Abstract
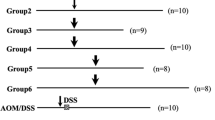
Colitis-associated colorectal cancer (CACC) is one of the devastating complications of long-term inflammatory bowel disease and is associated with substantial morbidity and mortality. Combination of azoxymethane (AOM) and dextran sulfate sodium (DSS) has been extensively used for inflammation-mediated colon tumor development due to its reproducibility, potency, histological and molecular changes, and resemblance to human CACC. In the tumor microenvironment and extra-intestinal tissues, PARP-1, NLRP3 inflammasome, and autophagy’s biological functions are complicated and encompass intricate interactions between these molecular components. The focus of the present investigation is to determine the colonic and extra-intestinal tissue damage induced by AOM-DSS and related molecular mechanisms. Azoxymethane (10 mg/kg, i.p.; single injection) followed by DSS (3 cycles, 7 days per cycle) over a period of 10 weeks induced colitis-associated colon cancer in male BALB/c mice. By initiating carcinogenesis with a single injection of azoxymethane (AOM) and then establishing inflammation with dextran sulfate sodium (DSS), a two-stage murine model for CACC was developed. Biochemical parameters, ELISA, histopathological and immunohistochemical analysis, and western blotting have been performed to evaluate the colonic, hepatic, testicular and pancreatic damage. In addition, the AOM/DSS-induced damage has been assessed by analyzing the expression of a variety of molecular targets, including proliferating cell nuclear antigen (PCNA), interleukin-10 (IL-10), AMP-activated protein kinase (AMPK), poly (ADP-ribose) polymerase-1 (PARP-1), cysteine-associated protein kinase-1 (caspase-1), NLR family pyrin domain containing 3 (NLRP3), beclin-1, and interleukin-1β (IL-1β). Present findings revealed that AOM/DSS developed tumors in colon tissue followed by extra-intestinal hepatic, testicular, and pancreatic damages.
Graphical abstract













Similar content being viewed by others
Data availability
All data and materials support their published claims and comply with field standards. Data will be available on demand.
References
Babu SSN, Singla S, Jena G (2022) Role of combination treatment of aspirin and zinc in DMH-DSS-induced colon inflammation, oxidative stress and tumour progression in male BALB/c mice. Biol Trace Elem Res. https://doi.org/10.1007/s12011-022-03241-3
Biasco G, Derenzini E, Grazi G, Ercolani G, Ravaioli M, Pantaleo MA, Brandi G (2006) Treatment of hepatic metastases from colorectal cancer: many doubts, some certainties. Cancer Treat Rev 32:214–228. https://doi.org/10.1016/j.ctrv.2005.12.011
Biasizzo M, Kopitar-Jerala N (2020) Interplay between NLRP3 inflammasome and autophagy. Front Immunol 11:591803. https://doi.org/10.3389/fimmu.2020.591803
Bush A, Humes R, Young P (2020) Colon cancer metastatic to the pancreas presenting as of diabetic ketoacidosis. ACG Case Rep J 7:e00455. 10.14309/crj.0000000000000455
Chandra R, Karalis JD, Liu C, Murimwa GZ, Voth Park J, Heid CA, Reznik SI, Huang E, Minna JD, Brekken RA (2021) The colorectal cancer tumor microenvironment and its impact on liver and lung metastasis. Cancers (Basel) 13 https://doi.org/10.3390/cancers13246206
Charaya A, Sahu C, Singla S, Jena G (2022) Zinc deficiency exacerbates bisphenol A-induced hepatic and renal damage: delineation of molecular mechanisms. Biol Trace Elem Res. https://doi.org/10.1007/s12011-022-03392-3
