Abstract
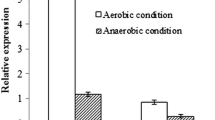
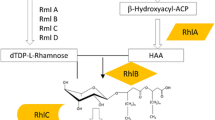
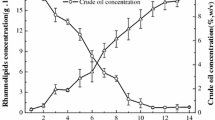
Pseudomonas spp. are the main producers of rhamnolipids. These products have applications in pharmaceuticals, cosmetics, food industry and bioremediation. The biosynthesis of rhamnolipids is influenced by nutrient composition, pH and temperature. In this study, the impact of nutrients on the expression levels of rhamnolipid synthesis genes was evaluated in P. aeruginosa ATCC 15442. Glucose and glycerol were used as carbon sources; while, NaNO3, NH4NO3 and yeast extract/peptone were employed as nitrogen sources. The effect of different concentrations of Fe2+ and Fe3+ on rhamnolipid synthesis genes was also evaluated. Highest biosurfactant production was obtained in minimal medium supplemented with glucose, NaNO3 and Fe2+. Two rhamnolipid synthesis genes, rhlA and rhlB, were amplified with PCR. CapLC ESI–Ion trap-MS/MS detected only mono-rhamnolipid Rha–C10–C10 in the extract. Although similar induction levels were recorded in the presence of 0.05 g/L iron ions, the presence of Fe2+ resulted in higher expression levels than Fe3+ at concentrations equivalent to 0.025 and 0.075 g/L.







Similar content being viewed by others
References
Agarry SE, Salam K, Olatunde A, Aremu MO (2015) Biosurfactant production by indigenous Pseudomonas and Bacillus species isolated from auto-mechanic soil environment towards microbial enhanced oil recovery. EJET 3:27–39
Amaral PF, Coelho MA, Marrucho IM, Coutinho JA (2010) Biosurfactants from yeasts: characteristics, production and application. Adv exp Med Bio 672:236–249. https://doi.org/10.1007/978-1-4419-5979-9_18
Banin E, Vasil ML, Greenbery EP (2005) Iron and Pseudomonas aeruginosa biofilm formation. PNAS 102:11076–11081. https://doi.org/10.1073/pnas.0504266102
Costa SGVAO, Nitschke M, Haddad R, Eberlin MN, Contiero J (2006) Production of Pseudomonas aeruginosa LBI rhamnolipids following growth on Brazilian native oils. Process Biochem 41:483–488. https://doi.org/10.1016/j.procbio.2005.07.002
Déziel E, Lépine F, Dennie D, Boismenu D, Mamer OA, Villemur R (1999) Liquid chromatography/mass spectrometry analysis of mixtures of rhamnolipids produced by Pseudomonas aeruginosa strain 57RP grown on mannitol or naphthalene. Biochim Biophys Acta 1440:244–252. https://doi.org/10.1016/S1388-1981(99)00129-8
Du J, Zhang A, Zhang X, Si X, Cao J (2019) Comparative analysis of rhamnolipid congener synthesis in neotype Pseudomonas aeruginosa ATCC 10145 and two marine isolates. Bioresour Technol 286:121380. https://doi.org/10.1016/j.biortech.2019.121380
Ehinmitola EO, Funmilayo Aransiola E, Adeahbo OP (2018) Comparative study of various carbon sources on rhamnolipid production. S Afr J Chem Eng 26:42–48. https://doi.org/10.1016/j.sajce.2018.09.001
Eraqi WA, Yassin AS, Ali AE, Amin MA (2016) Utilization of crude glycerol as a substrate for the production of rhamnolipid by Pseudomonas aeruginosa. Biotechnol Res Int. https://doi.org/10.1155/2016/3464509
Funston SJ, Tsaousi K, Rudden M, Smyth TJ, Stevenson PS, Marchant R, Banat IM (2016) Characterising rhamnolipid production in Burkholderia thailandensis E264, a non-pathogenic producer. Appl Microbiol Biotechnol 100:7945–7956. https://doi.org/10.1007/s00253-016-7564-y
Glick R, Gilmour C, Tremblay J, Satanower S, Avidan O, Déziel E, Greenberg EP, Poole K, Banin E (2010) Increase in rhamnolipid synthesis under iron-limiting conditions influences surface motility and biofilm formation in Pseudomonas aeruginosa. J Bacteriol 192:2973–2980. https://doi.org/10.1128/JB.01601-09
