Abstract
Herein, it was investigated whether a complex of lidocaine with 2-hydroxypropyl-β-cyclodextrin (HP-β-CD) would present a better antinociceptive profile in vivo when compared with plain lidocaine in models of orofacial pain. Plain lidocaine (LDC) and complexed lidocaine (LDC:HP-β-CD) were initially evaluated in vitro to determine the release rate of the two formulations. Subsequently, the effect of both formulations was evaluated in independent groups of rats submitted to the orofacial formalin test, induction of facial heat hyperalgesia by capsaicin and carrageenan, and induction of facial heat and mechanical hyperalgesia by constriction of the infraorbital nerve. LDC:HP-β-CD led to a reduction in the lidocaine release assessed in the in vitro release assay compared to plain LDC. Both formulations presented an antinociceptive effect in all models, but LDC:HP-β-CD showed a better effect in the second phase of the formalin response, in carrageenan-induced heat hyperalgesia, and in the heat hyperalgesia associated to infraorbital nerve constriction. Our results show that complexation improved in vivo antinociceptive effects of LDC, but further studies are necessary to elucidate what properties contribute to the better effect of the complexed formulation on this models and/or what characteristics of the pain model facilitate the action of the complexed formulation.




Similar content being viewed by others
References
Almeida RT, Duarte ID (2008) Nitric oxide/cGMP pathway mediates orofacial antinociception induced by electroacupuncture at the St36 acupoint. Brain Res 1188:54–60
Anirudhan TS, Nair SS, Nair AS (2016) Fabrication of a bioadhesive transdermal device from chitosan and hyaluronic acid for the controlled release of lidocaine. Carbohydr Polym 152:687–698
Araujo DR, Braga Ade F, Moraes CM, Fraceto LF, Paula E (2006) Complexation of 50% enantiomeric excess (S75-R25) bupivacaine with cyclodextrins and spinal block anesthesia in rats. Rev Bras Anestesiol 56:495–506
Araya EI, Nones CFM, Ferreira LEN, Kopruszinski CM, Cunha JMD, Chichorro JG (2017) Role of peripheral and central TRPV1 receptors in facial heat hyperalgesia in streptozotocin-induced diabetic rats. Brain Res 1670:146–155
Becker DE, Reed KL (2012) Local anesthetics: review of pharmacological considerations. Anesth Prog 59:90–101 quiz 102-103
Berk T, Silberstein SD (2018) The use and method of action of intravenous lidocaine and its metabolite in headache disorders. Headache 58:783–789
Braga MA, Martini MF, Pickholz M, Yokaichiya F, Franco MK, Cabeca LF, Guilherme VA, Silva CM, Limia CE, de Paula E (2016) Clonidine complexation with hydroxypropyl-beta-cyclodextrin: from physico-chemical characterization to in vivo adjuvant effect in local anesthesia. J Pharm Biomed Anal 119:27–36
Brederson JD, Kym PR, Szallasi A (2013) Targeting TRP channels for pain relief. Eur J Pharmacol 716:61–76
Casale R, Symeonidou Z, Bartolo M (2017) Topical treatments for localized neuropathic pain. Curr Pain Headache Rep 21:15
Cereda CM, Brunetto GB, de Araujo DR, de Paula E (2006) Liposomal formulations of prilocaine, lidocaine and mepivacaine prolong analgesic duration. Can J Anaesth 53:1092–1097
Chichorro JG, Lorenzetti BB, Zampronio AR (2004) Involvement of bradykinin, cytokines, sympathetic amines and prostaglandins in formalin-induced orofacial nociception in rats. Br J Pharmacol 141:1175–1184
Chichorro JG, Zampronio AR, Rae GA (2006) Endothelin ET(B) receptor antagonist reduces mechanical allodynia in rats with trigeminal neuropathic pain. Exp Biol Med (Maywood) 231:1136–1140
Chichorro JG, Zampronio AR, Cabrini DA, Franco CR, Rae GA (2009) Mechanisms operated by endothelin ETA and ETB receptors in the trigeminal ganglion contribute to orofacial thermal hyperalgesia induced by infraorbital nerve constriction in rats. Neuropeptides 43:133–142