Chung IC, OuYang CN, Yuan SN, Lin HC, Huang KY, Wu PS, Liu CY, Tsai KJ, Loi LK, Chen YJ, Chung AK, Ojcius DM, Chang YS, Chen LC (2019) Pretreatment with a heat-killed probiotic modulates the NLRP3 inflammasome and attenuates colitis-associated colorectal cancer in mice. Nutrients 11. https://doi.org/10.3390/nu11030516
Clapper ML, Cooper HS, Chang WC (2007) Dextran sulfate sodium-induced colitis-associated neoplasia: a promising model for the development of chemopreventive interventions. Acta Pharmacol Sin 28:1450–1459. https://doi.org/10.1111/j.1745-7254.2007.00695.x
Das S, Shukla N, Singh SS, Kushwaha S, Shrivastava R (2021) Mechanism of interaction between autophagy and apoptosis in cancer. Apoptosis 26:512–533. https://doi.org/10.1007/s10495-021-01687-9
Devenport SN, Singhal R, Radyk MD, Taranto JG, Kerk SA, Chen B, Goyert JW, Jain C, Das NK, Oravecz-Wilson K, Zhang L, Greenson JK, Chen YE, Soleimanpour SA, Reddy P, Lyssiotis CA, Shah YM (2021) Colorectal cancer cells utilize autophagy to maintain mitochondrial metabolism for cell proliferation under nutrient stress. JCI Insight 6 https://doi.org/10.1172/jci.insight.138835
Dörsam B, Seiwert N, Foersch S, Stroh S, Nagel G, Begaliew D, Diehl E, Kraus A, McKeague M, Minneker V, Roukos V, Reißig S, Waisman A, Moehler M, Stier A, Mangerich A, Dantzer F, Kaina B, Fahrer J (2018) PARP-1 protects against colorectal tumor induction, but promotes inflammation-driven colorectal tumor progression. Proc Natl Acad Sci U S A 115:E4061–e4070. https://doi.org/10.1073/pnas.1712345115
Dziaman T, Ludwiczak H, Ciesla JM, Banaszkiewicz Z, Winczura A, Chmielarczyk M, Wisniewska E, Marszalek A, Tudek B, Olinski R (2014) PARP-1 expression is increased in colon adenoma and carcinoma and correlates with OGG1. PLoS One 9:e115558. https://doi.org/10.1371/journal.pone.0115558
Farombi EO, Adedara IA, Ajayi BO, Idowu TE, Eriomala OO, Akinbote FO (2018) 6-Gingerol improves testicular function in mice model of chronic ulcerative colitis. Hum Exp Toxicol 37:358–372. https://doi.org/10.1177/0960327117703689
Fraczek M, Kurpisz M (2007) Inflammatory mediators exert toxic effects of oxidative stress on human spermatozoa. J Androl 28:325–333. https://doi.org/10.2164/jandrol.106.001149
Gäbele E, Dostert K, Hofmann C, Wiest R, Schölmerich J, Hellerbrand C, Obermeier F (2011) DSS induced colitis increases portal LPS levels and enhances hepatic inflammation and fibrogenesis in experimental NASH. J Hepatol 55:1391–1399. https://doi.org/10.1016/j.jhep.2011.02.035
Grivennikov SI (2013) Inflammation and colorectal cancer: colitis-associated neoplasia. Semin Immunopathol 35:229–244. https://doi.org/10.1007/s00281-012-0352-6
Guo Y, Su ZY, Zhang C, Gaspar JM, Wang R, Hart RP, Verzi MP, Kong AN (2017) Mechanisms of colitis-accelerated colon carcinogenesis and its prevention with the combination of aspirin and curcumin: transcriptomic analysis using RNA-seq. Biochem Pharmacol 135:22–34. https://doi.org/10.1016/j.bcp.2017.02.021
Halliwell B (2023) Understanding mechanisms of antioxidant action in health and disease. Nat Rev Mol Cell Biol. https://doi.org/10.1038/s41580-023-00645-4
Hatoum HA, Abi Saad GS, Otrock ZK, Barada KA, Shamseddine AI (2011) Metastasis of colorectal carcinoma to the testes: clinical presentation and possible pathways. Int J Clin Oncol 16:203–209. https://doi.org/10.1007/s10147-010-0140-z
