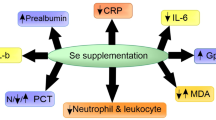
Abstract
Trace elements are cofactors in various enzymes in the antioxidant defense and cell homeostasis required in the tissue during inflammation. In acute kidney injury induced by lipopolysaccharide (LPS), renal cells are affected by cytotoxicity. Renal evacuation and gastrointestinal absorption rates are important in regulating plasma levels of trace elements. Simvastatin is a widely used anti-lipidemic drug with known anti-inflammatory effects. This study aimed to examine the effect of simvastatin on trace elements and electrolyte levels in kidney tissue in rats with LPS-induced sepsis. Adult male Wistar albino rats were divided into four groups: control, LPS (20 mg/kg, i.p., single dose), simvastatin (20 mg/kg, o.p., 5 days), and LPS + Simvastatin (LPS + Sim). Sodium, potassium, calcium, magnesium, selenium, zinc, copper, and histological structural changes were examined in kidney tissue samples 4 h after LPS execution. The inductively coupled plasma optical emission spectroscopy technique (ICP-OES) was used to determine the tissue trace element levels. In rats with sepsis-induced LPS, selenium, calcium, sodium, and magnesium levels significantly decreased while copper, potassium, and zinc levels significantly increased compared to other experimental groups. In sepsis treated with the simvastatin (LPS + Simvastatin) group, trace elements and electrolyte levels are like the control groups, apart from selenium levels. Selenium levels were significantly decreased in the LPS + Simvastatin group compared to the controls. As a result of examining the kidney tissues under a light microscope, simvastatin improved tissue damage caused by LPS-induced acute kidney injury. LPS-induced renal injury and simvastatin caused significant changes in the oxidant/antioxidant system. In septic rats, simvastatin was shown to balance some trace element levels, and it may improve damage in the kidney tissue.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Adekola OO, Soriyan OO, Meka I et al (2012) The incidence of electrolyte and acid-base abnormalities in critically ill patients using point of care testing (i-STAT portable analyser). Nig Q J Hosp Med 22(2):103–108
Agarwal A, Anand K, Manish G et al (2018) Trace elements and sepsis: is there a correlation. Int J Med Public Health 8(4):133–139
Akkaş İ, Ince N, Sungur MA (2020) Serum trace element and heavy metal levels in patients with sepsis. Aging Male 23(3):222–226. https://doi.org/10.1080/13685538.2020.1740200
Ateş G, Özkök E, Yorulmaz H et al (2023) Serum YKL-40 as candidate biomarker in sepsis-induced lung injury. Cerrahpaşa Med J 47(2):156–161. https://doi.org/10.5152/cjm.2023.22123
Ayoglu H, Sezer U, Akin M et al (2016) Selenium, copper, zinc, iron levels and mortality in patients with sepsis and systemic inflammatory response syndrome in Western Black Sea Region, Turkey. J Pak Med Assoc 66(4):447–452
Barin JS, Pereira JSF, Mello PA, Knorr CL, Moraes DP, Mesko MF, Flores EMM (2012) Focused microwave-induced combustion for digestion of botanical samples and metals determination by ICP OES and ICP-MS. Talanta 94:308–314
Biasucci LM, Biasillo G, Stefanelli A (2010) Inflammatory markers, cholesterol and statins: pathophysiological role and clinical importance. Clin Chem Lab Med 48(12):1685–1691
Cao L, Zheng J, Tsukada H, Pan S, Wang Z, Tagami K, Uchida S (2016) Simultaneous determination of radiocesium (135Cs, 137Cs) and plutonium (239Pu, 240Pu) isotopes in river suspended particles by ICP-MS/MS and SF-ICP-MS. Talanta 159:55–63
