Abstract
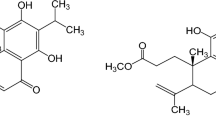
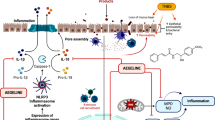
Chalcones present potential therapeutic activities reported on literature, which led us to evaluate the anti-inflammatory effects and the acute toxicity of 2′,6′-dihydroxy-4′-methoxydihydrochalcone (DHMDC) using in vitro and in vivo models. The anti-inflammatory activity was firstly in vitro investigated using macrophages (RAW 264.7) and neutrophils previously treated with DHMCD activated with lipopolysaccharide (LPS). Nitrite, IL-1β, and TNF levels were measured in the macrophage culture supernatant, and the adhesion molecule expression (CD62L, CD49D, and CD18) was evaluated in neutrophils. Then, carrageenan-induced inflammation was performed in the subcutaneous tissue of male Swiss mice. Leukocyte migration and histological analysis were performed in the pouches. Toxicological studies were carried out on female Swiss mice (600 mg/kg) through biochemical parameters and histopathological analysis. In vitro, the DHMCD significantly reduced the IL-1β, TNF, and nitrite levels. The DHMCD was also able to modulate the percentage of positive neutrophils for CD62L, without modifying the expression of CD18 or CD49d. In vivo, DHMCD (3 mg/kg, p.o.) significantly reduced neutrophil migration to inflammatory exudate and subcutaneous tissue. No evidence of toxic effect was observed considering the biochemical parameters and histopathological analysis of liver and kidney. Together, the obtained data shows that DHMCD presents anti-inflammatory activity by modulating the macrophage inflammatory protein secretion and also by blocking the CD62L cleavage in neutrophils. Furthermore, there was not any evidence of toxic effect in acute toxicological analysis.






Similar content being viewed by others
References
Bussmann AJC, Borghi SM, Zaninelli TH, dos Santos TS, Guazelli CFS, Fattori V, Domiciano TP, Pinho-Ribeiro FA, Ruiz-Miyazawa KW, Casella AMB, Vignoli JA, Camilios-Neto D, Casagrande R, Verri WA Jr (2019) The citrus flavanone naringenin attenuates zymosan-induced mouse joint inflammation: induction of Nrf2 expression in recruited CD45+ hematopoietic cells. Inflammopharmacology. 6:1229–1242. https://doi.org/10.1007/s10787-018-00561-6
Cartwright JA, Lucas CD, Rossi AG (2019) Inflammation resolution and the induction of granulocyte apoptosis by cyclin-dependent kinase inhibitor drugs. Front Pharmacol 10. https://doi.org/10.3389/fphar.2019.00055
Chen X, Yu W, Li W, Zhang H, Huang W, Wang J, Zhu W, Fang Q, Chen C, Li X, Liang G (2018) An anti-inflammatory chalcone derivative prevents heart and kidney from hyperlipidemia-induced injuries by attenuating inflammation. Toxicol Appl Pharmacol 338:43–53. https://doi.org/10.1016/j.taap.2017.11.003
Escribano-Ferrer E, Queralt Regué J, Garcia-Sala X, Boix Montañés A, Lamuela-Raventos RM (2019) In vivo anti-inflammatory and antiallergic activity of pure naringenin, naringenin chalcone, and quercetin in mice. J Nat Prod 82:177–182. https://doi.org/10.1021/acs.jnatprod.8b00366
Fathi MAA, El-Hafeezbcd AA, Abdelhamid D et al (2019) 1,3,4-Oxadiazole/chalcone hybrids: design, synthesis, and inhibition of leukemia cell growth and EGFR, Src, IL-6 and STAT3 activities. Bioorg Chem 84:150–163. https://doi.org/10.1016/j.bioorg.2018.11.032
Fu ZY, Jin QH, Qu YL, Guan LP (2019) Chalcone derivatives bearing chromen or benzo[f]chromen moieties: design, synthesis, and evaluations of anti-inflammatory, analgesic, selective COX-2 inhibitory activities. Bioorganic and Medicinal Chemistry Letters 29:1909–1912. https://doi.org/10.1016/j.bmcl.2019.05.051