Irrazabal T, Thakur BK, Croitoru K, Martin A (2021) Preventing colitis-associated colon cancer with antioxidants: a systematic review. Cell Mol Gastroenterol Hepatol 11:1177–1197. https://doi.org/10.1016/j.jcmgh.2020.12.013
Jiang H, Tang Y, Garg HK, Parthasarathy DK, Torregrossa AC, Hord NG, Bryan NS (2012) Concentration- and stage-specific effects of nitrite on colon cancer cell lines. Nitric Oxide 26:267–273. https://doi.org/10.1016/j.niox.2012.03.010
Karageorgou M, Myoteri D, Kotsis T, Polymeneas G, Bournakis E, Dellaportas D (2019) Solitary colorectal cancer metastasis to the pancreas. Case Rep Surg 2019:4891512. https://doi.org/10.1155/2019/4891512
Karki R, Kanneganti TD (2019) Diverging inflammasome signals in tumorigenesis and potential targeting. Nat Rev Cancer 19:197–214. https://doi.org/10.1038/s41568-019-0123-y
Kenific CM, Thorburn A, Debnath J (2010) Autophagy and metastasis: another double-edged sword. Curr Opin Cell Biol 22:241–245. https://doi.org/10.1016/j.ceb.2009.10.008
Kushwaha S, Jena GB (2013) Telmisartan ameliorates germ cell toxicity in the STZ-induced diabetic rat: studies on possible molecular mechanisms. Mutat Res 755:11–23. https://doi.org/10.1016/j.mrgentox.2013.04.013
Li C, Gao Y, Lu C, Guo M (2023) Identification of potential biomarkers for colorectal cancer by clinical database analysis and Kaplan-Meier curves analysis. Medicine (Baltimore) 102:e32877. https://doi.org/10.1097/md.0000000000032877
Li H, Chen YX, Wen JG, Zhou HH (2017) Metastasis-associated in colon cancer 1: a promising biomarker for the metastasis and prognosis of colorectal cancer. Oncol Lett 14:3899–3908. https://doi.org/10.3892/ol.2017.6670
Li W, Zhao T, Wu D, Li J, Wang M, Sun Y, Hou S (2022) Colorectal cancer in ulcerative colitis: mechanisms, surveillance and chemoprevention. Curr Oncol 29:6091–6114. https://doi.org/10.3390/curroncol29090479
Li X, Gao Y, Yang M, Zhao Q, Wang G, Yang YM, Yang Y, Liu H, Zhang Y (2014) Identification of gene expression changes from colitis to CRC in the mouse CAC model. PLoS One 9:e95347. https://doi.org/10.1371/journal.pone.0095347
Liang X, Hu JN, He JM (2019) An optimized protocol of azoxymethane-dextran sodium sulfate induced colorectal tumor model in mice. Chin Med Sci J 34:281–288. https://doi.org/10.24920/003495
Liu H, Lou J, Liu Y, Liu Z, Xie J, Sun J, Pan H, Han W (2022) Intestinal epithelial cell autophagy deficiency suppresses inflammation-associated colon tumorigenesis. Mol Ther Nucleic Acids 28:35–46. https://doi.org/10.1016/j.omtn.2022.02.012
Liu Y, Zhang Y, Zhao Y, Gao D, Xing J, Liu H (2016) High PARP-1 expression is associated with tumor invasion and poor prognosis in gastric cancer. Oncol Lett 12:3825–3835. https://doi.org/10.3892/ol.2016.5169
Martin J, Petrillo A, Smyth EC, Shaida N, Khwaja S, Cheow HK, Duckworth A, Heister P, Praseedom R, Jah A, Balakrishnan A, Harper S, Liau S, Kosmoliaptsis V, Huguet E (2020) Colorectal liver metastases: current management and future perspectives. World J Clin Oncol 11:761–808. https://doi.org/10.5306/wjco.v11.i10.761
Megaraj V, Ding X, Fang C, Kovalchuk N, Zhu Y, Zhang QY (2014) Role of hepatic and intestinal p450 enzymes in the metabolic activation of the colon carcinogen azoxymethane in mice. Chem Res Toxicol 27:656–662. https://doi.org/10.1021/tx4004769
