Abstract
Rationale
Chronic exposure to drugs of abuse changes glutamatergic transmission in human addicts and animal models. N-acetylcysteine (NAC) is a cysteine prodrug that indirectly activates cysteine-glutamate antiporters. In the extrasynaptic space, NAC restores basal glutamate levels during drug abstinence and normalizes increased glutamatergic tone in rats during reinstatement to drugs of abuse. In initial clinical trials, repeated NAC administration seems to be promising for reduced craving in cocaine addicts.
Objective
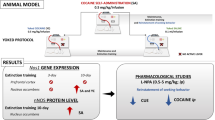
In this study, NAC-amide, called AD4 or NACA, was examined in intravenous cocaine self-administration and extinction/reinstatement procedures in rats. We investigated the behavioral effects of AD4 in the olfactory bulbectomized (OBX) rats, considered an animal model of depression. Finally, we tested rats injected with AD4 or NAC during 10-daily extinction training sessions to examine subsequent cocaine seeking.
Results
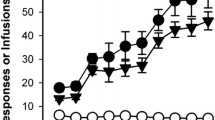
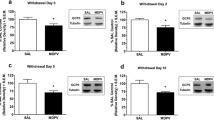
AD4 (25–75 mg kg−1) given acutely did not alter the rewarding effects of cocaine in OBX rats and sham-operated controls. However, at 6.25–50 mg kg−1, AD4 decreased dose-dependently cocaine seeking and relapse triggered by cocaine priming or drug-associated conditioned cues in both phenotypes. Furthermore, repeated treatment with AD4 (25 mg kg−1) or NAC (100 mg kg−1) during daily extinction trials reduced reinstatement of drug-seeking behavior in sham-operated controls. In the OBX rats only, AD4 effectively blocked cocaine-seeking behavior.
Conclusions
Our results demonstrate that AD4 is effective at blocking cocaine-seeking behavior, highlighting its potential clinical use toward cocaine use disorder.





Similar content being viewed by others
References
Amen SL, Piacentine LB, Ahmad ME, Li SJ, Mantsch JR, Risinger RC, et al. (2011) Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology 36:871–878
Amer J, Atlas D, Fibach E (2008) N-acetylcysteine amide (AD4) attenuates oxidative stress in beta-thalassemia blood cells. Biochim Biophys Acta 1780:249–255
Atlas D, Melamed E, Offen D (1999). Brain targeted low molecular weight hydrophobic antioxidant compounds. US Patent No. 5874468
Bachnoff N, Trus M, Atlas D (2011) Alleviation of oxidative stress by potent and selective thioredoxin-mimetic peptides. Free Radic Biol Med 50:1355–1367
Bahat-Stroomza M, Gilgun-Sherki Y, Offen D, Panet H, Saada A, Krool-Galron N, et al. (2005) A novel thiol antioxidant that crosses the blood brain barrier protects dopaminergic neurons in experimental models of Parkinson’s disease. Eur J Neurosci 21:637–646
Baker DA, Shen H, Kalivas PW (2002) Cystine/glutamate exchange serves as the source for extracellular glutamate: modifications by repeated cocaine administration. Amino Acids 23:161–162
Baker DA, McFarland K, Lake RW, Shen H, Tang XC, Toda S, et al. (2003a) Neuroadaptations in cystine-glutamate exchange underlie cocaine relapse. Nat Neurosci 6:743–749
Baker DA, McFarland K, Lake RW, Shen H, Toda S, Kalivas PW (2003b) N-acetyl cysteine-induced blockade of cocaine-induced reinstatement. Ann N Y Acad Sci 1003:349–351
Bavarsad Shahripour R, Harrigan MR, Alexandrov AV (2014) N-acetylcysteine (NAC) in neurological disorders: mechanisms of action and therapeutic opportunities. Brain Behav 4:108–122
Bouton ME (2002) Context, ambiguity, and unlearning: sources of relapse after behavioral extinction. Biol Psychiatry 52:976–986
Brebner K, Wong TP, Liu L, Liu Y, Campsall P, Gray S, et al. (2005) Nucleus accumbens long-term depression and the expression of behavioral sensitization. Science 310:1340–1343
Conklin CA, Tiffany ST (2002) Applying extinction research and theory to cue-exposure addiction treatments. Addiction 97:155–167
Curtis MJ, Bond RA, Spina D, Ahluwalia A, Alexander SP, Giembycz MA, et al. (2015) Experimental design and analysis and their reporting: new guidance for publication in BJP. Br J Pharmacol 172:3461–3471
Deepmala D, Slattery J, Kumar N, Delhey L, Berk M, Dean O, et al. (2015) Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev 55:294–321