Goodman SB, Pajarinen J, Yao Z, Lin T (2019) Inflammation and bone repair: from particle disease to tissue regeneration. Frontiers in Bioengineering and Biotechnology 7. https://doi.org/10.3389/fbioe.2019.00230
Grisham MB, Johnson GG, Lancaster JR (1996) Quantitation of nitrate and nitrite in extracellular fluids. Methods Enzymol 268:237–246. https://doi.org/10.1016/S0076-6879(96)68026-4
Herencia F, Ferrándiz ML, Ubeda A, Domínguez JN, Charris JE, Lobo GM, Alcaraz MJ (1998) Synthesis and anti-inflammatory activity of chalcone derivatives. Bioorganic and Medicinal Chemistry Letters. 8:1169–1174. https://doi.org/10.1016/S0960-894X(98)00179-6
Jain M, Parmar HS (2011) Evaluation of antioxidative and anti-inflammatory potential of hesperidin and naringin on the rat air pouch model of inflammation. Inflamm Res 60:483–491. https://doi.org/10.1007/s00011-010-0295-0
Karimi-Sales E, Alipour MR, Naderi R, Hosseinzadeh E, Ghiasi R (2019a) Protective effect of trans-chalcone against high-fat diet-induced pulmonary inflammation is associated with changes in miR-146a and pro-inflammatory cytokines expression in male rats. Inflammation. 42:2048–2055. https://doi.org/10.1007/s10753-019-01067-1
Karimi-Sales E, Ebrahimi-Kalan A, Alipour MR (2019b) Preventive effect of trans-chalcone on non-alcoholic steatohepatitis: improvement of hepatic lipid metabolism. Biomed Pharmacother 109:1306–1312. https://doi.org/10.1016/j.biopha.2018.10.196
Karimi-Sales E, Jeddi S, Ebrahimi-Kalan A, Alipour MR (2018) Trans-chalcone prevents insulin resistance and hepatic inflammation and also promotes hepatic cholesterol efflux in high-fat diet-fed rats: modulation of miR-34a-, miR-451-, and miR-33a-related pathways. Food Funct 9:4292–4298. https://doi.org/10.1039/c8fo00923f
Kawai T, Akira S (2010) The role of pattern-recognition receptors in innate immunity: update on toll-like receptors. Nat Immunol 11:373–384. https://doi.org/10.1038/ni.1863
Kong X, Wu G, Chen S, Zhang L, Li F, Shao T, Ren L, Chen SY, Zhang H, McClain CJ, Feng W (2019) Chalcone derivative L6H21 reduces EtOH + LPS-induced liver injury through inhibition of NLRP3 inflammasome activation. Alcohol Clin Exp Res 43:1662–1671. https://doi.org/10.1111/acer.14120
Ley K, Laudanna C, Cybulsky MI, Nourshargh S (2007) Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 7:678–689. https://doi.org/10.1038/nri2156
Lin WL, Chang CF, Shi CS, Shi GY, Wu HL (2013) Recombinant lectin-like domain of thrombomodulin suppresses vascular inflammation by reducing leukocyte recruitment via interacting with Lewis Y on endothelial cells. Arterioscler Thromb Vasc Biol 33:2366–2373. https://doi.org/10.1161/ATVBAHA.113.301221
Lin Y, Zhang M, Lu Q et al (2019) A novel chalcone derivative exerts anti-inflammatory and anti-oxidant effects after acute lung injury. Aging 11:7805–7816. https://doi.org/10.18632/aging.102288
Liu H, Liu L, Gao X, Liu Y, Xu W, He W, Jiang H, Tang J, Fan H, Xia X (2017) Novel ferulic amide derivatives with tertiary amine side chain as acetylcholinesterase and butyrylcholinesterase inhibitors: the influence of carbon spacer length, alkylamine and aromatic group. Eur J Med Chem 126:810–822. https://doi.org/10.1016/j.ejmech.2016.12.003
Liu X, Ai F, Li H, Xu Q, Mei L, Miao J, Wen Q, Zhang C, Zhang S, Zhou J, Chen X, Chu C, Guo J (2019) Anti-inflammatory effects of Shenfu injection against acute lung injury through inhibiting HMGB1-NF-B pathway in a rat model of endotoxin shock. Evid Based Complement Alternat Med 2019:1–10. https://doi.org/10.1155/2019/9857683
