Abstract
Perturbations produced by ionizing radiation on intestinal tissue are considered one of highly drastic challenges in radiotherapy. Animals were randomized into five groups. The first group was allocated as control, and the second was subjected to whole body γ-irradiation (10 Gy). The third was administered HA NP (17.6 mg/kg/day; i.p.) and then irradiated. The fourth one received MitoQ (2 mg/kg/day; i.p.) and then irradiated. The last group received MitoQ/HA NP (2 mg/kg/day; i.p.) for 5 days prior to irradiation. Mice were sacrificed a week post-γ-irradiation for evaluation. MitoQ/HA NP ameliorated mitochondrial oxidative stress as indicated by rising (TAC) and glutathione peroxidase and decreasing malondialdehyde, showing its distinguished antioxidant yield. That impacted the attenuation of apoptosis, which was revealed by the restoration of the anti-apoptotic marker and lessening proapoptotic caspase-3. Inflammatory parameters dwindled via treatment with MitoQ/HA NP. Moreover, this new NP exerts its therapeutic action through a distinguished radioprotective pathway (Hmgb1/TLR-4.) Subsequently, these antioxidants and their nanoparticles conferred protection to intestinal tissue as manifested by histopathological examination. These findings would be associated with its eminent antioxidant potential through high mitochondria targeting, enhanced cellular uptake, and ROS scavenging. This research underlines MitoQ/HA NP as a new treatment for the modulation of intestinal damage caused by radiotherapy modalities.
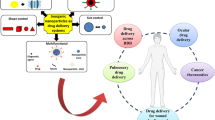
Graphical Abstract
This illustrates that gamma radiation–induced intestinal injury in mice is indicated by an increase in mitochondrial oxidative stress, reactive oxygen species, and intestinal inflammation. The apoptosis marker was increased after irradiation that leads to intestinal damage through the TLR-4 and Hmgb-1 pathways. MitoQ and its hyaluronic acid–based nanopreparation improve all previously mentioned parameters.














Similar content being viewed by others
Data availability
Not applicable.
References
Bavelaar BM, Lee BQ, Gill MR, Falzone N, Vallis KA (2018) Subcellular targeting of theranostic radionuclides. Front Pharmacol 9:996
Botting K, Skeffington K, Niu Y, Allison B, Brain K, Itani N, Beck C, Logan A, Murray A, Murphy M (2020) Translatable mitochondria-targeted protection against programmed cardiovascular dysfunction. Sci Adv 6:eabb1929
Brentnall M, Rodriguez-Menocal L, De Guevara RL, Cepero E, Boise LH (2013) Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol 14:32
Cao X, Wen P, Fu Y, Gao Y, Qi X, Chen B, Tao Y, Wu L, Xu A, Lu H (2019) Radiation induces apoptosis primarily through the intrinsic pathway in mammalian cells. Cell Signal 62:109337
Chaurasia M, Bhatt AN, Das A, Dwarakanath BS, Sharma K (2016) Radiation-induced autophagy: mechanisms and consequences. Free Radical Res 50:273–290
Cosentino D, Piro F (2018) Hyaluronic acid for treatment of the radiation therapy side effects: a systematic review. Eur Rev Med Pharmacol Sci 22:7562–7572
Cox B, Emili A (2006) Tissue subcellular fractionation and protein extraction for use in mass-spectrometry-based proteomics. Nat Protoc 1:1872–1878
Datta K, Suman S, Fornace AJ (2014) Radiation persistently promoted oxidative stress, activated mTOR via PI3K/Akt, and downregulated autophagy pathway in mouse intestine. Int J Biochem Cell Biol 57:167–176
de la Cruz Bonilla B, Stemler KM, Taniguchi CM, Piwnica-Worms H (2018) Stem cell enriched-epithelial spheroid cultures for rapidly assaying small intestinal radioprotectors and radiosensitizers in vitro. Sci Rep 8:1–8
Du J, Zhang P, Cheng Y, Liu R, Liu H, Gao F, Shi C, Liu C (2020) General principles of developing novel radioprotective agents for nuclear emergency. Radiat Med Protect 1:120–126
Dua R, Gill KD (2004) Effect of aluminium phosphide exposure on kinetic properties of cytochrome oxidase and mitochondrial energy metabolism in rat brain. Biochim Biophys Acta (BBA)-Gen Subj 1674:4–11
Duman DG, Kumral ZNÖ, Ercan F, Deniz M, Can G, Yeğen BÇ (2013) Saccharomyces boulardii ameliorates clarithromycin-and methotrexate-induced intestinal and hepatic injury in rats. Br J Nutr 110:493–499
