Abstract
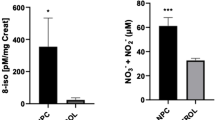
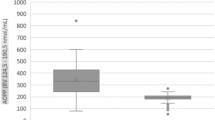
Niemann-Pick type C1 (NP-C1) is a lysosomal storage disease (LSD) caused by mutations in NPC1 gene that lead to defective synthesis of the respective lysosomal transporter protein and cholesterol accumulation in late endosomes/lysosomes (LE/L) compartments, as well as glycosphingolipids GM2 and GM3 in the central nervous system (CNS). Clinical presentation varies according to the age of onset and includes visceral and neurological symptoms, such as hepatosplenomegaly and psychiatric disorders. Studies have been associating the pathophysiology of NP-C1 with oxidative damage to lipids and proteins, as well as evaluating the benefits of adjuvant therapy with antioxidants for this disease. In this work, we evaluated the DNA damage in fibroblasts culture from patients with NP-C1 treated with miglustat, as well as the in vitro effect of the antioxidant compounds N-acetylcysteine (NAC) and Coenzyme Q10 (CoQ10), using the alkaline comet assay. Our preliminary results demonstrate that NP-C1 patients have increased DNA damage compared to healthy individuals and that the treatments with antioxidants can mitigate it. DNA damage may be due to an increase in reactive species since it has been described that NP-C1 patients have increased peripheral markers of damage to other biomolecules. Our study suggests that NP-C1 patients could benefit from the use of adjuvant therapy with NAC and CoQ10, which should be better evaluated in a future clinical trial.



Similar content being viewed by others
Data availability
Not applicable.
References
Biancini GB, Moura DJ, Manini PR, Faverzani JL, Netto CBO, Deon M, Giugliani R, Saffi J, Vargas CR (2015) Mutat Res/Genet Toxicol Environ Mutagen 784–785:31–36
Cuisset JM, Sukno S, Trauffler A, Latour P, Dobbelaere D, Michaud L, and Vallée L (n.d.) J Med Case Rep https://doi.org/10.1186/s13256-016-1038-9
Dardis A, Zampieri S, Canterini S, Newell KL, Stuani C, Murrell JR, Ghetti B, Fiorenza MT, Bembi B, Buratti E (2016) Acta Neuropathol Commun 4:1–13
Dean O, Giorlando F, Berk M (2011) J Psychiatry Neurosci 36:78–86
Diaz Jacques CE, de Souza HM, Sperotto NDM, Veríssimo RM, da Rosa HT, Moura DJ, Saffi J, Giugliani R, Vargas CR (2018) Mutat Res/Gen Toxicol Environ Mutagen 835:21–24
Dizdaroglu M (2012) Cancer Lett 327:26–47
Donida B, Raabe M, Tauffner B, Farias MA, Machado AZ, Timm F, Kessler RG, Hammerschmidt TG, Reinhardt LS, Brito VB, Portugal RV, Bernardi A, Frozza R, Moura DJ, Giugliani R, Poletto F, Vargas CR (2020) J Inherit Metab Dis 43:586–601
Forbes DP (2013) Biomarker validation for niemann-pick disease, type C: safety and efficacy of N-acetyl cysteine. https://clinicaltrials.gov/ct2/show/study/NCT00975689. Accessed 24 Jan 2023
Fu R, Wassif CA, Yanjanin NM, Watkins-Chow DE, Baxter LL, Incao A, Liscum L, Sidhu R, Firnkes S, Graham M, Ory DS, Porter FD, Pavan WJ (2013) Hum Mol Genet 22:3508–3523
Fu R, Yanjanin NM, Bianconi S, Pavan WJ, Porter FD (2010) Mol Genet Metab 101:214–218
Garver WS, Francis GA, Jelinek D, Shepherd G, Flynn J, Castro G, Vockley CW, Coppock DL, Pettit KM, Heidenreich RA, and Meaney FJ (2007) Am J Med Genet A https://doi.org/10.1002/ajmg.a.31735
Halliwell B (2001) Drugs Aging 18:685–16
Halliwell B (2006) Plant Physiol 141:312–322
Hammerschmidt TG, de Oliveira Schmitt Ribas G, Saraiva-Pereira ML, Bonatto MP, Kessler RG, Souza FTS, Trapp F, Michelin-Tirelli K, Burin MG, Giugliani R, Vargas CR (2018) Int J Dev Neurosci 66:18–23
Hammerschmidt TG, Donida B, Faverzani JL, Moura AP, dos Reis BG, Machado AZ, Kessler RG, Sebastião FM, Reinhardt LS, Moura DJ, Vargas CR (2022) Exp Cell Res 416:113175
Hargreaves I (2003) Ann Clin Biochem 40:207–218
Heese BA (2008) Semin Pediatr Neurol 15
Héron B, Valayannopoulos V, Baruteau J, Chabrol B, Ogier H, Latour P, Dobbelaere D, Eyer D, Labarthe F, Maurey H, Cuisset JM, de Villemeur TB, Sedel F, and Vanier M (2012) Orphanet J Rare Dis https://doi.org/10.1186/1750-1172-7-36
Isobe C, Abe T, Terayama Y (2010) J Neurol 257:399–404
Klein A, Maldonado C, Vargas LM, Gonzalez M, Robledo F, Perez de Arce K, Muñoz FJ, Hetz C, Alvarez AR, Zanlungo S (2011) Neurobiol Dis 41:209–218
Long Y, Xu M, Li R, Dai S, Beers J, Chen G, Soheilian F, Baxa U, Wang M, Marugan JJ, Muro S, Li Z, Brady R, and Zheng W (2016) Stem Cells Transl Med https://doi.org/10.5966/sctm.2015-0373
Marín T, Contreras P, Castro JF, Chamorro D, Balboa E, Bosch-Morató M, Muñoz FJ, Alvarez AR, Zanlungo S (2014) Nutrients 6:3000–3017