Mirończuk-Chodakowska I, Witkowska AM, Zujko ME (2018) Endogenous non-enzymatic antioxidants in the human body. Adv Med Sci 63:68–78. https://doi.org/10.1016/j.advms.2017.05.005
Modesto R, Estarreja J, Silva I, Rocha J, Pinto R, Mateus V (2022) Chemically induced colitis-associated cancer models in rodents for pharmacological modulation: a systematic review. J Clin Med 11. https://doi.org/10.3390/jcm11102739
Muanprasat C, Wongkrasant P, Satitsri S, Moonwiriyakit A, Pongkorpsakol P, Mattaveewong T, Pichyangkura R, Chatsudthipong V (2015) Activation of AMPK by chitosan oligosaccharide in intestinal epithelial cells: mechanism of action and potential applications in intestinal disorders. Biochem Pharmacol 96:225–236. https://doi.org/10.1016/j.bcp.2015.05.016
Mulcahy Levy JM, Thorburn A (2020) Autophagy in cancer: moving from understanding mechanism to improving therapy responses in patients. Cell Death Differ 27:843–857. https://doi.org/10.1038/s41418-019-0474-7
Muñoz-Gámez JA, Rodríguez-Vargas JM, Quiles-Pérez R, Aguilar-Quesada R, Martín-Oliva D, De Murcia G, De Murcia JM, Almendros A, De Almodóvar MR, Oliver FJ (2009) PARP-1 is involved in autophagy induced by DNA damage. Autophagy 5:61–74. https://doi.org/10.4161/auto.5.1.7272
Neufert C, Becker C, Neurath MF (2007) An inducible mouse model of colon carcinogenesis for the analysis of sporadic and inflammation-driven tumor progression. Nat Protoc 2:1998–2004. https://doi.org/10.1038/nprot.2007.279
Pai Bellare G, Saha B, Patro BS (2021) Targeting autophagy reverses de novo resistance in homologous recombination repair proficient breast cancers to PARP inhibition. Br J Cancer 124:1260–1274. https://doi.org/10.1038/s41416-020-01238-0
Pai RK, Jairath V, Vande Casteele N, Rieder F, Parker CE, Lauwers GY (2018) The emerging role of histologic disease activity assessment in ulcerative colitis. Gastrointest Endosc 88:887–898. https://doi.org/10.1016/j.gie.2018.08.018
Paldino E, D'Angelo V, Laurenti D, Angeloni C, Sancesario G, Fusco FR (2020) Modulation of inflammasome and pyroptosis by olaparib, a PARP-1 inhibitor, in the R6/2 mouse model of Huntington’s disease. Cells 9. https://doi.org/10.3390/cells9102286
Park JM, Lee DH, Kim DH (2023) Redefining the role of AMPK in autophagy and the energy stress response. Nat Commun 14:2994. https://doi.org/10.1038/s41467-023-38401-z
Parmar AR, Trivedi PP, Jena GB (2014) Dextran sulfate sodium-induced ulcerative colitis leads to testicular toxicity in mice: role of inflammation, oxidative stress and DNA damage. Reprod Toxicol 49:171–184. https://doi.org/10.1016/j.reprotox.2014.08.004
Qin Y, Yu Y, Yang C, Wang Z, Yang Y, Wang C, Zheng Q, Li D, Xu W (2021) Atractylenolide I inhibits NLRP3 inflammasome activation in colitis-associated colorectal cancer via suppressing Drp1-mediated mitochondrial fission. Front Pharmacol 12:674340. https://doi.org/10.3389/fphar.2021.674340
Roediger WE, Lawson MJ, Radcliffe BC (1990) Nitrite from inflammatory cells--a cancer risk factor in ulcerative colitis? Dis Colon Rectum 33:1034–1036. https://doi.org/10.1007/bf02139219
Rose M, Burgess JT, O’Byrne K, Richard DJ, Bolderson E (2020) PARP inhibitors: clinical relevance, mechanisms of action and tumor resistance. Front Cell Dev Biol 8:564601. https://doi.org/10.3389/fcell.2020.564601
