Abstract
Rationale
N-acetylcysteine can increase extrasynaptic glutamate and reduce nicotine self-administration in rats and smoking rates in humans.
Objectives
The aim of this study was to determine if N-acetylcysteine modulates the development of nicotine place conditioning and withdrawal in mice.
Methods
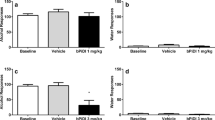
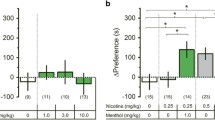
N-acetylcysteine was given to nicotine-treated male ICR mice. Experiment 1: reward-like behavior. N-acetylcysteine (0, 5, 15, 30, or 60 mg/kg, i.p.) was given 15 min before nicotine (0.5 mg/kg, s.c.) or saline (10 ml/kg, s.c.) in an unbiased conditioned place preference (CPP) paradigm. Conditioning for highly palatable food served as control. Experiment 2: spontaneous withdrawal. The effect of N-acetylcysteine (0, 15, 30, 120 mg/kg, i.p.) on anxiety-like behavior, somatic signs, and hyperalgesia was measured 18–24 h after continuous nicotine (24 mg/kg/day, 14 days). Experiment 3: mecamylamine-precipitated, withdrawal-induced aversion. The effect of N-acetylcysteine (0, 15, 30, 120 mg/kg, i.p.) on mecamylamine (3.5 mg/kg, i.p.)-precipitated withdrawal was determined after continuous nicotine (24 mg/kg, i.p., 28 days) using the conditioned place aversion (CPA) paradigm.
Results
Dose-related reductions in the development of nicotine CPP, somatic withdrawal signs, hyperalgesia, and CPA were observed after N-acetylcysteine pretreatment. No effect of N-acetylcysteine was found on palatable food CPP, anxiety-like behavior, or motoric capacity (crosses between plus maze arms). Finally, N-acetylcysteine did not affect any measure in saline-treated mice at doses effective in nicotine-treated mice.
Conclusions
These are the first data suggesting that N-acetylcysteine blocks specific mouse behaviors associated with nicotine reward and withdrawal, which adds to the growing appreciation that N-acetylcysteine may have high clinical utility in combating nicotine dependence.




Similar content being viewed by others
References
Baker DA, McFarland K, Lake RW, Shen H, Tang XC, Toda S, Kalivas PW (2003) Neuroadaptations in cystine-glutamate exchange underlie cocaine relapse. Nat Neurosci 6:743–749. doi:10.1038/nn1069
Barbour B (2001) An evaluation of synapse independence. J Neurosci 21:7969–7984
Bardo MT, Bevins RA (2000) Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology (Berl) 153:31–43
Blokhina EA, Kashkin VA, Zvartau EE, Danysz W, Bespalov AY (2005) Effects of nicotinic and NMDA receptor channel blockers on intravenous cocaine and nicotine self-administration in mice. Eur Neuropsychopharmacol 15:219–225. doi:10.1016/j.euroneuro.2004.07.005
Cohen A, Treweek J, Edwards S, Leao RM, Schulteis G, Koob GF, George O (2015) Extended access to nicotine leads to a CRF1 receptor dependent increase in anxiety-like behavior and hyperalgesia in rats. Addict Biol 20:56–68. doi:10.1111/adb.12077
Damaj MI, Kao W, Martin BR (2003) Characterization of spontaneous and precipitated nicotine withdrawal in the mouse. J Pharmacol Exp Ther 307:526–534. doi:10.1124/jpet.103.054908
De Biasi M, Dani JA (2011) Reward, addiction, withdrawal to nicotine. Annu Rev Neurosci 34:105–130. doi:10.1146/annurev-neuro-061010-113734
Dean O, Giorlando F, Berk M (2011) N-acetylcysteine in psychiatry: current therapeutic evidence and potential mechanisms of action. J Psychiatry Neurosci 36:78–86. doi:10.1503/jpn.100057
Deepmala, Slattery J, Kumar N, Delhey L, Berk M, Dean O, Spielholz C, Frye R (2015) Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev 55:294–321. doi:10.1016/j.neubiorev.2015.04.015
Dringen R, Hirrlinger J (2003) Glutathione pathways in the brain. Biol Chem 384:505–516. doi:10.1515/BC.2003.059
Haydon PG (2001) GLIA: listening and talking to the synapse. Nat Rev Neurosci 2:185–193
Hurst GA, Shaw PB, LeMaistre CA (1967) Laboratory and clinical evaluation of the mucolytic properties of acetylcysteine. Am Rev Respir Dis 96:962–970
Jackson KJ, Kota DH, Martin BR, Damaj MI (2009) The role of various nicotinic receptor subunits and factors influencing nicotine conditioned place aversion. Neuropharmacology 56:970–974. doi:10.1016/j.neuropharm.2009.01.023
Jackson KJ, Martin BR, Changeux JP, Damaj MI (2008) Differential role of nicotinic acetylcholine receptor subunits in physical and affective nicotine withdrawal signs. J Pharmacol Exp Ther 325:302–312. doi:10.1124/jpet.107.132977
