Abstract
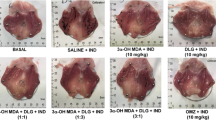
Therapeutic effect of non-steroidal anti-inflammatory drugs (NSAIDs) has been related with gastrointestinal injury. Docosahexaenoic acid (DHA), an omega-3 polyunsaturated fatty acid (PUFA), can prevent gastric and small intestinal damage. Nonetheless, contribution of antioxidative action in the protective effect of DHA has not been evaluated before in the small intestine injury after indomethacin treatment. Pathogenesis of NSAID-induced small intestinal injury is multifactorial, and reactive oxidative species have been related to indomethacin’s small intestinal damage. The present work aimed to evaluate antioxidative activity in the protective action of DHA in the indomethacin-induced small intestinal damage. Female Wistar rats were gavage with DHA (3 mg/kg) or omeprazole (3 mg/kg) for 10 days. Each rat received indomethacin (3 mg/kg, orally) daily to induce small intestinal damage. The total area of intestinal ulcers and histopathological analysis were performed. In DHA-treated rats, myeloperoxidase and superoxide dismutase activity, glutathione, malondialdehyde, leukotriene, and lipopolysaccharide (LPS) levels were measured. Furthermore, the relative abundance of selective bacteria was assessed. DHA administration (3 mg/kg, p.o.) caused a significant decrease in indomethacin-induced small intestinal injury in Wistar rats after 10 days of treatment. DHA’s enteroprotection resulted from the prevention of an increase in myeloperoxidase activity, and lipoperoxidation, as well as an improvement in the antioxidant defenses, such as glutathione levels and superoxide dismutase activity in the small intestine. Furthermore, we showed that DHA’s enteroprotective effect decreased significantly LPS levels in indomethacin-induced injury in small intestine. Our data suggest that DHA’s enteroprotective might be attributed to the prevention of oxidative stress.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aratani Y (2018) Myeloperoxidase: its role for host defense, inflammation, and neutrophil function. Arch Biochem Biophys 640:47–52. https://doi.org/10.1016/j.abb.2018.01.004
Arroyo-Lira AG, Rodríguez-Ramos F, Chávez-Piña AE (2014) Synergistic antinociceptive effect and gastric safety of the combination of docosahexaenoic acid and indomethacin in rats. Pharmacol Biochem Behav 122:74–81. https://doi.org/10.1016/j.pbb.2014.03.015
Arroyo-Lira AG, Rodríguez-Ramos F, Ortiz MI, Castañeda-Hernández G, Chávez-Piña AE (2017) Supra-additive interaction of docosahexaenoic acid and naproxen and gastric safety on the formalin test in rats. Drug Dev Res 78:332–339. https://doi.org/10.1002/DDR.21396
Bindu S, Mazumder S, Bandyopadhyay U (2020) Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: a current perspective. Biochem Pharmacol 180:114147. https://doi.org/10.1016/j.bcp.2020.114147
Cao W, Wang C, Chin Y, Chen X, Gao Y, Yuan S, Xue C, Wang Y, Tang Q (2019) DHA-phospholipids (DHA-PL) and EPA-phospholipids (EPA-PL) prevent intestinal dysfunction induced by chronic stress. Food Funct 10:277–288. https://doi.org/10.1039/C8FO01404C
Che H, Li H, Song L, Dong X, Yang X, Zhang T, Wang Y, Xie W (2021) Orally administered DHA-enriched phospholipids and DHA-enriched triglyceride relieve oxidative stress, improve intestinal barrier, modulate inflammatory cytokine and gut microbiota, and meliorate inflammatory responses in the brain in dextran sodium sulfate induced colitis in mice. Mol Nutr Food Res 65:2000986. https://doi.org/10.1002/mnfr.202000986
Cuervo-Zanatta D, Garcia-Mena J, Perez-Cruz C (2021) Gut microbiota alterations and cognitive impairment are sexually dissociated in a transgenic mice model of Alzheimer’s disease. J Alzheimers Dis 82(s1):S195–S214. https://doi.org/10.3233/JAD-201367
Djuricic I, Calder PC (2021) Beneficial outcomes of omega-6 and omega-3 polyunsaturated fatty acids on human health: an update for 2021. Nutrients 13:2421. https://doi.org/10.3390/nu13072421
