Abstract
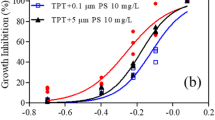
The effect of microplastic adsorption on marine microalgae Tetraselmis suecica, Amphora subtropica, and copepod Pseudodiaptomus annandalei was investigated in the present study. Fluorescence microscopic images were used to evaluate MP interactions with algae and copepods. T. suecica growth rate decreased with effects of 0.1 µm polystyrene exposure to 75 µl/100 ml (0.899 to 0.601 abs), 50 µl/100 ml (0.996 to 0.632 abs) and 25 µl/100 ml (0.996 to 0.632 abs), respectively. On the other hand, at 10th day of experiment, the control T. suecica showed the highest growth rate (0.965 abs), chlorophyll concentration (Chl-‘a' = 21.36 µg/L; Chl-‘b' = 13.65 µg/L), and cell density (3.3 × 106 cells/ml). A marine diatom A. subtropica absorbed 2.0 μm microplastics, and the maximal inhibition rate increased at higher MP concentration until 10th day. The highest MPs (75 μl/100 ml) treatment resulted in decreased growth rate of A. subtropica from 0.163 to 0.096 abs. A. subtropica (without MPs) had the highest lipid concentration of 27.15%, whereas T. suecica had the lowest lipid concentration of 11.2% (without MP). The maximum survival (80%) of P. annandalei was found in control on 15th day whereas on 12th day, the microplastics ingested copepod had the lowest survival rate (0%). On 15th day, the maximum Nauplii Production Rate (NPR) (19.33) female−1 was observed in control, whereas the minimum (17.33) female−1 NPR was observed in copepod ingested with MPs. The maximum lipid production (17.33% without MPs) was reported in control, whereas MPs fed copepods had the lowest lipid production (16%). Long-term exposure to polystyrene microplastics significantly reduced algae growth and chlorophyll concentration and also NPR and lipid concentration rate of copepod. We inferred that microplastic exposure of algae and copepods might results in persistent decreases in ingested carbon biomass over time.
Graphical abstract






Similar content being viewed by others
References
Alimi OS, Farner Budarz J, Hernandez LM, Tufenkji N (2018) Microplastics and nanoplastics in aquatic environments: aggregation, deposition, and enhanced contaminant transport. Environ Sci & Tech 52:1704–1724. https://doi.org/10.1021/acs.est.7b05559
Ananth S, Santhanam P (2011) Laboratory culture and biochemical profile of marine copepod, Macrosetella gracilis (Dana). Aquaculture 12(1):49–55
Andrady AL (2011) Microplastics in the marine environment. Mar Pollut Bll 62(8):1596–1605. https://doi.org/10.1016/j.marpolbul.2011.05.030
Andrady AL, Neal MA (2009) Applications and societal benefits of plastics. Phil Trans R Soc b 364(1526):1977–1984. https://doi.org/10.1098/rstb.2008.0304
Ballent A, Pando S, Purser A, Juliano MF, Thomsen L (2013) Modelled transport of benthic marine microplastic pollution in the Nazaré Canyon. Biogeosciences 10(12):7957–7970. https://doi.org/10.5194/bg-10-7957-2013
Besseling E, Wang B, Lürling M, Koelmans AA (2014) Nanoplastic affects growth of S. obliquus and reproduction of D. magna. Environ Sci Technol 48(20):12336–12343. https://doi.org/10.1021/es503001d.
