Abstract
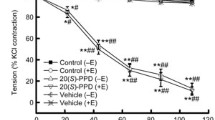
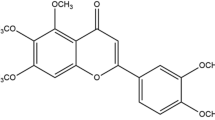
This study was designed to investigate the vasorelaxant effects and underlying mechanism of isocorynoxeine (ICN), one of the indole alkaloids from Uncaria hooks, on isolated mesenteric arteries in vitro. The myograph system was applied for isometric tension recording in the vascular rings. ICN relaxed both endothelium-intact and endothelium-denuded rat vascular rings precontracted with phenylephrine or KCl in a dose-dependent manner. Propranolol, tetraethylammonium, BaCl2, and glibenclamide had no influence on the vasodilator effect of ICN on phenylephrine-primed vascular rings, while 4-aminopyridine decreased the maximum relaxation. Furthermore, ICN produced a significant drop in maximum response in the PE log concentration-response curve without shifting to the right. In the Ca2+-free Kreb’s-Henseleit buffer, ICN inhibited the contraction in vascular rings evoked by PE, but not by KCl. The phasic contractions of segments in the Ca2+-free Kreb’s-Henseleit buffer induced by CaCl2 were restrained by ICN, while contractions elicited by caffeine displayed no differences. Furthermore, the phasic vasodilation of ICN was significantly lower than controls when pretreated with nifedipine and heparin. Both BAYK8644- and PE-evoked responses were significantly inhibited in the presence of 100 μM of ICN in human vascular smooth muscle cells loaded with the fluorescent Ca2+ indicator Fluo-4-AM. All these results suggest that ICN act in an endothelium-independent manner on the mesenteric artery. Its mechanisms of vasorelaxant action were produced by the inhibition of L-type calcium channel-mediated external Ca2+ influx and α1A-adrenoceptor-mediated intracellular Ca2+ release in vascular smooth muscle cells, and the participation of the Kv channel.






Similar content being viewed by others
References
Bai R, Wei Z, Liu J, Xie W, Yao H, Wu X, Jiang J, Wang Q, Xu J (2012) Synthesis and biological evaluation of 4′-[(benzimidazole-1-yl) methyl] biphenyl-2-sulfonamide derivatives as dual angiotensin II/endothelin A receptor antagonists. Bioorg Med Chem 20(15):4661–4667. https://doi.org/10.1016/j.bmc.2012.06.011
Baltoumas FA, Theodoropoulou MC, Hamodrakas SJ (2013) Interactions of the α-subunits of heterotrimeric G-proteins with GPCRs, effectors and RGS proteins: a critical review and analysis of interacting surfaces, conformational shifts, structural diversity and electrostatic potentials. J Struct Biol 182(3):209–218. https://doi.org/10.1016/j.jsb.2013.03.004
Berridge MJ (1993) Inositol trisphosphate and calcium signalling. Nature 361(6410):315–325. https://doi.org/10.1111/j.1749-6632.1995.tb26646
Carretero OA, Oparil S (2000) Essential hypertension. Circulation 101(3):329–335. https://doi.org/10.1161/01.CIR.101.3.329
Chen YP, Lu MN, Hao JC, Li MH, Hattori M, Wang W (2015) Metabolites of isocorynoxeine in rats after its oral administration. J Asian Nat Prod Res 17(4):384–390. https://doi.org/10.1080/10286020.2014.1003182
Cheng JT (2000) Drug therapy in Chinese traditional medicine. J Clin Pharmacol 40(5):445–450. https://doi.org/10.1177/00912700022009198
Chou CH, Gong CL, Chao CC, Lin CH, Kwan CY, Hsieh CL, Leung YM (2009) Rhynchophylline from Uncaria rhynchophylla functionally turns delayed rectifiers into A-Type K+ channels. J Nat Prod 72(5):830–834. https://doi.org/10.1021/np800729q
Crowley CM, Lee CH, Gin SA, Keep AM, Cook RC, van Breemen C (2002) The mechanism of excitation-contraction coupling in phenylephrine-stimulated human saphenous vein. Am J Physiol Heart Circ Physiol 283(4):1271–1281. https://doi.org/10.1152/ajpheart.01129.2001
Ghosh TK, Eis PS, Mullaney JM, Ebert CL, Gill DL (1988) Competitive, reversible, and potent antagonism of inositol 1, 4, 5-trisphosphate-activated calcium release by heparin. J Biol Chem 263(23):11075–11079