Cases J, Vacchina V, Napolitano A et al (2001) Selenium from selenium-rich spirulina is less bioavailable than selenium from sodium selenite and selenomethionine in selenium-deficient rats. J Nutr 131(9):2343–2350. https://doi.org/10.1093/jn/131.9.2343
Ceballos-Marquez A, Barkema HW, Stryhn H (2010) The effect of selenium supplementation before calving on early-lactation udder health in pastured dairy heifers. J Dairy Sci 93(10):4602–4612. https://doi.org/10.3168/jds.2010-3086
Dickson K, Lehmann C (2019) Inflammatory response to different toxins in experimental sepsis models. Int J Mol Sci 20(18):4341. https://doi.org/10.3390/ijms20184341
Duntas LH (2009) Selenium and inflammation: underlying antiinflammatory mechanisms. Horm Metab Res 41:443–447. https://doi.org/10.1055/s-0029-1220724
Durant R, Klouche K, Delbosc S et al (2004) Superoxide anion over production in sepsis: effects of vitamin and simvastatin. Shock 22(1):34–39
Escobedo-Monge MF, Barrado E, Parodi-Román J et al (2021) Copper and copper/Zn ratio in a series of children with chronic diseases: a cross-sectional study. Nutrients 13(10):3578. https://doi.org/10.3390/nu13103578
Ghayour-Mobarhan M, Lamb DJ, Taylor A et al (2005) Effect of statin therapy on serum trace element status in dyslipidaemic subjects. J Trace Elem Med Biol 19(1):61–67. https://doi.org/10.1016/j.jtemb.2005.06.003
Gitlin JD, Schroeder JJ, Lee-Ambrose LM et al (1992) Mechanisms of caeruloplasmin biosynthesis in normal and copper-deficient rats. Biochem 282(Pt 3):835–839
Goncalves DA, de Souza ID, Rosa ACG, Melo ESP, Goncalves AMB, de Oliveira LCS, do Nascimento VA (2019) Multi-wavelength calibration: determination of trace toxic elements in medicine plants by ICP OES. Microchem J 146:381–386
Guo CH, Chen PC, Yeh MS et al (2011) Cu/Zn ratios are associated with nutritional status, oxidative stress, inflammation, and immune abnormalities in patients on peritoneal dialysis. Clin Biochem 44(4):275–280. https://doi.org/10.1016/j.clinbiochem.2010.12.017
Han M, Lin Z, Zhang Y (2013) The alteration of copper homeostasis in inflammation induced by lipopolysaccharides. Biol Trace Elem Res 154:268–274. https://doi.org/10.1007/s12011-013-9725-5
Hendy GN, Canaff L (2016) Calcium-sensing receptor, proinflammatory cytokines and calcium homeostasis. Semin Cell Dev Biol 49:37–43. https://doi.org/10.1016/j.semcdb.2015.11.006
Heyland DK, Dhaliwal R, Suchner U et al (2005) Antioxidant nutrients: a systematic review of trace elements and vitamins in the critically ill patient. Intensive Care Med 31(3):327–337. https://doi.org/10.1007/s00134-004-2522-z
Jung SY, Kim H, Park S et al (2016) Electrolyte and mineral disturbances in septic acute kidney injury patients undergoing continuous renal replacement therapy. Medicine (baltimore) 95(36):e4542. https://doi.org/10.1097/MD.0000000000004542
Júnior ADFS, Korn MDGA (2018) Effects of furosemide administration on the concentration of essential and toxic elements in Wistar rats by inductively coupled plasma optical emission spectrometry. J Trace Elem Med Biol 48:25–29
Kim SH, Johnson VJ, Shin TY et al (2004) Selenium attenuates lipopolysaccharide-induced oxidative stress responses through modulation of p38 MAPK and NF-kappaB signaling pathways. Exp Biol Med 229(2):203–213. https://doi.org/10.1177/153537020422900209
Laine JT, Tuomainen TP, Salonen JT et al (2020) Serum copper-to-zinc-ratio and risk of incident infection in men: the Kuopio Ischaemic Heart Disease Risk Factor Study. Eur J Epidemiol 35:1149–1156. https://doi.org/10.1007/s10654-020-00644-1
