Abstract
Rationale
Cognitive flexibility is a key component of executive function and is disrupted in major psychiatric disorders. Brain-derived neurotrophic factor (BDNF) exerts neuromodulatory effects on synaptic transmission and cognitive/affective behaviors. However, the causal mechanisms linking BDNF hypofunction with executive deficits are not well understood.
Objectives
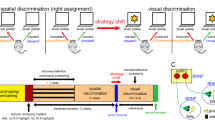
Here, we assessed the consequences of BDNF hemizygosity on cognitive flexibility in mice performing an operant conditioning task. As dopaminergic-glutamatergic interaction in the striatum is important for cognitive processing, and BDNF heterozygous (BDNF+/−) mice display a higher dopamine tone in the dorsal striatum, we also assessed the effects of partial striatal dopamine depletion on task performance and glutamate release.
Results
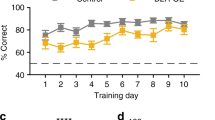
BDNF+/− mice acquired discrimination learning as well as new rule learning during set-shifting as efficiently as wild-type mice. However, partial removal of striatal dopaminergic inputs with 6-hydroxydopamine (6-OHDA) impaired these cognitive processes by impeding the maintenance of a new learning strategy in both genotypes. BDNF mutants exhibited performance impairments during reversal learning, and these deficits were associated with increased perseveration to the previously acquired strategy. Partial dopamine depletion of the striatum reversed these cognitive impairments. Additionally, reduction in depolarization-evoked glutamate release noted in the dorsal striatum of BDNF+/− mice was not observed in 6-OHDA-infused BDNF mutants indicating normalization of glutamatergic transmission in these animals.
Conclusions
Our data illustrate that BDNF signaling regulates cognitive control processes presumably by maintaining striatal dopamine-glutamate balance. Moreover, aberrations in BDNF signaling may act as a common neurobiological substrate that accounts for executive dysfunction observed in multiple psychiatric conditions.






Similar content being viewed by others
References
Agnoli L, Mainolfi P, Invernizzi RW, Carli M (2013) Dopamine D1-like and D2-like receptors in the dorsal striatum control different aspects of attentional performance in the five-choice serial reaction time task under a condition of increased activity of corticostriatal inputs. Neuropsychopharmacology 38:701–714
Altar CA, Boylan CB, Jackson C, Hershenson S, Miller J, Wiegand SJ, Lindsay RM, Hyman C (1992) Brain-derived neurotrophic factor augments rotational behavior and nigrostriatal dopamine turnover in vivo. Proc Natl Acad Sci U S A 89:11347–11351
Autry AE, Monteggia LM (2012) Brain-derived neurotrophic factor and neuropsychiatric disorders. Pharmacol Rev 64:238–258
Balleine BW, O’Doherty JP (2010) Human and rodent homologies in action control: corticostriatal determinants of goal-directed and habitual action. Neuropsychopharmacol 35:48–69
Balleine BW, Delgado MR, Hikosaka O (2007) The role of the dorsal striatum in reward and decision-making. J Neurosci 27:8161–8165
Bamford NS, Robinson S, Palmiter RD, Joyce JA, Moore C, Meshul CK (2004) Dopamine modulates release from corticostriatal terminals. J Neurosci 24:9541–9552
Birbeck JA, Khalid M, Mathews TA (2014) Potentiated striatal dopamine release leads to hyperdopaminergia in female brain-derived neurotrophic factor heterozygous mice. ACS Chem Neurosci 5:275–281
Bolanos CA, Nestler EJ (2004) Neurotrophic mechanisms in drug addiction. Neuromol Med 5:69–83
Bosse KE, Maina FK, Birbeck JA, France MM, Roberts JJ, Colombo ML, Mathews TA (2012) Aberrant striatal dopamine transmitter dynamics in brain-derived neurotrophic factor-deficient mice. J Neurochem 120:385–395
