Abstract
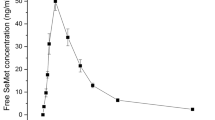
Chromium (Cr) is required for carbohydrate, lipid, and protein metabolisms in humans and animals. Cr insufficiency is associated with diabetes and cardiovascular disease. Chromium-enriched yeast (CrY) is a widely used Cr dietary supplement, but its pharmacokinetics remains unavailable. CrY was orally administered to rats at a single dose of 1 mg Cr/kg, and plasma Cr concentration at different time points was measured by inductively coupled plasma mass spectrometry. Pharmacokinetics of CrY in rats was well fitted to a non-compartmental model. Plasma Cr concentration reached the maximum of 8.68 ± 2.87 ng/mL at 0.25 h, and gradually decreased to 4.05 ± 0.47 ng/mL at 24 h. CrY was rapidly absorbed into the blood and was slowly eliminated after the oral administration, which could lead to the accumulation of Cr in vivo.

Similar content being viewed by others
Data availability
All the data generated or analyzed during this study are contained within the article.
References
Authority EFS (2009) Inability to assess the safety of chromium-enriched yeast added for nutritional purposes as a source of chromium in food supplements and the bioavailability of chromium from this source, based on the supporting dossiers. EFSA J 7(6):1083. https://doi.org/10.2903/j.efsa.2009.1083
Cheng HH, Lai MH, Hou WC, Huang CL (2004) Antioxidant effects of chromium supplementation with type 2 diabetes mellitus and euglycemic subjects. J Agric Food Chem 52(5):1385–1389. https://doi.org/10.1021/jf035074j
Chinese Nutrition Society (2014) Chinese dietary reference intakes 2013. Science Press, China
Collins BJ, Stout MD, Levine KE, Kissling GE, Melnick RL, Fennell TR, Walden R, Abdo K, Pritchard JB, Fernando RA, Burka LT, Hooth MJ (2010) Exposure to hexavalent chromium resulted in significantly higher tissue chromium burden compared with trivalent chromium following similar oral doses to male F344/N rats and female B6C3F1 mice. Toxicol Sci 118(2):368–379. https://doi.org/10.1093/toxsci/kfq263
Febel H, Szegedi B, Huszar S (2001) Absorption of inorganic, trivalent and hexavalent chromium following oral and intrajejunal doses in rats. Acta Vet Hung 49(2):203–209. https://doi.org/10.1556/004.49.2001.2.10
Feng W, Li Q, Wang W, Zhao T, Feng Y, Li F, Mao G, Chen Y, Ding Y, Yang L, Wu X (2018) Pharmacokinetics and bioavailability of chromium malate and its influence on trace metals absorption after oral or intravenous administration. Indian J Pharmacol 50(2):75–83. https://doi.org/10.4103/ijp.IJP_505_17
Food EPoFANSat (2010a) Scientific opinion on the safety of trivalent chromium as a nutrient added for nutritional purposes to foodstuffs for particular nutritional uses and foods intended for the general population (including food supplements). EFSA Journal 8 (12):1882. https://doi.org/10.2903/j.efsa.2010.1882
Food EPoFANSat (2010b) Scientific opinion on the safety of chromium picolinate as a source of chromium added for nutritional purposes to foodstuff for particular nutritional uses and to foods intended for the general population. EFSA Journal 8 (12):1883. https://doi.org/10.2903/j.efsa.2010.1883
Food EPoFaNSat (2012) Scientific opinion on ChromoPrecise® cellular bound chromium yeast added for nutritional purposes as a source of chromium in food supplements and the bioavailability of chromium from this source. EFSA Journal 10 (11):2951. https://doi.org/10.2903/j.efsa.2012.2951
Kerger BD, Paustenbach DJ, Corbett GE, Finley BL (1996) Absorption and elimination of trivalent and hexavalent chromium in humans following ingestion of a bolus dose in drinking water. Toxicol Appl Pharmacol 141(1):145–158. https://doi.org/10.1006/taap.1996.0271
Kottwitz K, Laschinsky N, Fischer R, Nielsen P (2009) Absorption, excretion and retention of 51Cr from labelled Cr-(III)-picolinate in rats. Biometals 22(2):289–295. https://doi.org/10.1007/s10534-008-9165-4
Laschinsky N, Kottwitz K, Freund B, Dresow B, Fischer R, Nielsen P (2012) Bioavailability of chromium (III)-supplements in rats and humans. Biometals 25(5):1051–1060. https://doi.org/10.1007/s10534-012-9571-5
O’Flaherty EJ (1993) A pharmacokinetic model for chromium. Toxicol Lett 68(1–2):145–158. https://doi.org/10.1016/0378-4274(93)90127-j
O’Flaherty EJ (1996) A physiologically based model of chromium kinetics in the rat. Toxicol Appl Pharmacol 138(1):54–64. https://doi.org/10.1006/taap.1996.0097
O’Flaherty EJ, Kerger BD, Hays SM, Paustenbach DJ (2001) A physiologically based model for the ingestion of chromium (III) and chromium (VI) by humans. Toxicol Sci 60(2):196–213. https://doi.org/10.1093/toxsci/60.2.196
Pechancová R, Pluháček T, Milde D (2019) Recent advances in chromium speciation in biological samples. Spectrochim Acta, Part B 152:109–122. https://doi.org/10.1016/j.sab.2018.12.008
Racek J, Trefil L, Rajdl D, Mudrová V, Hunter D, Senft V (2006) Influence of chromium-enriched yeast on blood glucose and insulin variables, blood lipids, and markers of oxidative stress in subjects with type 2 diabetes mellitus. Biol Trace Elem Res 109(3):215–230. https://doi.org/10.1385/bter:109:3:215
Racek J, Sindberg CD, Moesgaard S, Mainz J, Fabry J, Müller L, Rácová K (2013) Effect of chromium-enriched yeast on fasting plasma glucose, glycated haemoglobin and serum lipid levels in patients with type 2 diabetes mellitus treated with insulin. Biol Trace Elem Res 155(1):1–4. https://doi.org/10.1007/s12011-013-9758-9
Schlosser PM, Sasso AF (2014) A revised model of ex-vivo reduction of hexavalent chromium in human and rodent gastric juices. Toxicol Appl Pharmacol 280(2):352–361. https://doi.org/10.1016/j.taap.2014.08.010
Vincent JB (2007) The nutritional biochemistry of chromium (III) (2007). Elsevier, Netherlands
Zhang SQ, Chen GH, Lu WL, Zhang Q (2007) Effects on the bones of vanadyl acetylacetonate by oral administration: a comparison study in diabetic rats. J Bone Miner Metab 25(5):293–301. https://doi.org/10.1007/s00774-007-0759-7
Zhang SQ, Cheng SH, Shen S, Luo BY, Zhang Y (2021) Speciation analysis of chromium in chromium-enriched yeast by ion chromatography-inductively coupled plasma mass spectrometry. Biol Trace Elem Res 199(1):338–343. https://doi.org/10.1007/s12011-020-02149-0
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, and performance. The first draft of the manuscript was written by Shuang-Qing Zhang, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Ethics approval
All procedures were performed according to the Guide for the Care and Use of Laboratory Animals (NIH publication no. 85–23, eighth edition in 2011) and were approved by the Institutional Animal Care and Use Committee of the Hubei Provincial Key Laboratory of Yeast Function.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, SQ., Qin, XW. & Zhang, Y. Pharmacokinetics of chromium-enriched yeast in rats following oral administration. Naunyn-Schmiedeberg's Arch Pharmacol 396, 167–170 (2023). https://doi.org/10.1007/s00210-022-02334-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02334-z