Clavelou P, Pajot J, Dallel R, Raboisson P (1989) Application of the formalin test to the study of orofacial pain in the rat. Neurosci Lett 103:349–353
Dach F, Eckeli AL, Ferreira Kdos S, Speciali JG (2015) Nerve block for the treatment of headaches and cranial neuralgias - a practical approach. Headache 55(Suppl 1):59–71
Dale GJ, Phillips S, Falk GL (2016) The analgesic efficacy of intravenous lidocaine infusion after laparoscopic fundoplication: a prospective, randomized, double-blind, placebo-controlled trial. Local Reg Anesth 9:87–93
Dallel R, Raboisson P, Clavelou P, Saade M, Woda A (1995) Evidence for a peripheral origin of the tonic nociceptive response to subcutaneous formalin. Pain 61:11–16
de Araujo DR, Tsuneda SS, Cereda CM, Del GFCF, Prete PS, Fernandes SA, Yokaichiya F, Franco MK, Mazzaro I, Fraceto LF, de FABA, de Paula E (2008) Development and pharmacological evaluation of ropivacaine-2-hydroxypropyl-beta-cyclodextrin inclusion complex. Eur J Pharm Sci 33:60–71
de Paula E, Cereda CM, Tofoli GR, Franz-Montan M, Fraceto LF, de Araujo DR (2010) Drug delivery systems for local anesthetics. Recent Pat Drug Deliv Formul 4:23–34
Derry S, Wiffen PJ, Kalso EA, Bell RF, Aldington D, Phillips T, Gaskell H, Moore RA (2017) Topical analgesics for acute and chronic pain in adults - an overview of Cochrane Reviews. Cochrane Database Syst Rev 5:CD008609
Di Stani F, Ojango C, Dugoni D, Di Lorenzo L, Masala S, Delfini R, Bruti G, Simonetti G, Piovesan EJ, Ruggeri AG (2015) Combination of pharmacotherapy and lidocaine analgesic block of the peripheral trigeminal branches for trigeminal neuralgia: a pilot study. Arq Neuropsiquiatr 73:660–664
Dollo G, Thompson DO, Le Corre P, Chevanne F, Le Verge R (1998) Inclusion complexation of amide-typed local anesthetics with b-cyclodextrin and its derivatives. III. Biopharmaceutics of bupivacaine-SBE7-bCD complex following percutaneous sciatic nerve administration in rabbits. Int J Pharm 164:11–19
Ferreira LE, Muniz BV, Dos Santos CP, Volpato MC, de Paula E, Groppo FC (2016) Comparison of liposomal and 2-hydroxypropyl-beta-cyclodextrin-lidocaine on cell viability and inflammatory response in human keratinocytes and gingival fibroblasts. J Pharm Pharmacol 68:791–802
Ferreira LEN, Antunes GBM, Muniz BV, Burga-Sanchez J, de Melo NFS, Groppo FC, Fraceto LF, Volpato MC (2018) Effects of lidocaine and the inclusion complex with 2-hydroxypropyl-beta-cyclodextrin on cell viability and proliferation of oral squamous cell carcinoma. J Pharm Pharmacol 70:874–882
Fletcher D, Kayser V, Guilbaud G (1996) Influence of timing of administration on the analgesic effect of bupivacaine infiltration in carrageenin-injected rats. Anesthesiology 84:1129–1137
Franco de Lima RA, de Jesus MB, Saia Cereda CM, Tofoli GR, Cabeca LF, Mazzaro I, Fraceto LF, de Paula E (2012) Improvement of tetracaine antinociceptive effect by inclusion in cyclodextrins. J Drug Target 20:85–96
Franz-Montan M, Baroni D, Brunetto G, Sobral VR, da Silva CM, Venancio P, Zago PW, Cereda CM, Volpato MC, de Araujo DR, de Paula E, Groppo FC (2015) Liposomal lidocaine gel for topical use at the oral mucosa: characterization, in vitro assays and in vivo anesthetic efficacy in humans. J Liposome Res 25:11–19
Gambeta E, Kopruszinski CM, Dos Reis RC, Zanoveli JM, Chichorro JG (2016) Evaluation of heat hyperalgesia and anxiety like-behaviors in a rat model of orofacial cancer. Neurosci Lett 619:100–105
Giordano D, Raso MG, Pernice C, Agnoletti V, Barbieri V (2015) Topical local anesthesia: focus on lidocaine-tetracaine combination. Local Reg Anesth 8:95–100