Galicia-Moreno M, Rosique-Oramas D, Medina-Avila Z, Álvarez-Torres T, Falcón D, Higuera-de la tijera F, Béjar YL, Cordero-Pérez P, Muñoz-Espinosa L, Pérez-Hernández JL, Kershenobich D, Gutierrez-Reyes G (2016) Behavior of oxidative stress markers in alcoholic liver cirrhosis patients. Oxid Med Cell Longev 2016:1–10. https://doi.org/10.1155/2016/9370565
Handa O, Majima A, Onozawa Y, Horie H, Uehara Y, Fukui A, Omatsu T, Naito Y, Yoshikawa T (2014) The role of mitochondria-derived reactive oxygen species in the pathogenesis of non-steroidal anti-inflammatory drug-induced small intestinal injury. Free Radic Res 48:1095–1099. https://doi.org/10.3109/10715762.2014.928411
Kuzumoto T, Tanigawa T, Higashimori A, Kitamura H, Nadatani Y, Otani K, Fukunaga S, Hosomi S, Tanaka F, Kamata N, Nagami Y, Taira K, Watanabe T, Fujiwara Y (2021) Protective role of resolvin D1, a pro-resolving lipid mediator, in nonsteroidal anti-inflammatory drug-induced small intestinal damage. PLoS One. 16(5):e0250862. https://doi.org/10.1371/journal.pone.0250862
Lopez-Siles M, Enrich-Capó N, Aldeguer X, Sabat-Mir M, Duncan SH, Garcia-Gil LJ, Martinez-Medina M (2018) Alterations in the abundance and co-occurrence of Akkermansia muciniphila and Faecalibacterium prausnitzii in the colonic mucosa of inflammatory bowel disease subjects. Front Cell Infect Microbiol. 8:281. https://doi.org/10.3389/fcimb.2018.00281
Maseda D, Ricciotti E (2020) NSAID–gut microbiota interactions. Front Pharmacol 11. https://doi.org/10.3389/fphar.2020.01153
Maseda D, Zackular JP, Trindade B, Kirk L, Roxas JL, Rogers LM, Washington MK, Du L, Koyama T, Viswanathan VK, Vedantam G, Schloss PD, Crofford LJ, Skaar EP, Aronoff DM (2019) Nonsteroidal anti-inflammatory drugs alter the microbiota and exacerbate Clostridium difficile colitis while dysregulating the inflammatory response. mBio 10. https://doi.org/10.1128/mBio.02282-18
Miranda-Lara CA, Ortiz MI, Rodríguez-Ramos F, Chávez-Piña AE (2018) Synergistic interaction between docosahexaenoic acid and diclofenac on inflammation, nociception, and gastric security models in rats. Drug Dev Res 79:239–246. https://doi.org/10.1002/ddr.21438
Pineda-Peña EA, Jiménez-Andrade JM, Castañeda-Hernández G, Chávez-Piña AE (2012) Docosahexaenoic acid, an omega-3 polyunsaturated acid protects against indomethacin-induced gastric injury. Eur J Pharmacol 697:139–143. https://doi.org/10.1016/j.ejphar.2012.09.049
Pineda-Peña EA, Martínez-Pérez Y, Galicia-Moreno M, Navarrete A, Segovia J, Muriel P, Favari L, Castañeda-Hernández G, Chávez-Piña AE (2018) Participation of the anti-inflammatory and antioxidative activity of docosahexaenoic acid on indomethacin-induced gastric injury model. Eur J Pharmacol 818:585–592. https://doi.org/10.1016/j.ejphar.2017.11.015
Qiu S, Li P, Zhao H, Li X (2020) Maresin 1 alleviates dextran sulfate sodium-induced ulcerative colitis by regulating NRF2 and TLR4/NF-kB signaling pathway. Int Immunopharmacol 78:106018. https://doi.org/10.1016/j.intimp.2019.106018
Saleh A, Noguchi Y, Aramayo R, Ivanova ME, Stevens KM, Montoya A, Sunidhi S, Carranza NL, Skwark MJ, Speck C (2022) The structural basis of Cdc7-Dbf4 kinase dependent targeting and phosphorylation of the MCM2-7 double hexamer. Nat Commun 13:2915. https://doi.org/10.1038/s41467-022-30576-1
Salinas-Nolasco C, Barragán-Zarate GS, Lagunez-Rivera L, Solano R, Favari L, Jiménez-Andrade JM, Chávez-Piña AE (2023) Role of LTB4 and nitric oxide in the gastroprotective effect of Prosthechea karwinskii leaves extract in the indomethacin-induced gastric injury in the rat. Nat Prod Res. 37(5):819–822. https://doi.org/10.1080/14786419.2022.2089880
Sánchez-Tapia M, Miller AW, Granados-Portillo O, Tovar AR, Torres N (2020) The development of metabolic endotoxemia is dependent on the type of sweetener and the presence of saturated fat in the diet. Gut Microbes 12:1801301. https://doi.org/10.1080/19490976.2020.1801301
Sánchez-Trigueros MI, Méndez-Cruz F, Pineda-Peña EA, Rivera-Espinoza Y, Castañeda-Hernández G, Chávez-Piña AE (2021) Synergistic protective effects between docosahexaenoic acid and omeprazole on the gastrointestinal tract in the indomethacin-induced injury model. Drug Dev Res 82:543–552. https://doi.org/10.1002/DDR.21772