Lyck R, Enzmann G (2015) The physiological roles of ICAM-1 and ICAM-2 in neutrophil migration into tissues. Curr Opin Hematol 1:53–59. https://doi.org/10.1097/MOH.0000000000000103
Machado ID, Santin JR, Drewes CC, Gil CD, Oliani SM, Perretti M, Farsky SH (2017) Alterations in the profile of blood neutrophil membrane receptors caused by in vivo adrenocorticotrophic hormone actions. Am J Physiol Endocrinol Metab 307:E754–E763. https://doi.org/10.1152/ajpendo.00227.2014
Mahapatra DK, Bharti SK, Asati V (2017) Chalcone derivatives: anti-inflammatory potential and molecular targets perspectives. Curr Top Med Chem 17:3146–3169. https://doi.org/10.2174/1568026617666170914160446
Motta VT, (2009) Bioquímica clinica para o laboratorio: principios e interpretaçoes. Rio de Janeiro RJ
Naegelen I, Beaume N, Plançon S et al (2015) Regulation of neutrophil degranulation and cytokine secretion: a novel model approach based on linear fitting. Journal of Immunology Research. https://doi.org/10.1155/2015/817038
Nunes R, Broering MF, De Faveri R et al (2019) Effect of the metanolic extract from the leaves of Garcinia humilis Vahl (Clusiaceae) on acute inflammation. Inflammopharmacology. https://doi.org/10.1007/s10787-019-00645-x
OECD (2001) OECD guideline for testing of chemicals. Acute oral toxicity—fixed dose procedure. https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/oecd/oecd_gl420.pdf Accessed 10 May 2020
Pinho-Ribeiro FA, Hohmann MS, Borghi SM et al (2015) Protective effects of the flavonoid hesperidin methyl chalcone in inflammation and pain in mice: role of TRPV1, oxidative stress, cytokines and NF-κB. Chem Biol Interact 228:88–99. https://doi.org/10.1016/j.cbi.2015.01.011
Rasquel-Oliveira FS, Manchope MF, Staurengo-Ferrari L et al (2020) Hesperidin methyl chalcone interacts with NFκB Ser276 and inhibits zymosan-induced joint pain and inflammation, and RAW 264.7 macrophage activation. Inflammopharmacology. DOI: https://doi.org/10.1007/s10787-020-00686-7
Sameem B, Saeedi M, Mahdavi M, Shafiee A (2017) A review on tacrine-based scaffolds as multi-target drugs (MTDLs) for Alzheimer’s disease. Eur J Med Chem 128:332–345. https://doi.org/10.1016/j.ejmech.2016.10.060
Sasidharan R, Sreedharannair Leelabaiamma M, Mohanan R, Jose SP, Mathew B, Sukumaran S (2019) Anti-inflammatory effect of synthesized indole-based chalcone (2E)-3-(4-bromophenyl)-1-(1H-indol-3-yl) prop-2-en-1-one: an in vitro and in vivo studies. Immunopharmacol Immunotoxicol 41:568–576. https://doi.org/10.1080/08923973.2019.1672177
Sieve I, Ricke-Hoch M, Kasten M, Battmer K, Stapel B, Falk CS, Leisegang MS, Haverich A, Scherr M, Hilfiker-Kleiner D (2018) A positive feedback loop between IL-1β LPS and NEU1 may promote atherosclerosis by enhancing a pro-inflammatory state in monocytes and macrophages. Vasc Pharmacol 103-105:16–28. https://doi.org/10.1016/j.vph.2018.01.005
Sin YM, Sedgwick AD, Chea EP, Willoughby DA (1986) Mast cells in newly formed lining tissue during acute inflammation: a six day air pouch model in the mouse. Ann Rheum Dis 45:873–877. https://doi.org/10.1136/ard.45.10.873
Singh H, Sidhu S, Chopra K, Khan MU (2015) Hepatoprotective effect of trans-chalcone on experimentally induced hepatic injury in rats: inhibition of hepatic inflammation and fibrosis. Can J Physiol Pharmacol 94:879–887. https://doi.org/10.1139/cjpp-2016-0071
Staurengo-Ferrari L, Ruiz-Miyazawa KW, Pinho-Ribeiro FA, Fattori V, Zaninelli TH, Badaro-Garcia S, Borghi SM, Carvalho TT, Alves-Filho JC, Cunha TM, Cunha FQ, Casagrande R, Verri WA Jr (2018) Trans-chalcone attenuates pain and inflammation in experimental acute gout arthritis in mice. Front Pharmacol 9. https://doi.org/10.3389/fphar.2018.01123