Gould S, Scott RC (2005) 2-Hydroxypropyl-beta-cyclodextrin (HP-beta-CD): a toxicology review. Food Chem Toxicol 43:1451–1459
Hao JX, Yu W, Xu XJ, Wiesenfeld-Hallin Z (1996) Capsaicin-sensitive afferents mediate chronic cold, but not mechanical, allodynia-like behavior in spinally injured rats. Brain Res 722:177–180
Huang J, Zhang X, McNaughton PA (2006) Modulation of temperature-sensitive TRP channels. Semin Cell Dev Biol 17:638–645
Hummig W, Kopruszinski CM, Chichorro JG (2014) Pregabalin reduces acute inflammatory and persistent pain associated with nerve injury and cancer in rat models of orofacial pain. J Oral Facial Pain Headache 28:350–359
Jambhekar SS, Breen P (2016a) Cyclodextrins in pharmaceutical formulations I: structure and physicochemical properties, formation of complexes, and types of complex. Drug Discov Today 21:356–362
Jambhekar SS, Breen P (2016b) Cyclodextrins in pharmaceutical formulations II: solubilization, binding constant, and complexation efficiency. Drug Discov Today 21:363–368
Joo JD, Choi JW, In JH, Jung HS, Lee JA, Kim YS, Kim DW, Yeom JH, Shin EY, Jeon YS (2011) Lidocaine suppresses the increased extracellular signal-regulated kinase/cyclic AMP response element-binding protein pathway and pro-inflammatory cytokines in a neuropathic pain model of rats. Eur J Anaesthesiol 28:106–111
Kayser V, Guilbaud G (1987) Local and remote modifications of nociceptive sensitivity during carrageenin-induced inflammation in the rat. Pain 28:99–107
Kissin I, Lee SS, Bradley EL Jr (1998) Effect of prolonged nerve block on inflammatory hyperalgesia in rats: prevention of late hyperalgesia. Anesthesiology 88:224–232
Lang A, Ben Horin S, Picard O, Fudim E, Amariglio N, Chowers Y (2010) Lidocaine inhibits epithelial chemokine secretion via inhibition of nuclear factor kappa B activation. Immunobiology 215:304–313
Loftsson T, Duchene D (2007) Cyclodextrins and their pharmaceutical applications. Int J Pharm 329:1–11
Loftsson T, Masson M (2001) Cyclodextrins in topical drug formulations: theory and practice. Int J Pharm 225:15–30
Menezes Pdos P, Araujo AA, Doria GA, Quintans-Junior LJ, de Oliveira MG, dos Santos MR, de Oliveira JF, Matos Jdo R, Carvalho FM, Alves PB, de Matos IL, dos Santos DA, Marreto RN, da Silva GF, Serafini MR (2015) Physicochemical characterization and analgesic effect of inclusion complexes of essential oil from Hyptis pectinata L. Poit leaves with beta-cyclodextrin. Curr Pharm Biotechnol 16:440–450
Moraes CM, Abrami P, Araujo DRD, Braga AFA, Issa MG, Ferraz HG, Paula E, Fraceto LF (2007) Characterization of lidocaine:hydroxypropyl-b-cyclodextrininclusion complex. J Incl Phenom Macrocycl Chem 57:313–316
Motta EM, Chichorro JG, Rae GA (2009) Role of ET(A) and ET(B) endothelin receptors on endothelin-1-induced potentiation of nociceptive and thermal hyperalgesic responses evoked by capsaicin in rats. Neurosci Lett 457:146–150
Ni J, Wang X, Tang Y, Yang L, Zeng Y, Guo Y (2017) Subcutaneous injection of triamcinolone and lidocaine to prevent postherpetic neuralgia. Pain Physician 20:397–403
Ortiz MP, Godoy MC, Schlosser RS, Ortiz RP, Godoy JP, Santiago ES, Rigo FK, Beck V, Duarte T, Duarte MM, Menezes MS (2016) Effect of endovenous lidocaine on analgesia and serum cytokines: double-blinded and randomized trial. J Clin Anesth 35:70–77
Pedersen JL, Crawford ME, Dahl JB, Brennum J, Kehlet H (1996) Effect of preemptive nerve block on inflammation and hyperalgesia after human thermal injury. Anesthesiology 84:1020–1026
Pelissier T, Pajot J, Dallel R (2002) The orofacial capsaicin test in rats: effects of different capsaicin concentrations and morphine. Pain 96:81–87
Pinto LM, Fraceto LF, Santana MH, Pertinhez TA, Junior SO, de Paula E (2005) Physico-chemical characterization of benzocaine-beta-cyclodextrin inclusion complexes. J Pharm Biomed Anal 39:956–963