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc. 3(6):1101–8. https://doi.org/10.1038/nprot.2008.73
Sharma M, Kaur R, Kaushik K, Kaushal N (2019) Redox modulatory protective effects of ω-3 fatty acids rich fish oil against experimental colitis. Toxicol Mech Methods 29:244–254. https://doi.org/10.1080/15376516.2018.1553220
Singh DP, Borse SP, Nivsarkar M (2017) Overcoming the exacerbating effects of ranitidine on NSAID-induced small intestinal toxicity with quercetin: providing a complete GI solution. Chem Biol Interact. 272:53–64. https://doi.org/10.1016/j.cbi.2017.04.006
Somasundaram S, Rafi S, Hayllar J, Sigthorsson G, Jacob M, Price AB, Macpherson A, Mahmod T, Scott D, Wrigglesworth JM, Bjarnason I (1997) Mitochondrial damage: a possible mechanism of the “topical” phase of NSAID induced injury to the rat intestine. Gut 41:344–353. https://doi.org/10.1136/gut.41.3.344
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500. https://doi.org/10.1074/jbc.M113.471342
Takeuchi K, Satoh H (2015) NSAID-induced small intestinal damage - roles of various pathogenic factors. Digestion 91:218–232. https://doi.org/10.1159/000374106
Terán-Ventura E, Aguilera M, Vergara P, Martínez V (2014) Specific changes of gut commensal microbiota and TLRs during indomethacin-induced acute intestinal inflammation in rats. J Crohns Colitis 8:1043–1054. https://doi.org/10.1016/j.crohns.2014.02.001
Wallace JL (2013) Mechanisms, prevention and clinical implications of nonsteroidal anti-inflammatory drug-enteropathy. World J Gastroenterol 19:1861. https://doi.org/10.3748/wjg.v19.i12.1861
Watanabe T, Higuchi K, Kobata A, Nishio H, Tanigawa T, Shiba M, Tominaga K, Fujiwara Y, Oshitani N, Asahara T, Nomoto K, Takeuchi K, Arakawa T (2008) Non-steroidal anti-inflammatory drug-induced small intestinal damage is Toll-like receptor 4 dependent. Gut 57:181–187. https://doi.org/10.1136/gut.2007.125963
Zhu W, Ding Y, Kong W, Li T, Chen H (2018) Docosahexaenoic Acid (DHA) provides neuroprotection in traumatic brain injury models via activating Nrf2-ARE signaling. Inflammation 41:1182–1193. https://doi.org/10.1007/s10753-018-0765-z
Zhu X, Bi Z, Yang C, Guo Y, Yuan J, Li L, Guo Y, 2021. Effects of different doses of omega-3 polyunsaturated fatty acids on gut microbiota and immunity. Food Nutr Res 65. https://doi.org/10.29219/fnr.v65.6263
Acknowledgements
Martha Ivonne Sánchez Trigueros is a CONACyT, Mexico fellow (Grant Number 781715). Authors acknowledge to Q.F.B. Martha Patricia González García from Pharmacology Deparment-Cinvestav, M.V.Z. Ricardo Gaxiola Centeno, M.V.Z. Benjamín Chávez Álvarez, M.V.Z. Carlos Giovanni Sam Miranda from UPEAL-Cinvestav for their technical assistance in this project.
Funding
Financial support was provided by Consejo Nacional de Ciencia y Tecnología (CONACyT Project 285416) and SIP, Mexico 20220896.
Author information
Authors and Affiliations
Contributions
MIST conducted all experiments and wrote the draft. EAPP, GC, and AECP analyzed the data. EAP contributed to the experiments presented in Tables 1 and 2. CPC contributed to the design of the experiments performed in Figs. 4 and 5. IMV and DC contributed to the experiments conducted for Fig. 5. AECP wrote the final draft, supervised the research, and edited the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval
The study was evaluated and approved by the Bioethics Committee of Escuela Nacional de Medicina y Homeopatía, Instituto Politécnico Nacional, Mexico City, México (registration number: CBE/021/2019).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sánchez-Trigueros, M.I., Martínez-Vieyra, I.A., Pineda-Peña, E.A. et al. Role of antioxidative activity in the docosahexaenoic acid’s enteroprotective effect in the indomethacin-induced small intestinal injury model. Naunyn-Schmiedeberg's Arch Pharmacol (2023). https://doi.org/10.1007/s00210-023-02881-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-023-02881-z