Ferreira FR, Biojone C, Joca SR, Guimaraes FS (2008) Antidepressant-like effects of N-acetyl-L-cysteine in rats. Behav Pharmacol 19:747–750
Frankowska M, Jastrzębska J, Nowak E, Białko M, Przegaliński E, Filip M (2014) The effects of N-acetylcysteine on cocaine reward and seeking behaviors in a rat model of depression. Behav Brain Res 266:108–118
Gipson CD, Kupchik YM, Shen H, Reissner KJ, Thomas CA, Kalivas PW (2013) Relapse induced by cues predicting cocaine depends on rapid, transient synaptic potentiation. Neuron 77:867–872
Giustarini D, Milzani A, Dalle-Donne I, Tsikas D, Rossi R (2012) N-acetylcysteine ethyl ester (NACET): a novel lipophilic cell-permeable cysteine derivative with an unusual pharmacokinetic feature and remarkable antioxidant potential. Biochem Pharmacol 84:1522–1533
Grinberg L, Fibach E, Amer J, Atlas D (2005) N-acetylcysteine amide, a novel cell-permeating thiol, restores cellular glutathione and protects human red blood cells from oxidative stress. Free Radic Biol Med 38:136–145
Jastrzębska J, Frankowska M, Szumiec Ł, Sadakierska-Chudy A, Haduch A, Smaga I, et al. (2015) Cocaine self-administration in Wistar-Kyoto rats: a behavioral and biochemical analysis. Behav Brain Res 293:62–73
Kalivas PW (2009) The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 10:561–572
Kau KS, Madayag A, Mantsch JR, Grier MD, Abdulhameed O, Baker DA, et al. (2008) Blunted cystine-glutamate antiporter function in the nucleus accumbens promotes cocaine-induced drug seeking. Neuroscience 155:530–537
Kelly JP, Wrynn AS, Leonard BE (1997) The olfactory bulbectomized rat as a model ofdepression: an update. Pharmacol Ther 74:299–316
Knackstedt LA, Melendez RI, Kalivas PW (2010) Ceftriaxone restores glutamate homeostasis and prevents relapse to cocaine seeking. Biol Psychiatr 67:81–84
Lee KS, Kim SR, Park HS, Park SJ, Min KH, Lee KY, et al. (2007) A novel thiol compound, N-acetylcysteine amide, attenuates allergic airway disease by regulating activation of NF-kappaB and hypoxia-inducible factor-1alpha. Exp Mol Med 39:756–768
Leonard BE, Tuite M (1981) Anatomical physiological, and behavioral aspects of olfactory bulbectomy in the rat. Int Rev Neurobiol 22:251–286
Lominac KD, Sacramento AD, Szumlinski KK, Kippin TE (2012) Distinct neurochemical adaptations within the nucleus accumbens produced by a history of self-administered vs non-contingently administered intravenous methamphetamine. Neuropsychopharmacology 37:707–722
Madayag A, Lobner D, Kau KS, Mantsch JR, Abdulhameed O, Hearing M, et al. (2007) Repeated N-acetylcysteine administration alters plasticity-dependent effects of cocaine. J Neurosci 51:13968–13976
Madhavan A, Argilli E, Bonci A, Whistler JL (2013) Loss of D2 dopamine receptor function modulates cocaine-induced glutamatergic synaptic potentiation in the ventral tegmental area. J Neurosci 33:12329–12336
McBean GJ, Flynn J (2001) Molecular mechanisms of cystine transport. Biochem Soc Trans 29:717–722
McClure EA, Sonne SC, Winhusen T, Carroll KM, Ghitza UE, McRae-Clark AL, et al. (2014) Achieving cannabis cessation—evaluating N-acetylcysteine treatment (ACCENT): design and implementation of a multi-site, randomized controlled study in the National Institute on Drug Abuse clinical trials network. Contemp Clin Trials 39:211–223
McFarland K, Lapish CC, Kalivas PW (2003) Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci 23:3531–3537
Miguens M, Del Olmo N, Higuera-Matas A, Torres I, García-Lecumberri C, Ambrosio E (2008) Glutamate and aspartate levels in the nucleus accumbens during cocaine self-administration and extinction: a time course microdialysis study. Psychopharmacology 196:303–313
Moussawi K, Pacchioni A, Moran M, Olive MF, Gass JT, Lavin A, et al. (2009) N-acetylcysteine reverses cocaine-induced metaplasticity. Nat Neurosci 12:182–189
Moussawi K, Zhou W, Shen H, Reichel CM, See RE, Carr DB, et al. (2011) Reversing cocaine-induced synaptic potentiation provides enduring protection from relapse. Proc Natl Acad Sci U S A 108:385–390
O’Brien CP (1997) A range of research-based pharmacotherapies for addiction. Science 278:66–70