Bhattacharya P, Lin S, Turner JP, Ke PC (2010) Physical adsorption of charged plastic nanoparticles affects algal photosynthesis. J Phys Chem 114(39):16556–16561. https://doi.org/10.1021/jp1054759
Bonnet D, Richardson A, Harris R, Hirst A, Beaugrand G, de Edwards M, Puelles MLF (2005) An overview of Calanus helgolandicus ecology in European waters. Prog in Oceano 65(1):1–53. https://doi.org/10.1016/j.pocean.2005.02.002
Brown KS (1997) Diversity, disturbance, and sustainable use of Neotropical forests: insects as indicators for conservation monitoring. J Insect Conserv 1(1):25–42
Browne MA, Galloway TS, Thompson RC (2008) Spatial patterns of plastic debris along estuarine shorelines. Environ Sci Technol 44(9):3404–3409. https://doi.org/10.1021/es903784e
Cai L, Hu L, Shi H, Ye J, Zhang Y, Kim H (2018) Effects of inorganic ions and natural organic matter on the aggregation of nanoplastics. Chemosphere 197:142–151. https://doi.org/10.1016/j.chemosphere.2018.01.052
Cole M, Lindeque P, Halsband C, Galloway TS (2011) Microplastics as contaminants in the marine environment. A Review Mar Pollut Bll 62(12):2588–2597. https://doi.org/10.1016/j.marpolbul.2011.09.025
Cole M, Lindeque P, Fileman E, Halsband C, Goodhead R, Moger J, Galloway TS (2013) Microplastic ingestion by zooplankton. Environ Sci Technol 47(12):6646–6655. https://doi.org/10.1021/es400663f
Cole M, Lindeque P, Fileman E, Halsband C, Galloway TS (2015) The impact of polystyrene microplastics on feeding, function and fecundity in the marine copepod Calanus helgolandicus. Environ Sci & Tech 49(2):1130–1137
Costa JP, Santos PS, Duarte AC, Rocha-Santos T (2016) Nano plastics in the environment-sources, fates and effects. Sci Total Environ 566:15–26. https://doi.org/10.1016/j.scitotenv.2016.05.041
Cózar A, Echevarría F, González-Gordillo JI, Irigoien X, Úbeda B, Hernández-León S, Palma ÁT, Navarro S, García-de-Lomas J, Ruiz A, Fernández-de-Puelles ML (2014) Plastic debris in the open ocean. P Natl Acad Sci-Biol 111(28):10239–10244. https://doi.org/10.1073/pnas.1314705111
Davis C. C 1955 The marine and freshwater plankton. Michigan State University.
Folch J, Lees M, Stanley GS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J of Bio Chem 226(1):497–509
Galloway TS, Cole M, Lewis C (2017) Interactions of microplastic debris throughout the marine ecosystem. Nat Ecol Evol 1(5):0116. https://doi.org/10.1038/s41559-017-0116
Gigault J, Ter Halle A, Baudrimont M, Pascal PY, Gauffre F, Phi TL, El Hadri H, Reynaud GB, S, (2018) Current opinion: What is a nanoplastic? Environ Pollut 235:1030–1034. https://doi.org/10.1016/j.envpol.2018.01.024
Gregory MR, Ryan PG (1997) pelagic plastics and other seaborne persistent synthetic debris. A review of Southern Hemisphere perspectives. In Marine Debris (49–66). Springer. New York. NY.49–66. https://doi.org/10.1007/978-1-4613-8486-1_6.
Isobe A, Uchiyama-Matsumoto K, Uchida K, Tokai T (2014) Microplastics in the Southern Ocean. Mar Pollut Bull 114(1):623–626. https://doi.org/10.1016/j.marpolbul.2016.09.037
Jeffrey ST, Humphrey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem Phy 167(2):191–194. https://doi.org/10.1016/S0015-3796(17)30778-3
Kaiser MJ, Attrill MJ, Jennings S, Thomas DN, Barnes DK (2011) Marine ecology processes systems, and impacts. Oxford University Press
Kako S, Isobe A, Magome S, Hinata H, Seino S, Kojima A (2011) Establishment of numerical beach litter hindcast/forecast models: an application to Goto Islands. Japan Mar Pollut Bull 62:293–302. https://doi.org/10.1016/j.marpolbul.2010.10.011
Kako S, Isobe A, Kataoka T, Hinata H (2014) A decadal prediction of the quantity of plastic marine debris littered on beaches of the East Asian marginal seas. Mar Pollut Bull 81:174–184. https://doi.org/10.1016/j.marpolbul.2014.01.057
Kang JK, Kwon OY, Lee KW, Song YK, Shim WJ (2015) Marine neustonic microplastics around the south eastern coast of Korea. Mar Pollut Bull 96(1–2):304–312. https://doi.org/10.1016/j.marpolbul.2015.04.054
Kasturirangan LR (1963) A Key for the Identification of the More Common Planktonic Copepoda: Of Indian Coastal Waters (No. 2). Council of Scientific & Industrial Research.