Segnani C, Ippolito C, Antonioli L, Pellegrini C, Blandizzi C, Dolfi A, Bernardini N (2015) Histochemical detection of collagen fibers by Sirius Red/Fast Green is more sensitive than van Gieson or Sirius Red alone in normal and inflamed rat colon. PLoS One 10:e0144630. https://doi.org/10.1371/journal.pone.0144630
Shi F, Wei B, Lan T, Xiao Y, Quan X, Chen J, Zhao C, Gao J (2021) Low NLRP3 expression predicts a better prognosis of colorectal cancer. Biosci Rep 41. https://doi.org/10.1042/bsr20210280
Singla S, Jena G (2022) Intervention of 3-aminobenzamide against dextran sulphate sodium induced colitis in mice: investigations on molecular mechanisms. Eur J Pharmacol 920:174861. https://doi.org/10.1016/j.ejphar.2022.174861
Singla S, Sahu C, Jena G (2022) Association of type 1 diabetes with ulcerative colitis in BALB/c mice: investigations on sex-specific differences. J Biochem Mol Toxicol 36:e22980. https://doi.org/10.1002/jbt.22980
Snider AJ, Bialkowska AB, Ghaleb AM, Yang VW, Obeid LM, Hannun YA (2016) Murine model for colitis-associated cancer of the colon. Methods Mol Biol 1438:245–254. https://doi.org/10.1007/978-1-4939-3661-8_14
Sperti C, Pasquali C, Berselli M, Frison L, Vicario G, Pedrazzoli S (2009) Metastasis to the pancreas from colorectal cancer: is there a place for pancreatic resection? Dis Colon Rectum 52:1154–1159. https://doi.org/10.1007/DCR.0b013e31819f7397
Talero E, Alcaide A, Ávila-Román J, García-Mauriño S, Vendramini-Costa D, Motilva V (2016) Expression patterns of sirtuin 1-AMPK-autophagy pathway in chronic colitis and inflammation-associated colon neoplasia in IL-10-deficient mice. Int Immunopharmacol 35:248–256. https://doi.org/10.1016/j.intimp.2016.03.046
Tanimura Y, Fukui T, Horitani S, Matsumoto Y, Miyamoto S, Suzuki R, Tanaka T, Tomiyama T, Ikeura T, Ando Y, Nishio A, Okazaki K (2021) Long-term model of colitis-associated colorectal cancer suggests tumor spread mechanism and nature of cancer stem cells. Oncol Lett 21:7. https://doi.org/10.3892/ol.2020.12268
Thaker AI, Shaker A, Rao MS, Ciorba MA (2012) Modeling colitis-associated cancer with azoxymethane (AOM) and dextran sulfate sodium (DSS). J Vis Exp. https://doi.org/10.3791/4100
Tong Y, Yang W, Koeffler HP (2011) Mouse models of colorectal cancer. Chin J Cancer 30:450–462. https://doi.org/10.5732/cjc.011.10041
Trivedi PP, Jena GB (2012) Dextran sulfate sodium-induced ulcerative colitis leads to increased hematopoiesis and induces both local as well as systemic genotoxicity in mice. Mutat Res 744:172–183. https://doi.org/10.1016/j.mrgentox.2012.03.001
Vafaei S, Taheri H, Hajimomeni Y, Fakhre Yaseri A, Abolhasani Zadeh F (2022) The role of NLRP3 inflammasome in colorectal cancer: potential therapeutic target. Clin Transl Oncol 24:1881–1889. https://doi.org/10.1007/s12094-022-02861-4
Vatandoust S, Price TJ, Karapetis CS (2015) Colorectal cancer: metastases to a single organ. World J Gastroenterol 21:11767–11776. https://doi.org/10.3748/wjg.v21.i41.11767
Villarán RF, Espinosa-Oliva AM, Sarmiento M, De Pablos RM, Argüelles S, Delgado-Cortés MJ, Sobrino V, Van Rooijen N, Venero JL, Herrera AJ, Cano J, Machado A (2010) Ulcerative colitis exacerbates lipopolysaccharide-induced damage to the nigral dopaminergic system: potential risk factor in Parkinson’s disease. J Neurochem 114:1687–1700. https://doi.org/10.1111/j.1471-4159.2010.06879.x