Boulougouris V, Castane A, Robbins TW (2008) Dopamine D2/D3 receptor agonist quinpirole impairs spatial reversal learning in rats: investigation of D3 receptor involvement in persistent behavior. Psychopharmacology 202:611–620
Branchi I, D’Andrea I, Armida M, Carnevale D, Ajmone-Cat MA, Pezzola A, Potenza RL, Morgese MG, Cassano T, Minghetti L, Popoli P, Alleva E (2010) Striatal 6-OHDA lesion in mice: investigating early neurochemical changes underlying Parkinson’s disease. Behav Brain Res 208:137–143
Brigman JL, Feyder M, Saksida LM, Bussey TJ, Mishina M, Holmes A (2008) Impaired discrimination learning in mice lacking the NMDA receptor NR2A subunit. Learn Mem 15:50–54
Brigman JL, Graybeal C, Holmes A (2010) Predictably irrational: assaying cognitive inflexibility in mouse models of schizophrenia. Front Neurosci 4:19–28
Carola V, Gross C (2010) BDNF moderates early environmental risk factors for anxiety in mouse. Genes Brain Behv 9:379–389
Carvalho AL, Caldeira MV, Santos SD, Duarte CB (2008) Role of the brain-derived neurotrophic factor at glutamatergic synapses. Br J Pharmacol 153(Suppl 1):S310–324
Chao MV (2003) Neurotrophins and their receptors: a convergence point for many signalling pathways. Nat Rev Neurosci 4:299–309
Cole RD, Poole RL, Guzman DM, Gould TJ, Parikh V (2015) Contributions of beta2 subunit-containing nAChRs to chronic nicotine-induced alterations in cognitive flexibility in mice. Psychopharmacology 232:1207–1217
Cools R, Ivry RB, D’Esposito M (2006) The human striatum is necessary for responding to changes in stimulus relevance. J Cog Neurosci 18:1973–1983
D’Amore DE, Tracy BA, Parikh V (2013) Exogenous BDNF facilitates strategy set-shifting by modulating glutamate dynamics in the dorsal striatum. Neuropharmacology 75:312–323
Darvas M, Palmiter RD (2011) Contributions of striatal dopamine signaling to the modulation of cognitive flexibility. Biol Psychiatr 69:704–707
Darvas M, Henschen CW, Palmiter RD (2014) Contributions of signaling by dopamine neurons in dorsal striatum to cognitive behaviors corresponding to those observed in Parkinson’s disease. Neurobiol Dis 65:112–123
David HN, Ansseau M, Abraini JH (2005) Dopamine-glutamate reciprocal modulation of release and motor responses in the rat caudate-putamen and nucleus accumbens of “intact” animals. Brain Res Brain Res Rev 50:336–360
del Campo N, Fryer TD, Hong YT, Smith R, Brichard L, Acosta-Cabronero J, Chamberlain SR, Tait R, Izquierdo D, Regenthal R, Dowson J, Suckling J, Baron JC, Aigbirhio FI, Robbins TW, Sahakian BJ, Muller U (2013) A positron emission tomography study of nigro-striatal dopaminergic mechanisms underlying attention: implications for ADHD and its treatment. Brain 136:3252–3270
Ding X, Qiao Y, Piao C, Zheng X, Liu Z, Liang J (2014) N-methyl-D-aspartate receptor-mediated glutamate transmission in nucleus accumbens plays a more important role than that in dorsal striatum in cognitive flexibility. Front Behav Neurosci 8:304
Dirnberger G, Jahanshahi M (2013) Executive dysfunction in Parkinson’s disease: a review. J Neuropsychol 7:193–224
Dreyer JK, Herrik KF, Berg RW, Hounsgaard JD (2010) Influence of phasic and tonic dopamine release on receptor activation. J Neurosci 30:14273–14283
Duman RS, Monteggia LM (2006) A neurotrophic model for stress-related mood disorders. Biol Psychiatr 59:1116–1127
Duman CH, Schlesinger L, Kodama M, Russell DS, Duman RS (2007) A role for MAP kinase signaling in behavioral models of depression and antidepressant treatment. Biol Psychiatr 61:661–670