Ha KT, Lee TK, Kwak KH, Kim JK, Kim DI, Choi DY, Kim CH (2004) Inhibitory effect of Cho-Deung-San on human aortic smooth muscle cell migration induced by TNF-α through inhibition of matrix metalloproteinase-2 and-9 activity. Vasc Pharmacol 41(3):83–90. https://doi.org/10.1016/j.vph.2004.05.003
Habib GB, Basra SS (2014) Are there any new pharmacologic therapies on the horizon to better treat hypertension? A state-of-the-art paper. J Cardiovasc Pharmacol Ther 19(6):516–525. https://doi.org/10.1177/1074248414529620
Hanus JS, Amboni G, Rosa MI, Ceretta LB, Tuon L (2015) The quality and characteristics of sleep of hypertensive patients. Rev Esc Enferm U S P 49(4):0596–0602. https://doi.org/10.1590/S0080-623420150000400009
He J, Han S, Yang F, Zhou N, Wang S (2012) Prostate cell membrane chromatography–liquid chromatography–mass spectrometry for screening of active constituents from Uncaria rhynchophylla. J Chromatogr Sci 51(10):905–909. https://doi.org/10.1093/chromsci/bms188
Huang ZQ, Liu JL, Wang YY, Ling YP, Li D (2008) A study on the chemical change in the decoction of Uncaria hook. J Northwest 38(5):787–789
Kang TH, Murakami Y, Takayama H, Kitajima M, Aimi N, Watanabe H, Matsumoto K (2004) Protective effect of rhynchophylline and isorhynchophylline on in vitro ischemia-induced neuronal damage in the hippocampus: putative neurotransmitter receptors involved in their action. Life Sci 76(3):331–343. https://doi.org/10.1016/j.lfs.2004.08.012
Keji C, Hao X (2003) The integration of traditional Chinese medicine and Western medicine. Eur Rev 11(2):225–235. https://doi.org/10.1017/S106279870300022X
Kushida H, Fukutake M, Tabuchi M, Katsuhara T, Nishimura H, Ikarashi Y, Kanitani M, Kase Y (2013) Simultaneous quantitative analyses of indole and oxindole alkaloids of Uncaria Hook in rat plasma and brain after oral administration of the traditional Japanese medicine Yokukansan using high-performance liquid chromatography with tandem mass spectrometry. Biomed Chromatogr 27(12):1647–1656. https://doi.org/10.1002/bmc.2974
Laus G, Keplinger K (2003) Alkaloids of peruvian Uncaria guianensis (Rubiaceae). Phyton 43(1):1–8
Laus G, Teppner H (1996) Alkaloids of an Uncaria rhynchophylla (Rubiaceae-Coptosapelteae). Phyton 36:185–196
Li YJ, Bao JX, Xu JW, Murad F, Bian K (2010) Vascular dilation by paeonol—a mechanism study. Vasc Pharmacol 53(3):169–176. https://doi.org/10.1016/j.vph.2010.07.001
Li PY, Zeng XR, Cheng J, Wen J, Inoue I, Yang Y (2013) Rhynchophylline-induced vasodilation in human mesenteric artery is mainly due to blockage of L-type calcium channels in vascular smooth muscle cells. Naunyn Schmiedeberg’s Arch Pharmacol 386(11):973–982. https://doi.org/10.1007/s00210-013-0888-6
Liu SH, Zeng XR, Yin L (2007) The effect of rhynchophylline on large-conductance calcium-activated potassium channels of human mesenteric artery smooth muscle cells. Sichuan J Physiol Sci 29(3):97–99
Martini LC, Garcezdocarmo L, Jurkiewicz A (2010) Functional classification of non-competitive antagonism in vas deferens. Ann N Y Acad Sci 812(1):196–199. https://doi.org/10.1111/j.1749-6632.1997.tb48172.x
Metzler M, Pfeiffer E, Schulz SI, Dempe JS (2013) Curcumin uptake and metabolism. Biofactors 39(1):14–20. https://doi.org/10.1002/biof.1042
Qian LB, Wang HP, Qiu WL, Huang H, Bruce IC, Xia Q (2006) Interleukin-2 protects against endothelial dysfunction induced by high glucose levels in rats. Vasc Pharmacol 45(6):374–382. https://doi.org/10.1016/j.vph.2006.06.002
Ridsdale CE (1978) A revision of Mitragyna and Uncaria (Rubiaceae). Blumea 24(1):43–100
Sanae F, Komatsu Y, Amagaya S, Chisaki K, Hayashi H (2000) Effects of 9 Kampo medicines clinically used in hypertension on hemodynamic changes induced by theophylline in rats. Biol Pharm Bull 23(6):762–765. https://doi.org/10.1248/bpb.23.762