Migliore L, Molinu S, Naccarati A, Mancuso M, Rocchi A, and Siciliano G (2004) Mutagenesis https://doi.org/10.1093/mutage/geg036
Montero R, Yubero D, Salgado MC, González MJ, Campistol J, del M. O’Callaghan M, Pineda M, Delgadillo V, Maynou J, Fernandez G, Montoya J, Ruiz-Pesini E, Meavilla S, Neergheen V, García-Cazorla A, Navas P, Hargreaves I and Artuch R (2019) Sci Rep https://doi.org/10.1038/s41598-018-37542-2
Platt FM, d’Azzo A, Davidson BL, Neufeld EF, Tifft CJ (2018) Nat Rev Dis Primers 4:1–25
Patterson MC (2003) Neurologist 9:301–310
Patterson MC, Hendriksz CJ, Walterfang M, Sedel F, Vanier MT, and Wijburg F (2012) Mol Genet Metab https://doi.org/10.1016/j.ymgme.2012.03.012
Parasassi T, Brunelli R, Costa G, de Spirito M, Krasnowska EK, Lundeberg T, Pittaluga E, Ursini F (2010) Sci World J 10:1192–1202
Repetto G, del Peso A, Zurita JL (2008) Nat Protoc 3:1125–1131
Ribas GS, Pires R, Coelho JC, Rodrigues D, Mescka CP, Vanzin CS, Biancini GB, Negretto G, Wayhs CAY, Wajner M, Vargas CR (2012) Int J Dev Neurosci 30:439–444
Singh NP, McCoy MT, Tice RR, and Schneider EL (1988) Exp Cell Res. https://doi.org/10.1016/0014-4827(88)90265-0
Sitarska D, Ługowska A (2019) Metab Brain Dis 34:1253–1260
Smith D, Wallom KL, Williams IM, Jeyakumar M, Platt FM (2009) Neurobiol Dis 36:242–251
Souto EB, Campos JR, Da Ana R, Martins-Gomes C, Silva AM, Souto SB, Lucarini M, Durazzo A, and Santini A (2020) Int J Environ Res Public Health https://doi.org/10.3390/ijerph17062046
Sung HJ, Kim J, Kim Y, Jang S-W, Ko J (2012) Mol Biol Rep 39:3001–3007
Thomas R, Kermode AR (2019) Mol Genet Metab 126:83–97
Tomasetti M, Alleva R, Collins AR (2001) FASEB J 15:1425–1427
Torres S, Matías N, Baulies A, Nuñez S, Alarcon-Vila C, Martinez L, Nuño N, Fernandez A, Caballeria J, Levade T, Gonzalez-Franquesa A, Garcia-Rovés P, Balboa E, Zanlungo S, Fabrías G, Casas J, Enrich C, Garcia-Ruiz C, and Fernández-Checa JC (2017) Redox Biol https://doi.org/10.1016/j.redox.2016.11.010
Vanier M, Millat G (2003) Clin Genet 64:269–281
Vázquez MC, Balboa E, Alvarez AR, and Zanlungo S (2012) Oxid Med Cell Longev https://doi.org/10.1155/2012/205713
Völkner C, Liedtke M, Untucht R, Hermann A, and Frech MJ (2021) Int J Mol Sci. https://doi.org/10.3390/ijms222212184
Völkner C, Pantoom S, Liedtke M, Lukas J, Hermann A, and Frech MJ Cells https://doi.org/10.3390/cells11030319
Wassif CA, Cross JL, Iben J, Sanchez-Pulido L, Cougnoux A, Platt FM, Ory DS, Ponting CP, Bailey-Wilson JE, Biesecker LG, Porter FD (2016) Genet Med 18:41–48
Wheeler S, Sillence DJ (2020) J Neurochem 153:674–692
Acknowledgements
We would like to thank the patients and their families, as well as the medical staff of the Medical Genetics Service – HCPA for logistic and technical support in the development of this study.
Funding
This work was supported by: Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/MEC-Brasil); Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/MCTI-Brasil number 430443/2018–8); Fundo de Incentivo à Pesquisa e Eventos (FIPE/HCPA – number 2018–0648).
Author information
Authors and Affiliations
Contributions
TGH and CRV contributed to the study conception and design. Material preparation, data collection, and analysis were performed by TGH, BD, MR, RK, and MBF. RG, DJM, and CRV supervised the work. The first draft of the manuscript was written by TGH and GBG, as well as revised by CRV. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
We performed this study in line with the principles of the Declaration of Helsinki and the project was approved by the Research Ethics Committee of the Hospital de Clínicas de Porto Alegre (CEP/HCPA) under number 2018–0648.
Consent to participate
Informed consent was obtained from all individual participants, or legal guardians, included in the study.
Competing interests
RG received speaker honoraria, travel grants, educational grants, and/or investigator fees from Actelion, Amicus, Armagen, BioMarin, Inventiva, JCR Pharmaceuticals, Lysogene, RegenxBio, Sangamo, Sanofi-Genzyme, Takeda, Sobi and Ultragenyx. All other authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hammerschmidt, T.G., Guerreiro, G.B., Donida, B. et al. Beneficial in vitro effect of N-acetylcysteine and coenzyme Q10 on DNA damage in neurodegenerative Niemann-Pick type C 1 disease: preliminary results. Naunyn-Schmiedeberg's Arch Pharmacol 396, 1563–1569 (2023). https://doi.org/10.1007/s00210-023-02423-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02423-7