Legrand M, Dupuis C, Simon C et al (2013) Association between systemic hemodynamics and septic acute kidney injury in critically ill patients: a retrospective observational study. Crit Care 17:R278. https://doi.org/10.1186/cc13133
Leone N, Courbon D, Ducimetiere P et al (2006) Zinc, copper, and magnesium and risks for all-cause, cancer, and cardiovascular mortality. Epidemiology 17:308–314. https://doi.org/10.1097/01.ede.0000209454.41466.b7
Lepage R, Legare G, Racicot C et al (1999) Hypocalcemia induced during major and minor abdominal surgery in humans. J Clin Endocrinol Metab 84:2654–2658. https://doi.org/10.1210/jcem.84.8.5889
Malavolta M, Piacenza F, Basso A et al (2015) Serum copper to zinc ratio: relationship with aging and health status. Mech Ageing Dev 151:93–100. https://doi.org/10.1016/j.mad.2015.01.004
Manzanares W, Biestro A, Galusso F et al (2009) Serum selenium and glutathione peroxidase-3 activity: biomarkers of systemic inflammation in the critically ill? Intensive Care Med 35:882–889. https://doi.org/10.1007/s00134-008-1356-5
Mayeux PR, MacMillan-Crow LA (2012) Pharmacological targets in the renal peritubular microenvironment: implications for therapy for sepsis induced acute kidney injury. Pharmacol Ther 134:139–155. https://doi.org/10.1016/j.pharmthera.2012.01.004
McDonnell MA, Wang D, Khan SM et al (2003) Caspase-9 is activated in a cytochrome c-independent manner early during TNFα-induced apoptosis in murine cells. Cell Death Diff 10:1005–1015. https://doi.org/10.1038/sj.cdd.4401271
Merx MW, Liehn EA, Janssens U et al (2004) HMG-CoA reductase inhibitor simvastatin profoundly improves survival in a murine model of sepsis. Circulation 109:2560–2565
Nedjimi B (2021) Can trace element supplementations (Cu, Se, and Zn) enhance human immunity against COVID-19 and its new variants? Beni Suef Univ J Basic Appl Sci 10(1):33. https://doi.org/10.1186/s43088-021-00123-w
Nezić L, Skrbić R, Dobrić S et al (2009) Effect of simvastatin on proinflammatory cytokines production during lipopolysaccharide-induced inflammation in rats. Gen Physiol Biophys 28:119–126
Ota T, Ota K, Jono H et al (2010) Midkine expression in malignant salivary gland tumors and its role in tumor angiogenesis. Oral Oncol 46:657–661. https://doi.org/10.1016/j.oraloncology.2010.06.004
Palmer BF, Clegg DJ (2018) Hyperkalemia across the continuum of kidney function. Clin J Am Soc Nephrol 13(1):155–157. https://doi.org/10.2215/CJN.09340817
Prelack K, Sheridan RL (2001) Micronutrient supplementation in the critically ill patient: strategies for clinical practice. J Trauma 51(3):601–620. https://doi.org/10.1097/00005373-200109000-00037
Quasim T, McMillan DC, Talwar D et al (2003) Lower concentrations of carotenoids in the critically ill patient are related to a systemic inflammatory response and increased lipid peroxidation. Clin Nutr 22(5):459–462. https://doi.org/10.1016/s0261-5614(03)00044-x
Ramesh G, Reeves WB (2003) TNFR2-mediated apoptosis and necrosis in cisplatin-induced acute renal failure. Am J Physiol Renal Physiol 285:F610–F618. https://doi.org/10.1152/ajprenal.00101.2003
Saadat SH, Goodarzi R (2020) The relation of copper and zinc with incidence of neonatal sepsis and possible prediction biomarker role. Galen Med J 9:e1933. https://doi.org/10.31661/gmj.v9i0.1933
Saner G, Baysal SU, Unuvar E et al (2000) Serum zinc, copper levels, and copper/zinc ratios in infants with sepsis syndrome. J Trace Elem Exp Med 13:265–270
Schmidt C, Höcherl K, Schweda F et al (2007) Regulation of renal sodium transporters during severe inflammation. J Am Soc Nephrol 18(4):1072–1083. https://doi.org/10.1681/ASN.2006050454