Wang F, Liu W, Jiang Q, Gong M, Chen R, Wu H, Han R, Chen Y, Han D (2019a) Lipopolysaccharide-induced testicular dysfunction and epididymitis in mice: a critical role of tumor necrosis factor alpha†. Biol Reprod 100:849–861. https://doi.org/10.1093/biolre/ioy235
Wang X, Tu W, Chen D, Fu J, Wang J, Shao C, Zhang J (2019b) Autophagy suppresses radiation damage by activating PARP-1 and attenuating reactive oxygen species in hepatoma cells. Int J Radiat Biol 95:1051–1057. https://doi.org/10.1080/09553002.2019.1605461
Wu Y, Yao J, Xie J, Liu Z, Zhou Y, Pan H, Han W (2018) The role of autophagy in colitis-associated colorectal cancer. Signal Transduct Target Ther 3:31. https://doi.org/10.1038/s41392-018-0031-8
Yao J, Zhao L, Zhao Q, Zhao Y, Sun Y, Zhang Y, Miao H, You QD, Hu R, Guo QL (2014) NF-κB and Nrf2 signaling pathways contribute to wogonin-mediated inhibition of inflammation-associated colorectal carcinogenesis. Cell Death Dis 5:e1283. https://doi.org/10.1038/cddis.2014.221
Yashiro M (2014) Ulcerative colitis-associated colorectal cancer. World J Gastroenterol 20:16389–16397. https://doi.org/10.3748/wjg.v20.i44.16389
Zhang MY, Gou WF, Zhao S, Mao XY, Zheng ZH, Takano Y, Zheng HC (2014) Beclin 1 expression is closely linked to colorectal carcinogenesis and distant metastasis of colorectal carcinoma. Int J Mol Sci 15:14372–14385. https://doi.org/10.3390/ijms150814372
Zhao Y, Guo Q, Zhao K, Zhou Y, Li W, Pan C, Qiang L, Li Z, Lu N (2017) Small molecule GL-V9 protects against colitis-associated colorectal cancer by limiting NLRP3 inflammasome through autophagy. Oncoimmunology 7:e1375640. https://doi.org/10.1080/2162402x.2017.1375640
Zhou H, Liu Z, Wang Y, Wen X, Amador EH, Yuan L, Ran X, Xiong L, Ran Y, Chen W, Wen Y (2022) Colorectal liver metastasis: molecular mechanism and interventional therapy. Signal Transduct Target Ther 7:70. https://doi.org/10.1038/s41392-022-00922-2
Funding
The authors are thankful to the National Institute of Pharmaceutical Education and Research (NIPER), S.A.S. Nagar (Vide no. NPLC-GBJ-2021-2022), and Department of Science and Technology (DST), New Delhi (SERB File Number: CRG/2020/000412), for providing financial support to perform the experiment work related to the present study.
Author information
Authors and Affiliations
Contributions
S.S. conceived and conducted the experiments, analyzed the data, and wrote the manuscript. G.B.J. conceived the idea, reviewed the manuscript, and administered the project. Finally, both the authors have read and approved the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Competing interests
Authors of the current investigation declared no competing interests.
Ethical approval
All institutional and national guidelines for the care and use of laboratory animals were followed. The animal studies were approved by the Ethics Committee of the NIPER SAS Nagar (IAEC/19/21).
Informed consent
Informed consent is not applicable in the present study.
Consent for publication
Both authors are willing to publish their research work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singla, S., Jena, G. Studies on the mechanism of local and extra-intestinal tissue manifestations in AOM-DSS-induced carcinogenesis in BALB/c mice: role of PARP-1, NLRP3, and autophagy. Naunyn-Schmiedeberg's Arch Pharmacol (2023). https://doi.org/10.1007/s00210-023-02878-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-023-02878-8