O’Brien C, Parker K (2006) Drug addiction and drug abuse. In: Brunton JLL (ed) Goodman and Gilman’s the pharmacological basis of therapeutics, 11th edn. McGraw-Hill, New York, pp. 607–627
Offen D, Gilgun-Sherki Y, Barhum Y, Benhar M, Grinberg L, Reich R, et al. (2004) A low molecular weight copper chelator crosses the blood-brain barrier and attenuates experimental autoimmune encephalomyelitis. J Neurochem 89:1241–1251
Pandya JD, Readnower RD, Patel SP, Yonutas HM, Pauly JR, Goldstein GA, et al. (2014) N-acetylcysteine amide confers neuroprotection, improves bioenergetics and behavioral outcome following TBI. Exp Neurol 257:106–113
Penugonda S, Mare S, Goldstein G, Banks WA, Ercal N (2005) Effects of N-acetylcysteine amide (NACA), a novel thiol antioxidant against glutamate-induced cytotoxicity in neuronal cell line PC12. Brain Res 1056:132–138
Pierce RC, Bell K, Duffy P, Kalivas PW (1996) Repeated cocaine augments excitatory amino acid transmission in the nucleus accumbens only in rats having developed behavioral sensitization. J Neurosci 16:1550–1560
Pomierny-Chamioło L, Rup K, Pomierny B, Niedzielska E, Kalivas PW, Filip M (2014) Metabotropic glutamatergic receptors and their ligands in drug addiction. Pharmacol Ther 142:281–305
Quirk GJ, Mueller D (2008) Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology 33:56–72
Reichel CM, Moussawi K, Do PH, Kalivas PW, See RE (2011) Chronic N-acetylcysteine during abstinence or extinction after cocaine self-administration produces enduring reductions in drug seeking. J Pharmacol Exp Ther 337:487–493
Sari Y, Smith KD, Ali PK, Rebec GV (2009) Upregulation of GLT1 attenuates cue-induced reinstatement of cocaine-seeking behavior in rats. J Neurosci 29:9239–9243
Shaham Y, Shalev U, Lu L, De Wit H, Stewart J (2003) The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology 168:3–20
Shen HW, Toda S, Moussawi K, Bouknight A, Zahm DS, Kalivas PW (2009) Altered dendritic spine plasticity in cocaine-withdrawn rats. J Neurosci 29:2876–2884
Smaga I, Pomierny B, Krzyżanowska W, Pomierny-Chamioło L, Miszkiel J, Niedzielska E, et al. (2012) N-acetylcysteine possesses antidepressant-like activity through reduction of oxidative stress: behavioral and biochemical analyses in rats. Prog Neuro-Psychopharmacol Biol Psychiatry 39:280–287
Smaga I, Bystrowska B, Gawliński D, Pomierny B, Stankowicz P, Filip M (2014) Antidepressants and changes in concentration of endocannabinoids and N-acylethanolamines in rat brain structures. Neurotox Res 26:190–206
Wydra K, Golembiowska K, Zaniewska M, Kaminska K, Ferraro L, Fuxe K, et al. (2013) Accumbal and pallidal dopamine, glutamate and GABA overflow during cocaine self-administration and its extinction in rats. Addict Biol 18:307–324
Yan M, Shen J, Person MD, Kuang X, Lynn WS, Atlas D, et al. (2008) Endoplasmic reticulum stress and unfolded protein response in Atm-deficient thymocytes and thymic lymphoma cells are attributable to oxidative stress. Neoplasia 10:160–167
Zhou W, Kalivas PW (2008) N-acetylcysteine reduces extinction responding and induces enduring reductions in cue- and heroin-induced drug-seeking. Biol Psychiatry 63:338–340
Acknowledgments
This study was supported by the statutory funds of the Institute of Pharmacology Polish Academy of Sciences in Krakow (J.J., M.Fr., M.F..F.) and by the H. L. Lauterbach Foundation (D.A.).
Author contributions
J.J., M.Fr., and M.F. participated in research design, performed the experiments, and data analysis. J.J., M.Fr., M.F., and D.A. wrote or contributed to writing of the manuscript. All authors critically reviewed the content and approved the final version for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Joanna Jastrzębska and Malgorzata Frankowska contributed equally to the work.
Rights and permissions
About this article
Cite this article
Jastrzębska, J., Frankowska, M., Filip, M. et al. N-acetylcysteine amide (AD4) reduces cocaine-induced reinstatement. Psychopharmacology 233, 3437–3448 (2016). https://doi.org/10.1007/s00213-016-4388-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4388-5