Gudiña EJ, Rodrigues AI, Alves E, Rosario Domingues M, Teixeira JA, Rodrigues LR (2015) Bioconversion of agro-industrial by-products in rhamnolipids toward applications in enhanced oil recovery and bioremediation. Bioresour Technol 177:87–93. https://doi.org/10.1016/j.biortech.2014.11.069
Guerra-Santos L, Kappeli O, Fiecheter A (1984) Pseudomonas aeruginosa biosurfactant production in continuous culture with glucose as carbon source. Appl Environ Microbiol 48:301–305
Gunther NW IV, Nunez A, Fett W, Solaiman DKY (2005) Production of Rhamnolipids by Pseudomonas chloroaphis, a non-pathogenic bacterium. Appl Environ Microbiol 71:2288–2293. https://doi.org/10.1128/AEM.71.5.2288-2293.2005
Haba E, Pinazo A, Juaregui O, Espuny MJ, Infante MR, Manresa A (2003) Physicochemical characterization and antimicrobial properties of rhamnolipids produced by Pseudomonas aeruginosa 47T2 NCBIM 40044. Biotechnol bioeng 81:316–322. https://doi.org/10.1002/bit.10474
Heyd M, Kohnert A, Nusser M, Kirschhöfer F, Brenner-Weiss G, Berensmeir S (2008) Development and trends of biosurfactant analysis and purification using rhamnolipids as an example. Anal Bioanal Chem 391:1579–1590. https://doi.org/10.1007/s00216-007-1828-4
İkizler B, Arslan G, Kipcak E, Dirik C, Çelenk D, Aktuğlu T, Helvacı ŞŞ, Peker S (2017) Surface adsorption and spontaneous aggregation of rhamnolipid mixtures in aqueous solutions. Colloids Surf A Physicochem Eng Asp 519:125–136. https://doi.org/10.1016/j.colsurfa.2016.06.056
Kim BJ, Park JH, Park TH, Bronstein PA, Schneider DJ, Cartinhour SW, Shuler ML (2009) Effect of iron concentration on the growth rate of Pseudomonas syringae and the expression of virulence factors in hrp-Inducing Minimal medium. Appl Environ Microbiol 75:2720–2726. https://doi.org/10.1128/AEM.02738-08
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Maqsood MI, Jamal A (2011) Factors affecting the rhamnolipid biosurfactant production. Pak J Biotechnol 8:1–5
Medina G, Juárez K, Valderrama B, Soberón-Chávez G (2003) Mechanism of Pseudomonas aeruginosa RhlR Transcriptional Regulation of the rhlAB Promoter. J Bacteriol 185:5976–5983. https://doi.org/10.1128/JB.185.20.5976-5983.2003
Monteiro SA, Sassaki GL, de Souza LM, Meira JA, de Araújo JM, Mitchell DA, Ramos LP, Krieger N (2007) Molecular and structural characterization of the biosurfactant produced by Pseudomonas aeruginosa DAUPE 614. Chem Phys Lipids 147:1–13. https://doi.org/10.1016/j.chemphyslip.2007.02.001
Moussa TAA, Mohamed MS, Samak N (2014) Production and characterization of Di-rhamnolipid produced by Pseudomonas aeruginosa TMN. Braz J Chem Eng 31:867–880. https://doi.org/10.1590/0104-6632.20140314s00002473
Nickzad A, Guertin C, Deziel E (2018) Culture mediun optimization for production of rhamnolipids by Burkholderia glumae. Colloids Interfaces 2:49. https://doi.org/10.3390/colloids2040049
Nurfarahin AH, Mohamed MS, Phang LY (2018) Culture medium development for microbial-derived surfactants production—an overview. Molecules 23(5):1049. https://doi.org/10.3390/molecules23051049
Parthasarathi R, Sivakumaar PK (2009) Effect of different carbon sources on the production of biosurfactant by Pseudomonas fluorescens isolated from mangrove forests (Pichavaram), Tamil Nadu. India GJER 3:99–101
Praveesh BV, Soniyamby AR, Mariappan C, Kavithakumari P, Palaniswamy M, Lalitha S (2011) Biosurfactant production by Pseudomonas sp. from soil using whey as carbon source. NY Sci J 4:99–103
Rashid NFM, Azmi MMAF, Amirul ASA, Abdul Wahid ME, Bhubalan K (2015) Simultaneous production of biopolymer and biosurfactant by genetically modified Pseudomonas aeruginosa UMTKB-5. Int Proc Chem Biol Environ Eng 90:16–21
