Abstract
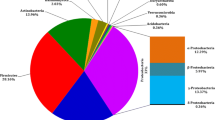
Earthworms are considered as a rich microhabitat for the growth and proliferation of diverse soil microorganisms. Hence, earthworms’ associated bacteria attracted interest due to their high metabolic profiles and benefits to soil fertility and plant growth. In this study, we aimed to isolate for the first-time aerobic bacteria present in the chloragogenous tissue of the earthworm Apporectodea molleri and test their Plant Growth-Promoting abilities and their resistance to heavy metals (Mn, Zn, Cu, Cd, and Ni). The 16S rRNA gene sequencing revealed the affiliation of the fifteen isolates to six main bacterial genera: Enterobacter, Citrobacter, Aeromonas, Pseudomonas, Bacillus, Terribacillus. These strains displayed different plant growth promoting traits (e.g., indole-3-acetic acid IAA, siderophores, nitrogen fixation, phosphate, and potassium solubilization), in addition, they were able to resist differently to heavy metals. Bacillus strains were most effective as three strains, namely B. subtilis strain TC34; B. circulans strain TC7 and Bacillus sp. strain TC10, were positive to all PGP traits and resisted to all heavy metals. This study illustrates the potential of bacteria from the chloragogenous tissue to exhibit multiple properties, which can be related to the functional feature of this tissue to stock metabolites and neutralize toxic elements.
Similar content being viewed by others
Data availability
The data underlying the funding are fully available within the manuscript.
References
Banerjee A, Biswas JK, Pant D et al (2019) Enteric bacteria from the earthworm (Metaphire posthuma) promote plant growth and remediate toxic trace elements. J Environ Manage 250:109530. https://doi.org/10.1016/j.jenvman.2019.109530
Biswas JK, Banerjee A, Rai M et al (2018) Potential application of selected metal resistant phosphate solubilizing bacteria isolated from the gut of earthworm (Metaphire posthuma) in plant growth promotion. Geoderma 330:117–124. https://doi.org/10.1016/j.geoderma.2018.05.034
Brown GG, Doube BM (2004) Functional interactions between earthworms, microorganisms, organic matter, and plants. In: Edwards CA (ed) Earthworm ecology, 2nd edn. CRC Press LLC, Boca Raton, pp 213–239
Brown AE, Smith HR (2010) Benson’s microbiological applications: laboratory manual in general microbiology, 14th edn. McGraw-Hill Education
Byzov BA, Nechitaylo TY, Bumazhkin BK et al (2009) Culturable microorganisms from the earthworm digestive tract. Microbiology 78:360–368. https://doi.org/10.1134/S0026261709030151
Courtois P, Rorat A, Lemiere S et al (2021) Accumulation, speciation and localization of silver nanoparticles in the earthworm Eisenia fetida. Environ Sci Pollut Res 28:3756–3765. https://doi.org/10.1007/s11356-020-08548-z
Das A, Osborne JW (2018) Bioremediation of heavy metals. In: Gothandam KM, Ranjan S, Dasgupta N et al (eds) Nanotechnology, food security and water treatment. Springer International Publishing, pp 277–311
Drake HL, Horn MA (2006) Earthworms as a transient heaven for terrestrial denitrifying microbes: a review. Eng Life Sci 6:261–265. https://doi.org/10.1002/elsc.200620126
Drake HL, Schramm A, Horn MA (2006) Earthworm gut microbial biomes: their importance to soil microorganisms, denitrification, and the terrestrial production of the greenhouse gas N2O. In: Varma A, König H (eds) Intestinal microorganisms of termites and other invertebrates. Springer-Verlag, Berlin Heidelberg, pp 65–87
Fischer E (1978) DOPA peroxidase activity in the chloragogen cells of the earthworm, Lumbricus terrestris L. Acta Histochem 63:219–223. https://doi.org/10.1016/S0065-1281(78)80028-2
Fischer E, Molnár L (1992) Environmental aspects of the chloragogenous tissue of earthworms. Soil Biol Biochem 24:1723–1727. https://doi.org/10.1016/0038-0717(92)90177-Y