Kalivas PW (2009) The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 10:561–572. doi:10.1038/nrn2515
Kenny PJ, Chartoff E, Roberto M, Carlezon WAJ, Markou A (2009) NMDA receptors regulate nicotine-enhanced brain reward function and intravenous nicotine self-administration: role of the ventral tegmental area and central nucleus of the amygdala. Neuropsychopharmacology 34:266–281. doi:10.1038/npp.2008.58
Kenny PJ, Gasparini F, Markou A (2003) Group II metabotropic and alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionate (AMPA)/kainate glutamate receptors regulate the deficit in brain reward function associated with nicotine withdrawal in rats. J Pharmacol Exp Ther 306:1068–1076. doi:10.1124/jpet.103.052027
Knackstedt LA, LaRowe S, Mardikian P, Malcolm R, Upadhyaya H, Hedden S, Markou A, Kalivas PW (2009) The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry 65:841–845. doi:10.1016/j.biopsych.2008.10.040
Kota D, Martin BR, Robinson SE, Damaj MI (2007) Nicotine dependence and reward differ between adolescent and adult male mice. J Pharmacol Exp Ther 322:399–407. doi:10.1124/jpet.107.121616
Lenoir M, Kiyatkin EA (2013) Intravenous nicotine injection induces rapid, experience-dependent sensitization of glutamate release in the ventral tegmental area and nucleus accumbens. J Neurochem 127:541–551. doi:10.1111/jnc.12450
Liechti ME, Lhuillier L, Kaupmann K, Markou A (2007) Metabotropic glutamate 2/3 receptors in the ventral tegmental area and the nucleus accumbens shell are involved in behaviors relating to nicotine dependence. J Neurosci 27:9077–9085. doi:10.1523/JNEUROSCI.1766-07.2007
Liechti ME, Markou A (2007) Interactive effects of the mGlu5 receptor antagonist MPEP and the mGlu2/3 receptor antagonist LY341495 on nicotine self-administration and reward deficits associated with nicotine withdrawal in rats. Eur J Pharmacol 554:164–174. doi:10.1016/j.ejphar.2006.10.011
Liechti ME, Markou A (2008) Role of the glutamatergic system in nicotine dependence: implications for the discovery and development of new pharmacological smoking cessation therapies. CNS Drugs 22:705–724
Lo M, Wang YZ, Gout PW (2008) The x(c)-cystine/glutamate antiporter: a potential target for therapy of cancer and other diseases. J Cell Physiol 215:593–602. doi:10.1002/jcp.21366
Madayag A, Lobner D, Kau KS, Mantsch JR, Abdulhameed O, Hearing M, Grier MD, Baker DA (2007) Repeated N-acetylcysteine administration alters plasticity-dependent effects of cocaine. J Neurosci 27:13968–13976. doi:10.1523/JNEUROSCI.2808-07.2007
McBean GJ (2002) Cerebral cystine uptake: a tale of two transporters. Trends Pharmacol Sci 23:299–302
McClure EA, Gipson CD, Malcolm RJ, Kalivas PW, Gray KM (2014) Potential role of N-acetylcysteine in the management of substance use disorders. CNS Drugs 28:95–106. doi:10.1007/s40263-014-0142-x
Moussawi K, Pacchioni A, Moran M, Olive MF, Gass JT, Lavin A, Kalivas PW (2009) N-Acetylcysteine reverses cocaine-induced metaplasticity. Nat Neurosci 12:182–189. doi:10.1038/nn.2250
Parpura V, Heneka MT, Montana V, Oliet SH, Schousboe A, Haydon PG, Stout RFJ, Spray DC, Reichenbach A, Pannicke T, Pekny M, Pekna M, Zorec R, Verkhratsky A (2012) Glial cells in (patho) physiology. J Neurochem 121:4–27. doi:10.1111/j.1471-4159.2012.07664.x
Paterson NE, Semenova S, Gasparini F, Markou A (2003) The mGluR5 antagonist MPEP decreased nicotine self-administration in rats and mice. Psychopharmacology (Berl) 167:257–264. doi:10.1007/s00213-003-1432-z
Prescott LF, Park J, Ballantyne A, Adriaenssens P, Proudfoot AT (1977) Treatment of paracetamol (acetaminophen) poisoning with N-acetylcysteine. Lancet 2:432–434
Ramirez-Nino AM, D’Souza MS, Markou A (2013) N-acetylcysteine decreased nicotine self-administration and cue-induced reinstatement of nicotine seeking in rats: comparison with the effects of N-acetylcysteine on food responding and food seeking. Psychopharmacology (Berl) 225:473–482. doi:10.1007/s00213-012-2837-3
Reid MS, Fox L, Ho LB, Berger SP (2000) Nicotine stimulation of extracellular glutamate levels in the nucleus accumbens: neuropharmacological characterization. Synapse 35:129–136. doi:10.1002/(SICI)1098-2396(200002)35:2<129::AID-SYN5>3.0.CO;2-D