Schild HO (1954) Non-competitive drug antagonism. J Physiol 124(2):33–34
Shimada Y, Yang Q, Yokoyama K, Goto H, Kasahara Y, Sekiya N, Hikiami H, Terasawa K (2003) Choto-san prevents occurrence of stroke and prolongs life span in stroke-prone spontaneously hypertensive rats. Am J Chin Med 31(01):79–85. https://doi.org/10.1142/S0192415X03000837
Song CQ, Fan Y, Huang WH (2003) Different hypotensive effects of various active constituents isolated from Uncaria rhynchophylla. Chin Tradit Herb Drugs 31(10):762–764
Tao SU, Rui LI, Cao YX (2011) Vasorelaxant and antihypertensive effects of formononetin through endothelium-dependent and -independent mechanisms. Acta Pharmacol Sin 32(8):1009–1018. https://doi.org/10.1038/aps.2011.51
Wu YT, Lin LC, Tsai TH (2014) Determination of rhynchophylline and hirsutine in rat plasma by UPLC-MS/MS after oral administration of Uncaria rhynchophylla extract. Biomed Chromatogr 28(3):439–445. https://doi.org/10.1002/bmc.3052
Yang ZD, Duan DZ, Du J, Yang MJ, Li S, Yao XJ (2012) Geissoschizine methyl ether, a corynanthean-type indole alkaloid from Uncaria rhynchophylla as a potential acetylcholinesterase inhibitor. Nat Prod Res 26(1):22–28. https://doi.org/10.1080/14786419.2010.529811
Zhang W, Liu GX, Huang XN (1987) Effect of rhynchophylline on the contraction of rabbit aorta. Acta Pharmacol Sin 8(5):425–429
Zhang WB, Chen CX, Sim SM, Kwan CY (2004) In vitro vasodilator mechanisms of the indole alkaloids rhynchophylline and isorhynchophylline, isolated from the hook of Uncaria rhynchophylla (Miquel). Naunyn Schmiedeberg's Arch Pharmacol 369(2):232–238. https://doi.org/10.1007/s00210-003-0854-9
Zhang YN, Yang YF, Xu W, Yang XW (2017) The blood-brain barrier permeability of six indole alkaloids from Uncariae Ramulus Cum Uncis in the MDCK-pHaMDR cell monolayer model. Molecules 22(11):1944. https://doi.org/10.3390/molecules22111944
Zhao F, Li J, Chen Y, Tian Y, Wu C, Xie Y, Zhou Y, Wang J, Xie X, Liu H (2016) Design, synthesis, and biological evaluation of indoline and Indole derivatives as potent and selective α1A-adrenoceptor antagonists. J Med Chem 59(8):3826–3839. https://doi.org/10.1021/acs.jmedchem.5b02023
Zheng JN, Wang DY (2009) Study on alkaloid constituents in Uncaria Macrophylla. Guiding J Trad Chin Med Pharm 15(1):80–86
Zhou J, Zhou S (2010) Antihypertensive and neuroprotective activities of rhynchophylline: the role of rhynchophylline in neurotransmission and ion channel activity. J Ethnopharmacol 132(1):15–27. https://doi.org/10.1016/j.jep.2010.08.041
Zhou JY, Zhou SW (2012) Isorhynchophylline: a plant alkaloid with therapeutic potential for cardiovascular and central nervous system diseases. Fitoterapia 83(4):617–626. https://doi.org/10.1016/j.fitote.2012.02.010
Acknowledgements
We gratefully acknowledge the National Natural Science Foundation of China (No. 81673398) for financial support.
Author contribution statement
SW and TL conceived of or designed study. TL and KX performed research. TL and NJ analyzed data. ZH and DC contributed new reagents and analytical tools. TL wrote the paper. All authors read and approved the manuscript.
Funding
This study was funded by National Natural Science Foundation of China (No. 81673398).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the Ethical Committee of Xi’an Jiaotong University.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Supplementary Fig 7
The LC/MS-IT-TOF MS spectra of ICN (GIF 8 kb)
Rights and permissions
About this article
Cite this article
Li, T., Xu, K., Che, D. et al. Endothelium-independent vasodilator effect of isocorynoxeine in vitro isolated from the hook of Uncaria rhynchophylla (Miquel). Naunyn-Schmiedeberg's Arch Pharmacol 391, 1285–1293 (2018). https://doi.org/10.1007/s00210-018-1536-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1536-y