Fukushima T, Allred BE, Raymond KN (2014) Direct evidence of iron uptake by the Gram-positive siderophore-shuttle mechanism without iron reduction. ACS Chem Biol 9:2092–2100. https://doi.org/10.1021/cb500319n
Gordon SA, Weber RP (1951) Colorimetric estimation of indoleacetic acid. Plant Physiol 26:192–195. https://doi.org/10.1104/pp.26.1.192
Ha TTT, Lam TT, Huyen NT, Canh NX (2018) Characterization and identification of nitrogen- fixing bacteria isolated from agricultural soil. Vietnam J Sci Technol 60:48–54. https://doi.org/10.31276/VJSTE.60(3).48
Halda-Alija L (2003) Identification of indole-3-acetic acid producing freshwater wetland rhizosphere bacteria associated with Juncus effusus L. Can J Microbiol 49:781–787. https://doi.org/10.1139/w03-103
Hepperle D (2011) DNA Dragon 1.4. 1–DNA sequence contig assembler software. https://www.dna-dragon.com/. Accessed 23 Apr 2019
Huang H, Zhao Y, Fan L et al (2020) Improvement of manganese phytoremediation by Broussonetia papyrifera with two plant growth promoting (PGP) Bacillus species. Chemosphere 260:127614. https://doi.org/10.1016/j.chemosphere.2020.127614
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Kramer J, Özkaya Ö, Kümmerli R (2020) Bacterial siderophores in community and host interactions. Nat Rev Microbiol 18:152–163. https://doi.org/10.1038/s41579-019-0284-4
Kumar SV, Menon S, Agarwal H, Gopalakrishnan D (2017) Characterization and optimization of bacterium isolated from soil samples for the production of siderophores. Bioresour Technol 3:434–439. https://doi.org/10.1016/j.reffit.2017.04.004
Kumar S, Stecher G, Li M et al (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Kuzyakov Y, Blagodatskaya E (2015) Microbial hotspots and hot moments in soil: concept & review. Soil Biol Biochem 83:184–199. https://doi.org/10.1016/j.soilbio.2015.01.025
Li H-B, Singh RK, Singh P et al (2017) Genetic diversity of nitrogen-fixing and plant growth promoting Pseudomonas species isolated from sugarcane rhizosphere. Front Microbiol 8:1–20. https://doi.org/10.3389/fmicb.2017.01268
Lopes R, Tsui S, Gonçalves PJRO, de Queiroz MV (2018) A look into a multifunctional toolbox: endophytic Bacillus species provide broad and underexploited benefits for plants. World J Microbiol Biotechnol 34:94. https://doi.org/10.1007/s11274-018-2479-7
Lynne AM, Haarmann D, Louden BC (2011) Use of blue agar CAS assay for siderophore detection. J Microbiol Biol Educ 12:51–53. https://doi.org/10.1128/jmbe.v12i1.249
Medina-Sauza RM, Álvarez-Jiménez M, Delhal A et al (2019) Earthworms building up soil microbiota, a review. Front Environ Sci 7:1–20. https://doi.org/10.3389/fenvs.2019.00081
Mudziwapasi R, Mlambo SS, Chigu NL et al (2016) Isolation and molecular characterization of bacteria from the gut of Eisenia fetida for biodegradation of 4,4 DDT. J Appl Biol Biotechnol. https://doi.org/10.7324/JABB.2016.40507
Pandey S, Gupta S (2020) Evaluation of Pseudomonas sp. for its multifarious plant growth promoting potential and its ability to alleviate biotic and abiotic stress in tomato (Solanum lycopersicum) plants. Sci Rep 10:20951. https://doi.org/10.1038/s41598-020-77850-0
Puga-Freitas R, Abbad S, Gigon A et al (2012) Control of cultivable IAA-producing bacteria by the plant Arabidopsis thaliana and the earthworm Aporrectodea caliginosa. Appl Environ Soil Sci 2012:1–5. https://doi.org/10.1155/2012/307415
Roubalová R, Procházková P, Hanč A et al (2019) Mutual interactions of E. andrei earthworm and pathogens during the process of vermicomposting. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-019-04329-5
Samanta TT, Das A (2016) Isolation, identification, and characterization of gut microflora of Perionyx excavatus collected from Midnapore, West Bengal. J Basic Microbiol 56:286–293. https://doi.org/10.1002/jobm.201500480