Tucureanu MM, Rebleanu D, Constantinescu AC et al (2018) Lipopolysaccharide-induced inflammation in monocytes/macrophages is blocked by liposomal delivery of Gi-protein inhibitor. Int J Nanomedicine 13:63–76. https://doi.org/10.2147/IJN.S150918
Vijaya Bhaskar Reddy M, Hung HY, Kuo PC, Huang GJ, Chan YY, Huang SC, Wu SJ, Morris-Natschke SL, Lee KH, Wu TS (2017) Synthesis and biological evaluation of chalcone, dihydrochalcone, and 1,3-diarylpropane analogs as anti-inflammatory agents. Bioorganic and Medicinal Chemistry Letters. 27:1547–1550. https://doi.org/10.1016/j.bmcl.2017.02.038
Yan M, Zhang X, Chen A, Gu W, Liu J, Ren X, Zhang J, Wu X, Place AT, Minshall RD, Liu G (2017) Endothelial cell SHP-2 negatively regulates neutrophil adhesion and promotes transmigration by enhancing ICAM-1-VE-cadherin interaction. FASEB J 31:4759–4769. https://doi.org/10.1096/fj.201700280R
Yang SC, Wang YH, Tsai YF, Chang YW, Wu TS, Ho CM, Hwang TL (2020) A synthesized heterocyclic chalcone inhibits neutrophilic inflammation through K+-dependent pH regulation. FASEB J 34:7127–7143. https://doi.org/10.1096/fj.201903123R
Yang T, Wang H, Li Y, Zeng Z, Shen Y, Wan C, Wu Y, Dong J, Chen L, Wen F (2019) Serotonin receptors 5-HTR2A and 5-HTR2B are involved in cigarette smoke-induced airway inflammation, mucus hypersecretion and airway remodeling in mice. Int Immunopharmacol 81:106036. https://doi.org/10.1016/j.intimp.2019.106036
Zeni ALB, De Albuquerque CAC, Gonçalves F et al (2013) Phytochemical profile, toxicity and antioxidant activity of Aloysia gratissima (Verbenaceae). Quim Nova 36:69–73. https://doi.org/10.1590/S0100-40422013000100013
Zermiani T, Santos MC, Lobato FM, Blödorn VB, Andrade FSM, Lacava LC, Inoue TY, Ferreira RA, Guimarães EF, Machado MS, Lucinda-Silva RM, Malheiros  (2019) Morphological and phytochemical characterization of Piper mosenii. Nat Prod Commun 14:1934578X1901400. https://doi.org/10.1177/1934578X1901400118
Zhou X, Gao XP, Fan J, Liu Q, Anwar KN, Frey RS, Malik AB (2005) LPS activation of toll-like receptor 4 signals CD11b/CD18 expression in neutrophils. Am J Physiol Lung Cell Mol Physiol 288:655–L662. https://doi.org/10.1152/ajplung.00327.2004
Acknowledgments
This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, 429505/2018-3), the Coordenação de Aperfeiçoamento Pessoal de Nível Superior (CAPES), and the Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (FAPESC), M.L.C., L.B., R.N., and S.R.S were post-graduate students in Pharmaceutical Science and are recipients of CAPES grants (Financial code 001) and L.L. was a graduate student in Pharmacy recipient of CNPq (PIBIC) grant (Financial code 001). N.L.M.Q. is a researcher granted by CNPq (305550/2018-7).
Author information
Authors and Affiliations
Contributions
J.R.S. and N.L.M.Q. conceived and designed the research. M.L.C., L.B., R.N., S.R.S., S.C.B., and L.C. conducted experiments. A.M. and M.M.S. contributed new reagents or analytical tools. J.R.S. and M.L.C. analyzed data. J.R.S., M.L.C., and N.L.M.Q. wrote the manuscript. All authors read and approved the manuscript and all data was generated in-house and that no paper mill was used.
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Cerutti, M.L., Benvenutti, L., Nunes, R. et al. Effects of 2′,6′-dihydroxy-4′-methoxydihidrochalcone on innate inflammatory response. Naunyn-Schmiedeberg's Arch Pharmacol 393, 2061–2072 (2020). https://doi.org/10.1007/s00210-020-01922-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01922-1