Abstract
Rationale
The selective N-methyl-d-aspartate (NMDA) channel blocker MK-801 is known to induce no loss of the righting reflex (LORR) and to stimulate catecholaminergic (CAergic) neurons in rodents, playing a crucial role in arousal.
Objectives
We examined whether MK-801 in combination with CA receptor ligands, which inhibit CAergic neuronal activities, could induce anesthesia including LORR.
Methods
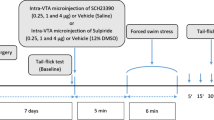
All drugs were administered systemically to mice. To assess anesthesia, three different behaviors were used: loss of nociceptive response (analgesia in the free-moving state without LORR), LORR, and loss of movement in response to noxious stimulation (immobility under LORR).
Results
A very large dose of MK-801 (50 mg/kg) induced neither analgesia nor LORR. In contrast, MK-801 in combination with a small dose of the dopamine (DA) receptor antagonist haloperidol (0.2 mg/kg) dose-dependently produced LORR with a 50 % effective dose (ED50) of 1.6 (0.9–3.0; 95 % confidence limit) mg/kg, but not immobility. The α2-adrenoceptor agonist dexmedetomidine induced not only analgesia, but also immobility in animals treated with MK-801 (5 mg/kg) plus haloperidol (0.2 mg/kg), which then lost their righting reflex. The ED50 value of 0.26 (0.10–0.66) mg/kg (various doses of dexmedetomidine plus a fixed dose of MK-801 and haloperidol) for immobility was approximately three-fold larger than that of 0.09 (0.03–0.23) mg/kg (dexmedetomidine plus vehicle saline) for analgesia. This may occur, as LORR induced by MK-801 plus haloperidol inhibits the pain suppression system. The other ligands had little or no effect.
Conclusions
The DAergic stimulant actions of MK-801 may mask its LORR effects by NMDA channel blockade.




Similar content being viewed by others
References
Aimone LD, Yaksh TL (1989) Opioid modulation of capsaicin-evoked release of substance P from rat spinal cord in vivo. Peptides 10:1127–1131
Bianchi C, Franceschini J (1954) Experimental observations on Haffner ’s method for testing analgesic drugs. Br J Pharmacol 9:280–284
Carter AJ (1995) Antagonists of the NMDA receptor-channel complex and motor coordination. Life Sci 57:917–929
Chen X, Shu S, Bayliss DA (2009) HCN1 channel subunits are a molecular substrate for hypnotic actions of ketamine. J Neurosci 29:600–609
Clarke PB, Reuben M (1995) Inhibition by dizocilpine (MK-801) of striatal dopamine release induced by MPTP and MPP+: possible action at the dopamine transporter. Br J Pharmacol 114:315–322
Cooper JR, Bloom FE, Roth RH (2003) Dopamine. In: Cooper JR, Bloom FE, Roth RH (eds) The biochemical basis of neuropharmacology, 8th edn. Oxford University Press, New York, pp 225–270
Daniell LC (1990) The noncompetitive N-methyl-D-aspartate antagonists, MK-801, phencyclidine and ketamine, increase the potency of general anesthetics. Pharmacol Biochem Behav 36:111–115
Del Arco A, Segovia G, Mora F (2008) Blockade of NMDA receptors in the prefrontal cortex increases dopamine and acetylcholine release in the nucleus accumbens and motor activity. Psychopharmacology 201:325–338
Doze VA, Chen BX, Maze M (1989) Dexmedetomidine produces a hypnotic-anesthetic action in rats via activation of central alpha-2 adrenoceptors. Anesthesiology 71:75–79
Dutton RC, Zhang Y, Stabernack CR, Laster MJ, Sonner JM, Eger EI II (2003) Temporal summation governs part of the minimum alveolar concentration of isoflurane anesthesia. Anesthesiology 98:1372–1377
Fields HL, Heinricher MM, Mason P (1991) Neurotransmitters in nociceptive modulatory circuits. Annu Rev Neurosci 14:219–245