Farhood B, Goradel NH, Mortezaee K, Khanlarkhani N, Salehi E, Nashtaei MS, Mirtavoos-mahyari H, Motevaseli E, Shabeeb D, Musa AE, Najafi M (2019) Melatonin as an adjuvant in radiotherapy for radioprotection and radiosensitization. Clin Transl Oncol 21:268–279
Flecknell PA (1993) Anaesthesia of animals for biomedical research. Br J Anaesth 71:885–894
Gheita HA, El-Sabbagh WA, Abdelsalam RM, Attia AS, El-Ghazaly MA (2019) Promising role of filgrastim and α-tocopherol succinate in amelioration of gastrointestinal acute radiation syndrome (GI-ARS) in mice. Naunyn Schmiedebergs Arch Pharmacol 392:1537–1550
Han C, Guan Q, Guo L, Yang Y, Ruan S, Zhang X (2019) The effects and mechanisms of myeloid differentiation protein 2 on intestinal mucosal permeability in mice with chronic colitis. J Cell Physiol 234:21089–21099
Harb AH, Abou Fadel C, Sharara AI (2014) Radiation enteritis. Curr Gastroenterol Rep 16:1–9
Ibrahim AA, Karam HM, Shaaban EA, Safar MM, El-Yamany MF (2019) MitoQ ameliorates testicular damage induced by gamma irradiation in rats: Modulation of mitochondrial apoptosis and steroidogenesis. Life Sci 232:116655
Jd B, Gamble M (2013) Theory and practice of histological techniques. 2013, Churchill Livingstone. Elsevier, Oxford
Kam WW, Banati RB (2013) Effects of ionizing radiation on mitochondria. Free Radic Biol Med 65:607–619
Karam HM, Radwan RR (2019) Metformin modulates cardiac endothelial dysfunction, oxidative stress and inflammation in irradiated rats: a new perspective of an antidiabetic drug. Clin Exp Pharmacol Physiol 46:1124–1132
Kelso GF, Porteous CM, Coulter CV, Hughes G, Porteous WK, Ledgerwood EC, Smith RA, Murphy MP (2001) Selective targeting of a redox-active ubiquinone to mitochondria within cells antioxidant and antiapoptotic properties. J Biol Chem 276:4588–4596
Kiang JG, Olabisi AO (2019) Radiation: a poly-traumatic hit leading to multi-organ injury. Cell Biosci 9:1–15
Koracevic D, Koracevic G, Djordjevic V, Andrejevic S, Cosic V (2001) Method for the measurement of antioxidant activity in human fluids. J Clin Pathol 54:356–361
Kowalczyk P, Sulejczak D, Kleczkowska P, Bukowska-Ośko I, Kucia M, Popiel M, Wietrak E, Kramkowski K, Wrzosek K, Kaczyńska K (2021) Mitochondrial oxidative stress—a causative factor and therapeutic target in many diseases. Int J Mol Sci 22:13384
Kulkarni R, Marples B, Balasubramaniam M, Thomas RA, Tucker JD (2010) Mitochondrial gene expression changes in normal and mitochondrial mutant cells after exposure to ionizing radiation. Radiat Res 173:635–644
Lierova A, Kasparova J, Pejchal J, Kubelkova K, Jelicova M, Palarcik J, Korecka L, Bilkova Z, Sinkorova Z (2020) Attenuation of radiation-induced lung injury by hyaluronic acid nanoparticles. Front Pharmacol 11:1199
Luo H, Guo P, Zhou Q (2012) Role of TLR4/NF-κB in damage to intestinal mucosa barrier function and bacterial translocation in rats exposed to hypoxia
McManus MJ, Murphy MP, Franklin JL (2011) The mitochondria-targeted antioxidant MitoQ prevents loss of spatial memory retention and early neuropathology in a transgenic mouse model of Alzheimer’s disease. J Neurosci 31:15703–15715
Mihara M, Uchiyama M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278
Murphy MP, Smith RA (2007) Targeting antioxidants to mitochondria by conjugation to lipophilic cations. Annu Rev Pharmacol Toxicol 47:629–656
Oyewole AO, Birch-Machin MA (2015) Mitochondria-targeted antioxidants. FASEB J 29:4766–4771
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Radwan RR, Karam HM (2020) Resveratrol attenuates intestinal injury in irradiated rats via PI3K/Akt/mTOR signaling pathway. Environ Toxicol 35:223–230
Riehl TE, Foster L, Stenson WF (2012) Hyaluronic acid is radioprotective in the intestine through a TLR4 and COX-2-mediated mechanism. Am J Physiol Gastrointest Liver Physiol 302:G309-316
Schoenberg MD, Brooks RE, Hall JJ, Schneiderman H (1951) Effect of x-irradiation on the hyaluronïdasehyaluronic acid system. Arch Biochem 30:333–340
Shinn LJ, Lagalwar S (2021) Treating neurodegenerative disease with antioxidants: efficacy of the bioactive phenol resveratrol and mitochondrial-targeted MitoQ and SkQ. Antioxidants 10:573