Reis RS, Rererira AG, Freire DM (2011) Gene regulation of rhamnolipid production in Pseudomonas aeruginosa—a review. Bioresour Technol 102:6377–6384. https://doi.org/10.1016/j.biortech.2011.03.074
Rudden M, Tsaousi K, Marchant R, Banat IM, Smith TJ (2015) Development and validation of an ultra-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS) method for the quantitative determination of rhamnolipid congeners. Appl microbiol biotechnol 99:9177–9187. https://doi.org/10.1007/s00253-015-6837-1
Santa Anna LM, Sebastian GV, Menezes EP, Alves TLM, Santos AS, Periera N Jr, Freire DMG (2002) Production of biosurfactants from Pseudomonas aeruginosa PA1 isolated in oil environments. Braz J Chem Eng 19:159–166. https://doi.org/10.1590/S0104-66322002000200011
Sekhon Randhawa KK, Kamaljeet K, Rahman Pattanathu KSM (2014) Rhamnolipid biosurfactants—past, present, and future scenario of global market. Front Microbiol 5:454. https://doi.org/10.3389/fmicb.2014.00454
Sobrinho HBS, Luna JM, Rufino RD, Lucia A, Porto F, Sarubbo L (2014) Biourfactants: classification, Properties and Environmental Applications. In: Govil JN (ed) Recent Developments in Biotechnology. Studium Press LLC, Houston, pp 1–29
Tavares LF, Silva PM, Junqueira M, Mariano DC, Nogueira FC, Domont GB, Freire DM, Neves BC (2013) Characterization of rhamnolipids produced by wild-type and engineered Burkholderia kururiensis. Appl Microbiol Biotechnol 97:1909–1921. https://doi.org/10.1007/s00253-012-4454-9
Tomar GS, Srinikethan G (2016) Studies on production of biosurfactant from Pseudomonas aeruginosa (MTCC7815) & its application in microbial enhanced oil recovery. Res J Chem Environ Sci 4:84–91
Twigg MS, Tripathi L, Zompra A, Salek K, Irorere VU, Gutierrez T, Spyroulias GA (2018) Identification and characterisation of short chain rhamnolipid production in a previously un investigated, non pathogenic marine pseudomonad. Appl Microbiol Biotechnol 102:8537–8549. https://doi.org/10.1007/s00253-018-9202-3
Wittgens A, Kovacic F, Müller MM, Gerlitzki M, Santiago-Schübel B, Hofmann D, Tiso T, Blank LM, Henkel M, Hausmann R, Syldatk C, Wilhelm S, Rosenau F (2017) Novel insights into biosynthesis and uptake of rhamnolipids and their precursors. Appl Microbio Biotechnol 101:2865–2878. https://doi.org/10.1007/s00253-016-8041-3
Wu LM, Lai L, Lu Q, Mei P, Wang YQ, Cheng L, Liu Y (2019) Comparative studies on the surface/interface properties and aggregation behavior of mono-rhamnolipid and di-rhamnolipid. Colloids Surf B Biointerfaces 181:593–601. https://doi.org/10.1016/j.colsurfb.2019.06.012
Zhang YI, Miller RM (1992) Enhanced octadecane dispersion and biodegradation by a Pseudomonas rhamnolipid surfactant (biosurfactant). Appl Environ Microbiol 5:3276–3282
Zhao F, Jiang H, Sun H, Liu C, Han S, Zhang Y (2019) Production of rhamnolipids with different proportions of mono-rhamnolipids using crude glycerol and a comparison of their application potential for oil recovery from oily sludge. RSC Adv 9:2885–2891. https://doi.org/10.1039/C8RA09351B
Zhao F, Shi R, Ma F, Han S, Zhang Y (2018) Oxygen effects on rhamnolipids production by Pseudomonas aeruginosa. Microb Cell fact 17:39. https://doi.org/10.1186/s12934-018-0888-9
Acknowledgements
The authors acknowledge the personals of Ege Microbiological Analysis Laboratory (EGEMIKAL) for providing Pseudomonas aeruginosa ATCC 15442.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shatila, F., Diallo, M.M., Şahar, U. et al. The effect of carbon, nitrogen and iron ions on mono-rhamnolipid production and rhamnolipid synthesis gene expression by Pseudomonas aeruginosa ATCC 15442. Arch Microbiol 202, 1407–1417 (2020). https://doi.org/10.1007/s00203-020-01857-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-01857-4