Koelmans AA, Besseling E, Shim WJ (2015) Nanoplastics in the aquatic environment. Critical review. In Marine Anthropogenic Litter (325–340). Springer, Cham. https://doi.org/10.1007/978-3-319-16510-3.
Kubota M (1994) A mechanism for the accumulation of floating marine debris north of Hawaii. J Phys Oceanogr 24:1059–1064. https://doi.org/10.1175/1520-0485(1994)024%3c1059:AMFTAO%3e2.0.CO;2
Lee KW, Shim WJ, Kwon OY, Kang JH (2013) Size-dependent effects of micro polystyrene particles in the marine copepod Tigriopus japonicus. Environ Sci & Tech 47(19):11278–11283. https://doi.org/10.1021/es401932b
Li J, Qu X, Su L, Zhang W, Yang D, Kolandhasamy P, Shi H (2016) Microplastics in mussels along the coastal waters of China. Environ Pollut 214:177–184. https://doi.org/10.1016/j.envpol.2016.04.012
Li J, Liu H, Chen JP (2018) Microplastics in freshwater systems: A review on occurrence, environmental effects, and methods for microplastics detection. Water Res 137:362–374. https://doi.org/10.1016/j.watres.2017.12.056
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Method Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Long M, Moriceau B, Gallinari M, Lambert C, Huvet A, Raffray J, Soudant P (2015) Interactions between microplastics and phytoplankton aggregates: Impact on their respective fates. Mar Chem 175:39–46. https://doi.org/10.1016/j.marchem.2015.04.003
Lusher A (2015) Microplastics in the marine environment: distribution, interactions and effects. In Marine Anthropogenic Litter (245–307): Springer Cham 245–307. https://doi.org/10.1007/978-3-319-16510-3.
Mao Y, Ai H, Chen Y, Zhang Z, Zeng P, Kang L, Li W, Gu W, He Q, Li H (2018) Phytoplankton response to polystyrene microplastics: perspective from an entire growth period. Chemosphere 208:59–68. https://doi.org/10.1016/j.chemosphere.2018.05.170
Marshall S. M, Orr A. P (1955) Experimental feeding of the copepod Calanus finmarchicus (Gunner) on phytoplankton cultures labelled with radioactive carbon (14C). Deep-Sea Research (1953) (Supplement).
Martínez-Fernández E, Acosta-Salmón H, Southgate PC (2006) The nutritional value of seven species of tropical microalgae for black-lip pearl oyster (Pinctada margaritifera, L.) larvae. Aquaculture. 257: (1–4)491–503. https://doi.org/10.1016/j.aquaculture.2006.03.022.