Egan MF, Kojima M, Callicott JH, Goldberg TE, Kolachana BS, Bertolino A, Zaitsev E, Gold B, Goldman D, Dean M, Lu B, Weinberger DR (2003) The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112:257–269
Eisenberg DP, Berman KF (2010) Executive function, neural circuitry, and genetic mechanisms in schizophrenia. Neuropsychopharmacol 35:258–277
Floresco SB (2013) Prefrontal dopamine and behavioral flexibility: shifting from an “inverted-U” toward a family of functions. Front Neurosci 7:62
Floresco SB, Magyar O, Ghods-Sharifi S, Vexelman C, Tse MT (2006) Multiple dopamine receptor subtypes in the medial prefrontal cortex of the rat regulate set-shifting. Neuropsychopharmacology 31:297–309
Floresco SB, Block AE, Tse MT (2008) Inactivation of the medial prefrontal cortex of the rat impairs strategy set-shifting, but not reversal learning, using a novel, automated procedure. Behav Brain Res 190:85–96
Floresco SB, Zhang Y, Enomoto T (2009) Neural circuits subserving behavioral flexibility and their relevance to schizophrenia. Behav Brain Res 204:396–409
Flores-Hernandez J, Cepeda C, Hernandez-Echeagaray E, Calvert CR, Jokel ES, Fienberg AA, Greengard P, Levine MS (2002) Dopamine enhancement of NMDA currents in dissociated medium-sized striatal neurons: role of D1 receptors and DARPP-32. J Neurophysiol 88:3010–3020
Goggi J, Pullar IA, Carney SL, Bradford HF (2002) Modulation of neurotransmitter release induced by brain-derived neurotrophic factor in rat brain striatal slices in vitro. Brain Res 941:34–42
Goto Y, Otani S, Grace AA (2007) The Yin and Yang of dopamine release: a new perspective. Neuropharmacology 53:583–587
Granseth B, Andersson FK, Lindström SH (2015) The initial stage of reversal learning is impaired in mice hemizygous for the vesicular glutamate transporter (VGluT1). Genes Brain Behav 14:477–485
Haber SN, Fudge JL, McFarland NR (2000) Striatonigrostriatal pathways in primates form an ascending spiral from the shell to the dorsolateral striatum. J Neurosci 20:2369–2382
Haluk DM, Floresco SB (2009) Ventral striatal dopamine modulation of different forms of behavioral flexibility. Neuropsychopharmacol 34:2041–2052
Hascup KN, Hascup ER, Pomerleau F, Huettl P, Gerhardt GA (2008) Second-by-second measures of L-glutamate in the prefrontal cortex and striatum of freely moving mice. J Pharmacol Expt Ther 324:725–731
Howes OD, Kapur S (2009) The dopamine hypothesis of schizophrenia: version III—the final common pathway. Schizophr Bull 35:549–562
Hu Y, Russek SJ (2008) BDNF and the diseased nervous system: a delicate balance between adaptive and pathological processes of gene regulation. J Neurochem 105:1–17
Huang EJ, Reichardt LF (2001) Neurotrophins: roles in neuronal development and function. Ann Rev Neurosci 24:677–736
Izquierdo A, Wiedholz LM, Millstein RA, Yang RJ, Bussey TJ, Saksida LM, Holmes A (2006) Genetic and dopaminergic modulation of reversal learning in a touchscreen-based operant procedure for mice. Behav Brain Res 171:181–188
Jia Y, Gall CM, Lynch G (2010) Presynaptic BDNF promotes postsynaptic long-term potentiation in the dorsal striatum. J Neurosci 30:14440–14445
Jones GH, Robbins TW (1992) Differential effects of mesocortical, mesolimbic, and mesostriatal dopamine depletion on spontaneous, conditioned, and drug-induced locomotor activity. Pharmacol Biochem Behav 43:887–895
Kalivas PW, O’Brien C (2008) Drug addiction as a pathology of staged neuroplasticity. Neuropsychopharmacology 33:166–180
Kalivas PW, Volkow ND (2005) The neural basis of addiction: a pathology of motivation and choice. Am J Psychiatr 162:1403–1413