Schroeder JJ, Cousins RJ (1990) Interleukin 6 regulates metallothionein gene expression and zinc metabolism in hepatocyte monolayer cultures. Proc Natl Acad Sci U S A 87(8):3137–3141
Singer M, Deutschman CS, Seymour CW et al (2016) The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 315(8):801–810. https://doi.org/10.1001/jama.2016.0287
Singh A, Smoak BL, Patterson KY et al (1991) Biochemical indices of selected trace minerals in men: effect of stress. Am J Clin Nutr 53(1):126–131. https://doi.org/10.1093/ajcn/53.1.126
Stafford SL, Bokil NJ, Achard MES et al (2013) Metal ions in macrophage antimicrobial pathways: emerging roles for zinc and copper. Biosci Rep 33:e00049. https://doi.org/10.1042/bsr20130014
Steele T, Kolamunnage-Dona R, Downey C et al (2013) Assessment and clinical course of hypocalcemia in critical illness. Crit Care 17:R106. https://doi.org/10.1186/cc12756
Taccone-Gallucci M, Noce A, Bertucci P et al (2010) Chronic treatment with statins increases the availability of selenium in the antioxidant defence systems of hemodialysis patients. J Trace Elem Med Biol 24(1):27–30. https://doi.org/10.1016/j.jtemb.2009.06.005
Vunta H, Belda BJ, Arner RJ et al (2008) Selenium attenuates pro-inflammatory gene expression in macrophages. Mol Nutr Food Res 52(11):1316–1323. https://doi.org/10.1002/mnfr.200700346
Wang D, Zheng J, Hu Q et al (2020) Magnesium protects against sepsis by blocking gasdermin D N-terminal-induced pyroptosis. Cell Death Differ 27(2):466–481. https://doi.org/10.1038/s41418-019-0366-x
Yalcin IE, Altay V (2023) Investigation of water-soil-plant relationships based on hazardous and macro-micro element concentrations on Orontes River, Türkiye. Int J Phytoremediation 25(14):1859–1880. https://doi.org/10.1080/15226514.2023.2202241
Yasuda H, Yuen PS, Hu X et al (2006) Simvastatin improves sepsis-induced mortality and acute kidney injury via renal vascular effects. Kidney Int 69(9):1535–1542. https://doi.org/10.1038/sj.ki.5000300
Yorulmaz H, Ozkok E, Demir G et al (2020) Pretreatment of simvastatin on liver trace element levels during endotoxemia. Arch Physiol Biochem 126(3):196–200. https://doi.org/10.1080/13813455.2018.1508234
Zager RA, Johnson AC, Lund S et al (2006) Levosimendan protects against experimental endotoxemic acute renal failure. Am J Physiol Renal Physiol 290(6):F1453–F1462. https://doi.org/10.1152/ajprenal.00485.2005
Zhang T, Lu X, Li J et al (2012) Inhibition of Na/K-ATPase promotes myocardial tumor necrosis factor-alpha protein expression and cardiac dysfunction via calcium/mTOR signaling in endotoxemia. Basic Res Cardiol 107(2):254. https://doi.org/10.1007/s00395-012-0254-8
Zivin JR, Cooley T, Zager RA et al (2001) Hypocalcemia: a pervasive metabolic abnormality in the critically ill. Am J Kid Dis 37:689–698
Zou ZG, Rios FJ, Montezano AC et al (2019) TRPM7, magnesium, and signaling. Int J Mol Sci 20(8):1877. https://doi.org/10.3390/ijms20081877
Funding
There was no financial support used in this study.
Author information
Authors and Affiliations
Contributions
G.A, E.O., S.T., wrote the main manuscript. G.A. prepared figures. All author reviewed the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval
This study was conducted at the Animal Experiments Local Ethics Committee, Istanbul University (Resolution No: 2012/138).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ates, G., Tamer, S., Ozkok, E. et al. Determination of trace elements and electrolyte levels in kidney tissue of simvastatin-treated septic rats. Naunyn-Schmiedeberg's Arch Pharmacol 397, 3513–3521 (2024). https://doi.org/10.1007/s00210-023-02835-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02835-5