:320–337
Weiss JM, Boss-Williams KA, Moore JP, Demetrikopoulos MK, Ritchie JC, West CH (2005) Testing the hypothesis that locus coeruleus hyperactivity produces depression-related changes via galanin. Neuropeptides 39:281–287
Wrenn CC, Kinney JW, Marriott LK, Holmes A, Harris AP, Saavedra MC, Starosta G, Innerfield CE, Jacoby AS, Shine J, Iismaa TP, Wenk GL, Crawley JN (2004) Learning and memory performance in mice lacking the GAL-R1 subtype of galanin receptor. Eur J Neurosci 19:1384–1396
Wrenn CC, Turchi JN, Schlosser S, Dreiling JL, Stephenson DA, Crawley JN (2006) Performance of galanin transgenic mice in the 5-choice serial reaction time attentional task. Pharmacol Biochem Behav 83:428–440
Xu ZQ, Shi TJ, Hokfelt T (1998) Galanin/GMAP- and NPY-like immunoreactivities in locus coeruleus and noradrenergic nerve terminals in the hippocampal formation and cortex with notes on the galanin-R1 and -R2 receptors. J Comp Neurol 392:227–251
Xu ZQ, Tong YG, Hokfelt T (2001) Galanin enhances noradrenaline-induced outward current on locus coeruleus noradrenergic neurons. Neuroreport 12:1779-82.
Yoshitake S, Ijiri S, Kehr J, Yoshitake T (2013) Inhibition of histamine release by local and intracerebroventricular infusion of galanin in hypothalamus, hippocampus and prefrontal cortex of awake rat: a microdialysis study. Neurosci Lett 534:58–63
Yu YJ, Yu YJ, Lin YG (2019) Anxiety and depression aggravate impulsiveness: the mediating and moderating role of cognitive flexibility. Psychol Health Med
Zini S, Roisin MP, Langel U, Bartfai T, Ben-Ari Y (1993) Galanin reduces release of endogenous excitatory amino acids in the rat hippocampus. Eur J Pharmacol 245:1–7
Funding
This research was supported by the Intramural Research Program of the National Institute of Mental Health (ZIA MH002951 and ZIC MH002952 to Y.C.). AP is now at Icahn School of Medicine at Mount Sinai, New York, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Messanvi, F., Perkins, A., du Hoffmann, J. et al. Fronto-temporal galanin modulates impulse control. Psychopharmacology 237, 291–303 (2020). https://doi.org/10.1007/s00213-019-05365-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-019-05365-2