Kambeitz JP, Bhattacharyya S, Kambeitz-Ilankovic LM, Valli I, Collier DA, McGuire P (2012) Effect of BDNF val(66)met polymorphism on declarative memory and its neural substrate: a meta-analysis. Neurosci Biobehav Rev 36:2165–2177
Klanker M, Feenstra M, Denys D (2013) Dopaminergic control of cognitive flexibility in humans and animals. Front Neurosci 7:201
Lawrence AD, Sahakian BJ, Hodges JR, Rosser AE, Lange KW, Robbins TW (1996) Executive and mnemonic functions in early Huntington’s disease. Brain 119:1633–1645
Lee B, Groman S, London ED, Jentsch JD (2007) Dopamine D2/D3 receptors play a specific role in the reversal of a learned visual discrimination in monkeys. Neuropsychopharmacology 32:2125–2134
Lindholm JS, Castren E (2014) Mice with altered BDNF signaling as models for mood disorders and antidepressant effects. Front Behav Neurosci 8:143
Mega MS, Cummings JL (1994) Frontal-subcortical circuits and neuropsychiatric disorders. J Neuropsychiatr Clin Neurosci 6:358–370
Mehta MA, Manes FF, Magnolfi G, Sahakian BJ, Robbins TW (2004) Impaired set-shifting and dissociable effects on tests of spatial working memory following the dopamine D2 receptor antagonist sulpiride in human volunteers. Psychopharmacology 176:331–342
Minichiello L (2009) TrkB signalling pathways in LTP and learning. Nat Rev Neurosci 10:850–860
Mizuno M, Yamada K, He J, Nakajima A, Nabeshima T (2003) Involvement of BDNF receptor TrkB in spatial memory formation. Learn Mem 10:108–115
Moghaddam B, Krystal JH (2012) Capturing the angel in “angel dust”: twenty years of translational neuroscience studies of NMDA receptor antagonists in animals and humans. Schizophr Bull 38:942–949
Monchi O, Petrides M, Petre V, Worsley K, Dagher A (2001) Wisconsin Card Sorting revisited: distinct neural circuits participating in different stages of the task identified by event-related functional magnetic resonance imaging. J Neurosci 21:7733–7741
Monchi O, Ko JH, Strafella AP (2006) Striatal dopamine release during performance of executive functions: A [(11)C] raclopride PET study. NeuroImage 33:907–912
Moussawi K, Riegel A, Nair S, Kalivas PW (2011) Extracellular glutamate: functional compartments operate in different concentration ranges. Front Syst Neurosci 5:94
Muir JL (1996) Attention and stimulus processing in the rat. Brain Res Cog Brain Res 3:215–225
Nestler EJ, Carlezon WA Jr (2006) The mesolimbic dopamine reward circuit in depression. Biol Psychiatr 59:1151–1159
Nikiforuk A (2012) Dopamine D1 receptor modulation of set shifting: the role of stress exposure. Behav Pharmacol 23:434–438
Notaras M, Hill R, van den Buuse M (2015) The BDNF gene Val66Met polymorphism as a modifier of psychiatric disorder susceptibility: progress and controversy. Mol Psychiatr 20:916–930
Ortega LA, Tracy BA, Gould TJ, Parikh V (2013) Effects of chronic low- and high-dose nicotine on cognitive flexibility in C57BL/6J mice. Behav Brain Res 238:134–145
Oualian C, Gisquet-Verrier P (2010) The differential involvement of the prelimbic and infralimbic cortices in response conflict affects behavioral flexibility in rats trained in a new automated strategy-switching task. Learn Mem 17:654–668
Palmiter RD (2008) Dopamine signaling in the dorsal striatum is essential for motivated behaviors: lessons from dopamine-deficient mice. Ann NY Acad Sci 1129:35–46
Parikh V, Man K, Decker MW, Sarter M (2008) Glutamatergic contributions to nicotinic acetylcholine receptor agonist-evoked cholinergic transients in the prefrontal cortex. J Neurosci 28:3769–3780
Parikh V, Ji J, Decker MW, Sarter M (2010) Prefrontal beta2 subunit-containing and alpha7 nicotinic acetylcholine receptors differentially control glutamatergic and cholinergic signaling. J Neurosci 30:3518–3530