Prado AR, Yokaichiya F, Franco M, Silva C, Oliveira-Nascimento L, Franz-Montan M, Volpato MC, Cabeca LF, de Paula E (2017) Complexation of oxethazaine with 2-hydroxypropyl-beta-cyclodextrin: increased drug solubility, decreased cytotoxicity and analgesia at inflamed tissues. J Pharm Pharmacol 69:652–662
Provinciali L, Lattanzi S, Chiarlone R, Fogliardi A, Intelligente F, Irace C, Lanzilotta M, Palomba R, Storelli E, Zampi M (2014) Topical pharmacologic approach with 5% lidocaine medicated plaster in the treatment of localized neuropathic pain. Minerva Med 105:515–527
Ramos Campos EV, Silva de Melo NF, Guilherme VA, de Paula E, Rosa AH, de Araujo DR, Fraceto LF (2013) Preparation and characterization of poly (epsilon-caprolactone) nanospheres containing the local anesthetic lidocaine. J Pharm Sci 102:215–226
Sabatowski R, Bosl I, Konig S, Buchheister B, Meier T, Baron R (2017) Treatment of postherpetic neuralgia with 5% lidocaine medicated plaster in elderly patients - subgroup analyses from three European clinical trials. Curr Med Res Opin 33:595–603
Shen J, Burgess DJ (2012) Accelerated in-vitro release testing methods for extended-release parenteral dosage forms. J Pharm Pharmacol 64:986–996
Shir Y, Seltzer Z (1990) A-fibers mediate mechanical hyperesthesia and allodynia and C-fibers mediate thermal hyperalgesia in a new model of causalgiform pain disorders in rats. Neurosci Lett 115:62–67
Suzuki R, Arai YC, Hamayasu K, Fujita K, Hara K, Yamaguchi T, Sasaguri S (2009) Complex of branched cyclodextrin and lidocaine prolonged the duration of peripheral nerve block. J Anesth 23:295–297
Ta LE, Bieber AJ, Carlton SM, Loprinzi CL, Low PA, Windebank AJ (2010) Transient receptor potential vanilloid 1 is essential for cisplatin-induced heat hyperalgesia in mice. Mol Pain 6:15
Tamburin S, Schweiger V, Magrinelli F, Brugnoli MP, Zanette G, Polati E (2014) Effect of 5% lidocaine medicated plaster on pain intensity and paroxysms in classical trigeminal neuralgia. Ann Pharmacother 48:1521–1524
Tender GC, Li YY, Cui JG (2008) Vanilloid receptor 1-positive neurons mediate thermal hyperalgesia and tactile allodynia. Spine J 8:351–358
Tjolsen A, Berge OG, Hunskaar S, Rosland JH, Hole K (1992) The formalin test: an evaluation of the method. Pain 51:5–17
Uekama K, Hirayama F, Irie T (1998) Cyclodextrin drug carrier systems. Chem Rev 98:2045–2076
Vos BP, Strassman AM, Maciewicz RJ (1994) Behavioral evidence of trigeminal neuropathic pain following chronic constriction injury to the rat’s infraorbital nerve. J Neurosci 14:2708–2723
Wang CF, Pancaro C, Gerner P, Strichartz G (2011) Prolonged suppression of postincisional pain by a slow-release formulation of lidocaine. Anesthesiology 114:135–149
Wei Y, Nedley MP, Bhaduri SB, Bredzinski X, Boddu SH (2015) Masking the bitter taste of injectable lidocaine HCl formulation for dental procedures. AAPS PharmSciTech 16:455–465
You P, Yuan R, Chen C (2017) Design and evaluation of lidocaine- and prilocaine-coloaded nanoparticulate drug delivery systems for topical anesthetic analgesic therapy: a comparison between solid lipid nanoparticles and nanostructured lipid carriers. Drug Des Devel Ther 11:2743–2752
Zakrzewska JM, Palmer J, Morisset V, Giblin GM, Obermann M, Ettlin DA, Cruccu G, Bendtsen L, Estacion M, Derjean D, Waxman SG, Layton G, Gunn K, Tate S, study i (2017) Safety and efficacy of a Nav1.7 selective sodium channel blocker in patients with trigeminal neuralgia: a double-blind, placebo-controlled, randomised withdrawal phase 2a trial. Lancet Neurol 16:291–300
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16:109–110
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brazil (CAPES) - Finance Code 001.