Sapkota R, Santos S, Farias P et al (2020) Insights into the earthworm gut multi-kingdom microbial communities. Sci Total Environ 727:138301. https://doi.org/10.1016/j.scitotenv.2020.138301
Schramm A, Davidson SK, Dodsworth JA et al (2003) Acidovorax-like symbionts in the nephridia of earthworms. Environ Microbiol 5:804–809. https://doi.org/10.1046/j.1462-2920.2003.00474.x
Sessitsch A, Kuffner M, Kidd P et al (2013) The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol Biochem 60:182–194. https://doi.org/10.1016/j.soilbio.2013.01.012
Singh A, Singh DP, Tiwari R et al (2015) Taxonomic and functional annotation of gut bacterial communities of Eisenia foetida and Perionyx excavatus. Microbiol Res 175:48–56. https://doi.org/10.1016/j.micres.2015.03.003
Singh A, Tiwari R, Sharma A et al (2016) Taxonomic and functional diversity of the culturable microbiomes of epigeic earthworms and their prospects in agriculture. J Basic Microbiol 56:1009–1020. https://doi.org/10.1002/jobm.201500779
Stackebrandt E, Goebel BM (1994) Taxonomic note: a place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. Int J Syst Bacteriol 44:846–849. https://doi.org/10.1099/00207713-44-4-846
Tillinghast EK (1968) Variations in blood and coelomic fluid ammonia and urea levels in the earthworm Lumbricus terrestris L. Comp Biochem Physiol 24:621–623. https://doi.org/10.1016/0010-406X(68)91014-1
Verma A, Ali D, Farooq M et al (2011) Expression and inducibility of endosulfan metabolizing gene in Rhodococcus strain isolated from earthworm gut microflora for its application in bioremediation. Bioresour Technol 102:2979–2984. https://doi.org/10.1016/j.biortech.2010.10.005
Vogel J, Seifert G (1992) Histological changes in the chloragogen tissue of the earthworm Eisenia fetida after administration of sublethal concentrations of different fluorides. J Invertebr Pathol 60:192–196. https://doi.org/10.1016/0022-2011(92)90096-M
Wani KA, Mamta SR, Lone RA (2017) Earthworms and associated microbiome: natural boosters for agro-ecosystems. In: Kumar V, Kumar M, Sharma S, Prasad R (eds) Probiotics in agroecosystem. Springer Singapore, Singapore, pp 469–489
Acknowledgements
This work was done partially at Ataturk University, Faculty of Agriculture, Department of Soil Science, Erzurum, Turkey. We express our gratitude to all the department staff for their help and support. Our thanks to Prof. Dr. Jorge Dominguez for performing the earthworm molecular identification sampled from Rabat – Morocco. This work was supported by the National Center for Scientific and Technical Research (CNRST—Morocco) under the Research Excellence Scholarship Program.
Author information
Authors and Affiliations
Contributions
SH: conceived and designed the experience, conducted the experiences, and collected data, wrote the manuscript; LY: conceived, designed and contributed during the experiences; SB: conceived and designed the experience, supervised the work, and contribute to the final form of the manuscript; MR: helped in the data analysis and contribute in the final form of the manuscript; AEH: conceived the original idea and contribute in the final form of the manuscript; SA: conceived and designed the experience, supervised the work, and contribute in the final form of the manuscript; all authors provided critical feedback and helped shape the research, analysis, and manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We have no conflict of interest to declare.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Houida, S., Yakkou, L., Bilen, S. et al. Taxonomic and functional characteristics of aerobic bacteria isolated from the chloragogenous tissue of the earthworm Aporrectodea molleri. Arch Microbiol 203, 4805–4812 (2021). https://doi.org/10.1007/s00203-021-02396-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02396-2