Parikh V, Naughton SX, Shi X, Kelley LK, Yegla B, Tallarida CS, Rawls SM, Unterwald EM (2014) Cocaine-induced neuroadaptations in the dorsal striatum: glutamate dynamics and behavioral sensitization. Neurochem Int 75:54–65
Pillai A, Kale A, Joshi S, Naphade N, Raju MS, Nasrallah H, Mahadik SP (2010) Decreased BDNF levels in CSF of drug-naive first-episode psychotic subjects: correlation with plasma BDNF and psychopathology. Int J Neuropsychopharmacol 13:535–539
Ragozzino ME (2007) The contribution of the medial prefrontal cortex, orbitofrontal cortex, and dorsomedial striatum to behavioral flexibility. Ann NY Aca d Sci 1121:355–375
Sanacora G, Treccani G, Popoli M (2012) Towards a glutamate hypothesis of depression: an emerging frontier of neuropsychopharmacology for mood disorders. Neuropharmacology 62:63–77
Scheggia D, Bebensee A, Weinberger DR, Papaleo F (2014) The ultimate intra-/extra-dimensional attentional set-shifting task for mice. Biol Psychiatr 75:660–670
Shirayama Y, Chen AC, Nakagawa S, Russell DS, Duman RS (2002) Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci 22:3251–3261
Stephens ML, Pomerleau F, Huettl P, Gerhardt GA, Zhang Z (2010) Real-time glutamate measurements in the putamen of awake rhesus monkeys using an enzyme-based human microelectrode array prototype. J Neurosci Methods 185:264–272
Tripanichkul W, Jaroensuppaperch EO (2013) Ameliorating effects of curcumin on 6-OHDA-induced dopaminergic denervation, glial response, and SOD1 reduction in the striatum of hemiparkinsonian mice. Eur Rev Med Pharmacol Sci 17:1360–1368
van Schouwenburg MR, O’Shea J, Mars RB, Rushworth MF, Cools R (2012) Controlling human striatal cognitive function via the frontal cortex. J Neurosci 32:5631–5637
Volkow ND, Wang GJ, Fowler JS, Tomasi D, Telang F, Baler R (2010) Addiction: decreased reward sensitivity and increased expectation sensitivity conspire to overwhelm the brain’s control circuit. BioEssays 32:748–755
West AR, Floresco SB, Charara A, Rosenkranz JA, Grace AA (2003) Electrophysiological interactions between striatal glutamatergic and dopaminergic systems. Ann NY Acad Sci 1003:53–74
Xu J, Zhu Y, Kraniotis S, He Q, Marshall JJ, Nomura T, Stauffer SR, Lindsley CW, Conn PJ, Contractor A (2013) Potentiating mGluR5 function with a positive allosteric modulator enhances adaptive learning. Learn Mem 20:438–445
Zurkovsky L, Bychkov E, Tsakem EL, Siedlecki C, Blakely RD, Gurevich EV (2013) Cognitive effects of dopamine depletion in the context of diminished acetylcholine signaling capacity in mice. Dis Model Mech 6:171–183
Acknowledgments
This work was supported by grants from the Brain and Behavioral Research Foundation, Pennsylvania Department of Health (no. 4100050909) and the National Institute of Health (NIH DA 037421) to V.P. MARC Undergraduate Student Training in Academic Research (NIH 5T34 GM 087239) provided research training support to D.G. We thank Brittany Tracy for the assistance with the genotyping.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experimental procedures were approved by the Institutional Care and Use Committee (IACUC) of Temple University and were in accordance with the National Institute of Health guidelines.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Sean X. Naughton and Brittney Yegla contributed equally to this work.
Rights and permissions
About this article
Cite this article
Parikh, V., Naughton, S.X., Yegla, B. et al. Impact of partial dopamine depletion on cognitive flexibility in BDNF heterozygous mice. Psychopharmacology 233, 1361–1375 (2016). https://doi.org/10.1007/s00213-016-4229-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4229-6