Author information
Authors and Affiliations
Contributions
JGC and RFC conceived and designed research. SBO, EIA, and EG conducted the in vivo experiments. LENF and MFM prepared lidocaine complexed formulation and conducted the in vitro assay. JGC analyzed data and wrote the manuscript. All authors read, revised, and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures were conducted in accordance with the Institutional Committee on the Ethical Use of Animals (authorization no. 1039) and performed in accordance with ethical guidelines, including the policies and recommendations of the International Association for the Study of Pain and according to the recommendations of the Brazilian Agency CONCEA (National Council of Control in Experimental Animals).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary materials
Supplementary Figure 1
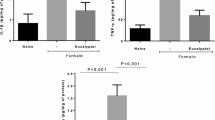
Experimental procedures timeline. In the formalin test animals received plain lidocaine (LDC, 2% in 50 μL), complexed lidocaine (LDC:HP-β-CD, 2% in 50 μL) or vehicle (VEH, 50 μL) into the right upper lip, and after 10 minutes, formalin (FOR, 2.5% in 50 μL) or saline (SAL, 50 μL) in the same site. The grooming response was assessed for 30 min (first phase: 0-3 minutes, second phase: 12-30 min). In the capsaicin test, heat hyperalgesia was assessed (BL), and animals was treated with plain lidocaine (LDC, 2% in 50 μL), complexed lidocaine (LDC:HP-β-CD, 2% in 50 μL) or vehicle (VEH, 50 μL) into the right upper lip, and after 10 minutes, capsaicin (CAP, 3 μg in 50 μL) or saline (SAL, 50 μL) was injected in the same site. Heat hyperalgesia was measured 0.5, 1, 2, 3 and 4 h after CAP/SAL injection. In the Carrageenan test, animals were subjected to the same procedure, but the treatments were performed 150 min after carrageenan (CAR, 100 μg in 50 μL) or saline (SAL, 50 μL) into the right upper lip. Heat and mechanical hyperalgesia were evaluated 5 and 15 days, respectively, after CION, as well as hourly after treatments with plain lidocaine (LDC, 2% in 50 μL), complexed lidocaine (LDC:HP-β-CD, 2% in 50 μL) or vehicle (VEH, 50 μL) into the right upper lip. (PNG 615 kb)
Supplementary Figure 2
Effect of plain lidocaine (LDC) and lidocaine complexed with 2-hydroxypropyl-β-cyclodextrin (LDC:HP-β-CD) in control animals (related to Fig 2). Animals was pre-treated with plain lidocaine (LDC, 2% in 50 μL) or complexed lidocaine (LDC:HP-β-CD, 2% in 50 μL) into the right upper lip, and after 10 minutes, vehicle (VEH, 50 μL) was injected in the same site. The grooming response was assessed for 30 minutes after VEH injection, and the first phase was considered from 0-3 minutes and the second phase was considered from 12-30 minutes. Data are expressed as mean ± SEM. Two-way ANOVA with repeated measures followed by the Bonferroni post hoc test (n=9-11). (PNG 50 kb)
Supplementary Figure 3
Effect of plain lidocaine (LDC) and lidocaine complexed with 2-hydroxypropyl-β-cyclodextrin (LDC:HP-β-CD) in control animals (related to Fig 3). Heat hyperalgesia baseline (BL) was assesses and animals was treated with plain lidocaine (LDC, 2% in 50 μL) or complexed lidocaine (LDC:HP-β-CD, 2% in 50 μL) into the right upper lip, and 10 or 150 minutes later they were treated with capsaicin vehicle (50 μL, Panel A) or with carrageenan vehicle (50 μL, Panel B), respectively. Data are expressed as mean ± SEM. Two-way ANOVA with repeated measures followed by the Bonferroni post hoc test (n=6-10). (PNG 37 kb)
Supplementary Figure 4
Effect of plain lidocaine (LDC) and lidocaine complexed with 2-hydroxypropyl-β-cyclodextrin (LDC:HP-β-CD) in SHAM animals (related to Fig 4). Heat or mechanical hyperalgesia baseline (BL) was assessed and SHAM surgery was performed. The heat and mechanical hyperalgesia was measured 5 or 15 days after surgery, respectively. Animals was treated with plain lidocaine (LDC, 2% in 50 μL) or complexed lidocaine (LDC:HP-β-CD, 2% in 50 μL) into the right upper lip, and heat or mechanical hyperalgesia (Panel A and B, respectively) was assessed for up to 4 hours after treatment. Data are expressed as mean ± SEM. Two-way ANOVA with repeated measures followed by the Bonferroni post hoc test (n=6-10). (PNG 40 kb)
Rights and permissions
About this article
Cite this article
de Oliveira, S.B., Araya, E.I., Gambeta, E. et al. Comparison of antinociceptive effects of plain lidocaine versus lidocaine complexed with hydroxypropyl-β-cyclodextrin in animal models of acute and persistent orofacial pain. Naunyn-Schmiedeberg's Arch Pharmacol 392, 573–583 (2019). https://doi.org/10.1007/s00210-018-01609-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-01609-8