Samet JM (2013) Tobacco smoking: the leading cause of preventable disease worldwide. Thorac Surg Clin 23:103–112. doi:10.1016/j.thorsurg.2013.01.009
Sanjakdar SS,Maldoon PP,Marks MJ,Brunzell DH,Maskos U,McIntosh JM,Bowers MS, Damaj MI (2014) Differential roles of alpha6beta2* and alpha4beta2* neuronal nicotinic receptors in nicotine- and cocaine-conditioned reward in mice. Neuropsychopharmacology 10.1038/npp.2014.177
Santello M, Cali C, Bezzi P (2012) Gliotransmission and the tripartite synapse. Adv Exp Med Biol 970:307–331. doi:10.1007/978-3-7091-0932-8_14
Sharma S,Hryhorczuk C, Fulton S (2012) Progressive-ratio responding for palatable high-fat and high-sugar food in mice. J Vis Exp e3754. 10.3791/3754
Shiffman S, Brockwell SE, Pillitteri JL, Gitchell JG (2008) Use of smoking-cessation treatments in the United States. Am J Prev Med 34:102–111. doi:10.1016/j.amepre.2007.09.033
Stoker AK, Olivier B, Markou A (2012) Involvement of metabotropic glutamate receptor 5 in brain reward deficits associated with cocaine and nicotine withdrawal and somatic signs of nicotine withdrawal. Psychopharmacology (Berl) 221:317–327. doi:10.1007/s00213-011-2578-8
Stoker AK, Semenova S, Markou A (2008) Affective and somatic aspects of spontaneous and precipitated nicotine withdrawal in C57BL/6J and BALB/cByJ mice. Neuropharmacology 54:1223–1232. doi:10.1016/j.neuropharm.2008.03.013
Timmerman W, Westerink BH (1997) Brain microdialysis of GABA and glutamate: what does it signify? Synapse 27:242–261. doi:10.1002/(SICI)1098-2396(199711)27:3<242::AID-SYN9>3.0.CO;2-D
Turner JR, Ray R, Lee B, Everett L, Xiang J, Jepson C, Kaestner KH, Lerman C, Blendy JA (2014) Evidence from mouse and man for a role of neuregulin 3 in nicotine dependence. Mol Psychiatry 19:801–810. doi:10.1038/mp.2013.104
Wang F, Chen H, Sharp BM (2008) Neuroadaptive changes in the mesocortical glutamatergic system during chronic nicotine self-administration and after extinction in rats. J Neurochem 106:943–956. doi:10.1111/j.1471-4159.2008.05456.x
Wang F, Chen H, Steketee JD, Sharp BM (2007) Upregulation of ionotropic glutamate receptor subunits within specific mesocorticolimbic regions during chronic nicotine self-administration. Neuropsychopharmacology 32:103–109. doi:10.1038/sj.npp.1301033
Warr O, Takahashi M, Attwell D (1999) Modulation of extracellular glutamate concentration in rat brain slices by cystine-glutamate exchange. J Physiol 514:783–793
Watkins SS, Koob GF, Markou A (2000) Neural mechanisms underlying nicotine addiction: acute positive reinforcement and withdrawal. Nicotine Tob Res 2:19–37
Xi ZX, Baker DA, Shen H, Carson DS, Kalivas PW (2002) Group II metabotropic glutamate receptors modulate extracellular glutamate in the nucleus accumbens. J Pharmacol Exp Ther 300:162–171
Zhao-Shea R, DeGroot SR, Liu L, Vallaster M, Pang X, Su Q, Gao G, Rando OJ, Martin GE, George O, Gardner PD, Tapper AR (2015) Increased CRF signalling in a ventral tegmental area-interpeduncular nucleus-medial habenula circuit induces anxiety during nicotine withdrawal. Nat Commun 6:6770. doi:10.1038/ncomms7770
Zhou W, Kalivas PW (2008) N-acetylcysteine reduces extinction responding and induces enduring reductions in cue- and heroin-induced drug-seeking. Biol Psychiatry 63:338–340. doi:10.1016/j.biopsych.2007.06.008
Acknowledgments
MSB is supported by the Alcohol Beverage Medical Research Foundation, a Center for Translational Research Award (UL1 TR000058), the National Institutes for Alcohol Abuse and Alcoholism (P50 AA022537), and ABMRF/The Foundation for Alcohol Research. MID is supported by the National Institutes for Drug Abuse (R01 DA032246 and R21 DA032246).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Experiments were performed during the light cycle and were approved by the Institutional Animal Care and Use Committee of Virginia Commonwealth University and followed the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Disclosures
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Bowers, M.S., Jackson, A., Maldoon, P.P. et al. N-acetylcysteine decreased nicotine reward-like properties and withdrawal in mice. Psychopharmacology 233, 995–1003 (2016). https://doi.org/10.1007/s00213-015-4179-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4179-4