Fishman RHB, Feigenbaum JJ, Yanai J, Klawans HL (1983) The relative importance of dopamine and norepinephrine in mediating locomotor activity. Prog Neurobiol 20:55–88
Foster AC (1991) Channel blocking drugs for the NMDA receptor. In: Meldrum BS (ed) Excitatory amino acid antagonists. Blackwell Scientific Publications, Oxford, pp 164–179
Franks NP (2008) General anaesthesia: from molecular targets to neuronal pathways of sleep and arousal. Nat Rev Neurosci 9:370–386
Gattaz WF, Schummer B, Behrens S (1994) Effects of zotepine, haloperidol and clozapine on MK-801-induced stereotypy and locomotion in rats. J Neural Transm Gen Sect 96:227–232
Gilsbach R, Röser C, Beetz N, Brede M, Hadamek K, Haubold M, Leemhuis J, Philipp M, Schneider J, Urbanski M, Szabo B, Weinshenker D, Hein L (2009) Genetic dissection of α2-adrenoceptor functions in adrenergic versus nonadrenergic cells. Mol Pharmacol 75:1160–1170
Guo TZ, Jiang JY, Buttermann AE, Maze M (1996) Dexmedetomidine injection into the locus ceruleus produces antinociception. Anesthesiology 84:873–881
Guyton AC, Hall JE (2006) Somatic sensations: II. Pain, headache, and thermal sensations. In: Guyton AC, Hall JE (eds) Textbook of medical physiology, 11th edn. Elsevier Saunders, Philadelphia, pp 598–609
Hu FY, Hanna GM, Han W, Mardini F, Thomas SA, Wyner AJ, Kelz MB (2012) Hypnotic hypersensitivity to volatile anesthetics and dexmedetomidine in dopamine β-hydroxylase knockout mice. Anesthesiology 117:1006–1017
Hynes MD, Berkowitz BA (1983) Catecholamine mechanisms in the stimulation of mouse locomotor activity by nitrous oxide and morphine. Eur J Pharmacol 90:109–114
Imperato A, Scrocco MG, Bacchi S, Angelucci L (1990) NMDA receptors and in vivo dopamine release in the nucleus accumbens and caudatus. Eur J Pharmacol 187:555–556
Iravani MM, Muscat R, Kruk ZL (1999) MK-801 interaction with the 5-HT transporter: a real-time study in brain slices using fast cyclic voltammetry. Synapse 32:212–224
Irifune M, Shimizu T, Nomoto M (1991) Ketamine-induced hyperlocomotion associated with alteration of presynaptic components of dopamine neurons in the nucleus accumbens of mice. Pharmacol Biochem Behav 40:399–407
Irifune M, Shimizu T, Nomoto M, Fukuda T (1995) Involvement of N-methyl-D-aspartate (NMDA) receptors in noncompetitive NMDA receptor antagonist-induced hyperlocomotion in mice. Pharmacol Biochem Behav 51:291–296
Irifune M, Katayama S, Takarada T, Shimizu Y, Endo C, Takata T, Morita K, Dohi T, Sato T, Kawahara M (2007) MK-801 enhances gabaculine-induced loss of the righting reflex in mice, but not immobility. Can J Anaesth 54:998–1005
Iwata S, Izumi K, Shimizu T, Fukuda T (1989) Effects of repeated testing on the incidence of haloperidol-induced catalepsy in mice. Pharmacol Biochem Behav 33:705–707
Jackson DM, Johansson C, Lindgren LM, Bengtsson A (1994) Dopamine receptor antagonists block amphetamine and phencyclidine-induced motor stimulation in rats. Pharmacol Biochem Behav 48:465–471
Katayama S, Irifune M, Kikuchi N, Takarada T, Shimizu Y, Endo C, Takata T, Dohi T, Sato T, Kawahara M (2007) Increased γ-aminobutyric acid levels in mouse brain induce loss of righting reflex, but not immobility, in response to noxious stimulation. Anesth Analg 104:1422–1429
Koblin DD (2000) Mechanisms of action. In: Miller RD (ed) Anesthesia, 5th edn. Churchill Livingstone, Philadelphia, pp 48–73
Kovacic P, Somanathan R (2010) Clinical physiology and mechanism of dizocilpine (MK-801): electron transfer, radicals, redox metabolites and bioactivity. Oxidative Med Cell Longev 3:13–22