Maximenko N, Hafner J, Niiler P (2012) Pathways of marine debris derived from trajectories of Lagrangian drifters. Mar Pollut Bull 65(1–3):51–62. https://doi.org/10.1016/j.marpolbul.2011.04.016
Mazurais D, Ernande B, Quazuguel P, Severe A, Huelvan C, Madec L, Mouchel O, Soudant P, Robbens J, Huvet A, Zambonino-Infante J (2015) Evaluation of the impact of polyethylene microbeads ingestion in European sea bass (Dicentrarchus labrax) larvae. Mar Environ Res 112:78–85. https://doi.org/10.1016/j.marenvres.2015.09.009
Passow U (2002) Production of transparent exopolymer particles (TEP) by phyto- and bacterioplankton. Mar Ecol Prog Ser 236:1–12. https://doi.org/10.3354/meps236001
Renaud SM, Thinh LV, Lambrinidis G, Parry DL (2002) Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 211(1–4):195–214. https://doi.org/10.1016/S0044-8486(01)00875-4
Sjollema SB, Redondo-Hasselerharm P, Leslie HA, Kraa MH, Vethaak AD (2016) Do plastic particles affect microalgal photosynthesis and growth? Aquat Toxicol 170:259–261. https://doi.org/10.1016/j.aquatox.2015.12.002
Smolders E, Degryse F (2002) Fate and effect of zinc from tire debris in soil. Environ Sci & Tech 36(17):3706–3710. https://doi.org/10.1021/es025567p
Staats N, Stal LJ, Mur LR (2000) Exopolysaccharide production by the epipelic diatom Cylindrotheca closterium: effects of nutrient conditions. J Exp Mar Biol Ecol 249(1):13–27. https://doi.org/10.1016/S0022-0981(00)00166-0
Thompson PA, Guo M, Harrison PJ (1993) The influence of irradiance on the biochemical composition of three phytoplankton species and their nutritional value for larvae of the Pacific oyster (Crassostrea gigas). Mar Biol 117(2):259–268. https://doi.org/10.1016/j.marpolbul.2010.05.026
Valenzuela S (2013) Unpacking the use of social media for protest behavior: The roles of information, opinion expression, and activism. Am Behav Sci 57(7):920–942. https://doi.org/10.1177/0002764213479375
Valenzuela-Espinoza E, Millán-Núñez R, Núñez-Cebrero F (2002) Protein, carbohydrate, lipid and chlorophyll a content in Isochrysis aff. galbana (clone T-Iso) cultured with a low cost alternative to the f/2 medium. Aquac Eng.25(4), 207–216.
Wright SL, Thompson RC, Galloway TS (2013) The physical impacts of microplastics on marine organisms: a review. Environ Pollut 178:483–492. https://doi.org/10.1016/j.envpol.2013.02.031
Xu ZB, Yan XJ, Pei LQ, Luo QJ, Xu JL (2008) Changes in fatty acids and sterols during batch growth of Pavlova viridis in photobioreactor. J Appl Phycol 20:237–243
Yoon TP, Ischay MA, Du J (2010) Visible light photocatalysis as a greener approach to photochemical synthesis. Nat Chem 2(7):527
Zarfl C, Matthies M (2010) Are marine plastic particles transport vectors for organic pollutants to the Arctic? Mar Pollut Bull 60(10):1810–1814
Acknowledgements
The authors are extremely thankful to the Bharathidasan University for providing the necessary facilities. The Ministry of Environment, Forest and Climate Change (MoEF & CC), Govt. of India, New Delhi, for providing financial support through a research Project (F. No. 220180/06/2015-RE (Tax), 05.10.2016). The Department of Biotechnology (DBT), Govt. of India, is gratefully acknowledged with thanks for the copepod culture facility provided (BT/PR 5856/AAQ/3/598/2012).
Funding
Ministry of Environment, Forest and Climate Change (F. No. 220180/06/2015-RE (Tax), 5th October 2016) to Santhanam Perumal.
Author information
Authors and Affiliations
Contributions
PR: conceptualization, methodology, resources, writing—original draft, writing—review and editing. PS: conceptualization, methodology, writing—review and editing. SSP, MD, and AA: data analysis. KND, SA, and JR: writing—review and editing and PP: writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raju, P., Santhanam, P., Pandian, S.S. et al. Impact of polystyrene microplastics on major marine primary (phytoplankton) and secondary producers (copepod). Arch Microbiol 204, 84 (2022). https://doi.org/10.1007/s00203-021-02697-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02697-6