Terra WR, Ferreira C (2005) 4.5 - Biochemistry of Digestion. In: Gilbert LI (ed) Comprehensive molecular insect science. Elsevier, Amsterdam, pp 171–224
Vajjhala PR, Ve T, Bentham A, Stacey KJ, Kobe B (2017) The molecular mechanisms of signaling by cooperative assembly formation in innate immunity pathways. Mol Immunol 86:23–37
Valenti D, Vacca RA, de Pinto MC, De Gara L, Marra E, Passarella S (2007) In the early phase of programmed cell death in Tobacco Bright Yellow 2 cells the mitochondrial adenine nucleotide translocator, adenylate kinase and nucleoside diphosphate kinase are impaired in a reactive oxygen species-dependent manner. Biochim Biophys Acta (BBA) Bioenerg 1767:66–78
Vergeade A, Mulder P, Vendeville-Dehaudt C, Estour F, Fortin D, Ventura-Clapier R, Thuillez C, Monteil C (2010) Mitochondrial impairment contributes to cocaine-induced cardiac dysfunction: prevention by the targeted antioxidant MitoQ. Free Radical Biol Med 49:748–756
Wang C, Xie J, Dong X, Mei L, Zhao M, Leng Z, Hu H, Li L, Gu Z, Zhao Y (2020) Clinically approved carbon nanoparticles with oral administration for intestinal radioprotection via protecting the small intestinal crypt stem cells and maintaining the balance of intestinal flora. Small 16:1906915
Zhang C, Chen K, Wang J, Zheng Z, Luo Y, Zhou W, Zhuo Z, Liang J, Sha W, Chen H (2020a) Protective effects of crocetin against radiation-induced injury in intestinal epithelial cells. BioMed Res Int 2020
Zhang X-m, Hu X, Ou J-y, Chen S-s, Nie L-h, Gao L, Zhu L-l (2020b) Glycyrrhizin ameliorates radiation enteritis in mice accompanied by the regulation of the HMGB1/TLR4 pathway. Evid-Based Complement Alternat Med 2020:8653783
Zhen J, Dai Y, Villani T, Giurleo D, Simon JE, Wu Q (2017) Synthesis of novel flavonoid alkaloids as α-glucosidase inhibitors. Bioorg Med Chem 25:5355–5364
Zheng Q, Li L, Liu M, Huang B, Zhang N, Mehmood R, Nan K, Li Q, Chen W, Lin S (2020) In situ scavenging of mitochondrial ROS by anti-oxidative MitoQ/hyaluronic acid nanoparticles for environment-induced dry eye disease therapy. Chem Eng J 398:125621
Acknowledgements
We thank Prof. Ahmed Osman, Professor of Pathology at the Faculty of Veterinary Medicine, Cairo University, Egypt, for carrying out the histological examination.
Funding
The authors would like to thank the Deanship of Scientific Research at Umm Al-Qura University, Makkah, Saudi Arabia, for supporting this work by grant code 19-MED-1–01-0038.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Heba M. Karam and Ayman A. Ibrahim. The first draft of the manuscript was written by Ayman A. Ibrahim. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All animals’ procedures were approved by the Animal Care Committee of the National Centre for Radiation Research and Technology (NCRRT), Cairo, Egypt, which complies with the Guide for Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85–23, revised 2011).
Rats were obtained from the animal house belonging to the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt. Mice were maintained in colony cages at 25˚C ± 5˚C, a humidity of 60 ± 5% and housed in suitable illumination conditions (12 h/ 12 h light/dark cycle) and were fed standard pellet diet and fresh water ad libitum. Animals were left one week for acclimatization in the lab environment before starting the experiment. Animals were treated gently; pressure, squeezing, and tough maneuvers were avoided.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Study highlights
• MitoQ is a potent antioxidant and ameliorates γ-radiation-induced intestinal injury.
• MitoQ/HA NP mitigated intestinal damage produced by γ-irradiation.
• MitoQ/HA NP reduced radiation-induced intestinal oxidative stress and apoptosis.
• MitoQ/HA NP attenuated radiation-induced inflammation in the intestine and Hmgb1/TLR4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dawoud, M., Attallah, K.M., Ibrahim, I.T. et al. MitoQ and its hyaluronic acid–based nanopreparation mitigating gamma radiation–induced intestinal injury in mice: alleviation of oxidative stress and apoptosis. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-02948-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-02948-5