Lapin IP, Rogawski MA (1995) Effects of D1 and D2 dopamine receptor antagonists and catecholamine depleting agents on the locomotor stimulation induced by dizocilpine in mice. Behav Brain Res 70:145–151
Masuzawa M, Nakao S, Miyamoto E, Yamada M, Murao K, Nishi K, Shingu K (2003) Pentobarbital inhibits ketamine-induced dopamine release in the rat nucleus accumbens: a microdialysis study. Anesth Analg 96:148–152
Mathé JM, Nomikos GG, Hildebrand BE, Hertel P, Svensson TH (1996) Prazosin inhibits MK-801-induced hyperlocomotion and dopamine release in the nucleus accumbens. Eur J Pharmacol 309:1–11
Mathé JM, Nomikos GG, Blakeman KH, Svensson TH (1999) Differential actions of dizocilpine (MK-801) on the mesolimbic and mesocortical dopamine systems: role of neuronal activity. Neuropharmacology 38:121–128
O'Brien CP (2011) Drug addiction. In: Brunton LL, Chabner BA, Knollmann BC (eds) Goodman & Gilman’s the pharmacological basis of therapeutics, 12th edn. McGraw-Hill, New York, pp 649–668
Patel PM, Patel HH, Roth DM (2011) General anesthetics and therapeutic gases. In: Brunton LL, Chabner BA, Knollmann BC (eds) Goodman & Gilman’s the pharmacological basis of therapeutics, 12th edn. McGraw-Hill, New York, pp 527–564
Ramoa AS, Alkondon M, Aracava Y, Irons J, Lunt GG, Deshpande SS, Wonnacott S, Aronstam RS, Albuquerque EX (1990) The anticonvulsant MK-801 interacts with peripheral and central nicotinic acetylcholine receptor ion channels. J Pharmacol Exp Ther 254:71–82
Sakamoto S, Nakao S, Masuzawa M, Inada T, Maze M, Franks NP, Shingu K (2006) The differential effects of nitrous oxide and xenon on extracellular dopamine levels in the rat nucleus accumbens: a microdialysis study. Anesth Analg 103:1459–1463
Sinner B, Graf BM (2008) Ketamine. In: Schüttler J, Schwilden H (eds) Modern anesthetics. Springer, Berlin, pp 313–333
Spyraki C, Fibiger HC (1982) Clonidine-induced sedation in rats: evidence for mediation by postsynaptic α2-adrenoceptors. J Neural Transm 54:153–163
Takano M, Takano Y, Yaksh TL (1993) Release of calcitonin gene-related peptide (CGRP), substance P (SP), and vasoactive intestinal polypeptide (VIP) from rat spinal cord: modulation by α2 agonists. Peptides 14:371–378
Taylor NE, Chemali JJ, Brown EN, Solt K (2013) Activation of D1 dopamine receptors induces emergence from isoflurane general anesthesia. Anesthesiology 118:30–39
Westfall TC, Westfall DP (2011) Adrenergic agonists and antagonists. In: Brunton LL, Chabner BA, Knollmann BC (eds) Goodman & Gilman’s the pharmacological basis of therapeutics, 12th edn. McGraw-Hill, New York, pp 277–333
Woods JH, Koek W, France CP, Moerschbaecher JM (1991) Behavioural effects of NMDA antagonists. In: Meldrum BS (ed) Excitatory amino acid antagonists. Blackwell Scientific Publications, Oxford, pp 237–264
Yan Q-S, Reith MEA, Jobe PC, Dailey JW (1997) Dizocilpine (MK-801) increases not only dopamine but also serotonin and norepinephrine transmissions in the nucleus accumbens as measured by microdialysis in freely moving rats. Brain Res 765:149–158
Acknowledgments
This study was supported in part by Grant-in-Aid No. 18592037 for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology of Japan. We thank Medical English Service Corp. (Kyoto, Japan) for English language editing.
Conflict of interest
Authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kikuchi, N., Irifune, M., Shimizu, Y. et al. Selective blockade of N-methyl-d-aspartate channels in combination with dopamine receptor antagonism induces loss of the righting reflex in mice, but not immobility. Psychopharmacology 232, 39–46 (2015). https://doi.org/10.1007/s00213-014-3634-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3634-y